类异戊二烯(Isoprenoids)在自然界中无处不在,包括古细菌、细菌、真核生物等,是种类最多的一类次生代谢产物,它们是细胞一级和二级代谢的组成部分,在信号转导、适应气候、繁殖及防御机制等方面发挥作用[1,2]。类异戊二烯化合物不仅参与植物生长发育和环境应答等重要的生理过程,而且在制药、天然乳胶、香料和有机合成领域也有应用[3]。负责形成和修饰超过六万种类异戊二烯化合物的是异戊烯基转移酶(Prenyltransferase,PT),又称类异戊二烯基焦磷酸合酶(Isoprenyl diphosphate synthases,IPPS或IDS),广泛存在于生物界[4]。异戊烯基转移酶可分为顺式异戊烯基转移酶(Cis-prenyltransferase)和反式异戊烯基转移酶(Trans-prenyltransferase)两个类型。顺式异戊烯基转移酶通常催化合成碳骨架长度大于C50的聚异戊二烯,而反式异戊烯基转移酶催化合成碳骨架长度小于C50的类异戊二烯化合物[5],前者所得产物表现出混合的双反式-多顺式或三反式-多顺式立体异构,后者所得产物则表现出反式立体异构[6]。聚异戊二烯(Polyisoprenoid)及其衍生物具有多种生物学功能[7],如叶绿体中含量丰富的聚戊烯醇(Polyprenols)会影响光合效率,多萜醇(Dolichols)是蛋白质糖基化过程中必需的糖载体,有的聚异戊二烯具有调节膜性质的能力,可作为蛋白质异戊酮的供体[8]。目前对植物细胞内聚异戊二烯合成途径及其功能的认识还相当有限,并且对聚异戊二烯生物合成的调控机制还不清楚。异戊烯转移酶不仅能催化产生一个C-C键(N-C键),还能在终产物中引入一个与类异戊二烯化合物生物活性有关的双键[4]。根据生成的碳链长度,反式异戊烯基转移酶又可分为短链异戊烯基转移酶(C10-25)、中链异戊烯基转移酶(C30-35)和长链异戊烯基转移酶(C40-50)[9]。短链异戊烯基转移酶有香叶基焦磷酸合酶(Geranyl diphosphate synthase,GPPS,C10)、法尼基焦磷酸合酶(Farnesyl diphosphate synthase,FPPS,C15)、香叶基香叶基焦磷酸合酶(Geranylgeranyl diphosphate synthase,GGPPS,C20)和香叶基法尼基焦磷酸合酶(Geranylfarnesyl diphosphate synthase,GFPPS,C25),它们各自的产物GPP、FPP、GGPP和GFPP可作为几种下游酶,如萜类合成酶(Terpene synthase,TPS)、角鲨烯合成酶(Squalene synthase,SQS)及八氢番茄红素合成酶(Phytoene synthase,PSY)等的底物,生成萜烯化合物(Terpenoids)、角鲨烯(Squalene)及八氢番茄红素(Phytoene)等,这些异戊烯基转移酶是类异戊二烯生物合成的重要分支点[10]。中链异戊烯基转移酶包括细菌所具有的六异戊烯基二磷酸合酶(HexPPS)、七异戊烯基二磷酸合酶(HepPPS)等。长链异戊烯基转移酶催化合成长度超过40个C的反式化合物,此类酶需要异戊烯载体蛋白(Prenyl carrier protein)才能把疏水反应产物转移出反应位点[11]。因植物异戊烯基转移酶大多为多次跨膜的膜结合蛋白,且植物遗传背景不清楚,克隆相关基因并进行外源表达有一定的难度,因此在分子水平上的研究报道甚少,但在微生物中异戊烯基转移酶的研究进展较快,大多进行了克隆及功能鉴定,个别酶类还完成了X衍射三维晶体结构分析[12]。由于植物体中类异戊二烯化合物的形成主要依赖异戊烯基转移酶及其基因,因此对异戊烯基转移酶及其基因家族的研究不仅有助于解析类异戊二烯化合物生物合成与调控的机制,还可以为研究合成有生物活性的类异戊二烯化合物提供新的路径[13]。
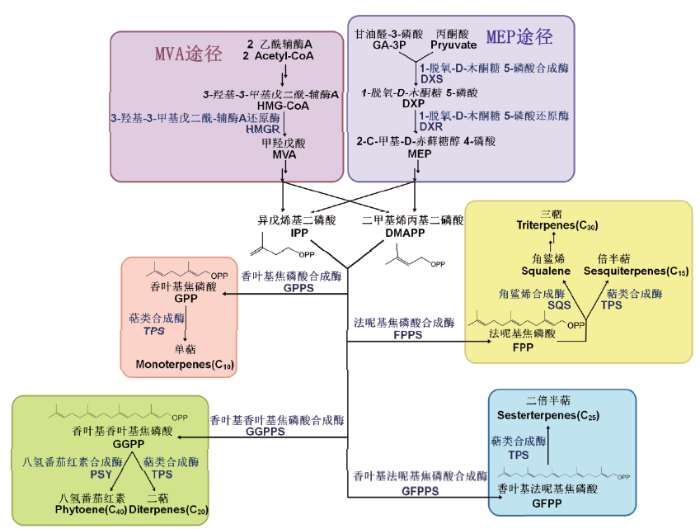
1 植物类异戊二烯生物合成途径
异戊烯基二磷酸(Isopentenyl diphosphate,IPP)和二甲丙烯焦磷酸(Dimethylallyl diphosphate,DMAPP)是所有萜烯化合物生物合成的C5分子前体。它们来源于两种不同的代谢途径,即甲羟戊酸(Mevalonate-independent,MVA)途径和磷酸甲基赤藓糖(Methyl-erythritol 4-phosphate,MEP)途径[14,15,16]。其中,MVA途径是以乙酰辅酶A为底物,在植物细胞质中主要参与倍半萜烯、油菜素内酯、甾醇、三萜等的合成[17,18];而MEP途径是以丙酮酸或磷酸甘油醛为底物,在植物质体中主要参与合成赤霉素、脱落酸、叶绿素、类胡萝卜素、单萜、二萜等物质[19,20]。IPP在IPP异构酶(Isopentenyl diphosphate isomerase,IPPI)的作用下可以转化成为DMAPP,在多种异戊烯基转移酶的作用下发生缩合反应。IPP和DMAPP在异戊烯基转移酶的作用下可形成二聚体、三聚体、四聚体或五聚体等多聚体,分别为香叶基焦磷酸(Geranyl diphosphate,GPP,C10)、法尼基焦磷酸(Farnesyl diphosphate,FPP,C15)、香叶基香叶基焦磷酸(Geranylgeranyl diphosphate,GGPP,C20)和香叶基法呢基焦磷酸(Geranylfarnesyl diphosphate,GFPP,C25)[14,15]。这些异戊烯基聚合物作为中间体在萜烯合成酶(Terpene synthases,TPS)的作用下最终转化为萜烯化合物,根据其产物碳链长度可分为:单萜(Monoterpenes,C10)、倍半萜(Sesquiterpenes,C15)、二萜(Diterpenes,C20)、二倍半萜(Sesterterpenes,C25)和三萜(Triterpenes,C30)等(图1)[14-15,21]。
图1
2 植物异戊烯基转移酶及其基因的研究现状
类异戊二烯化合物是自然界中化学成分最丰富的一类代谢产物,它是植物质膜结构的重要组成部分,在生物体内介导氧化还原反应,还具有调节繁殖周期、吸引配偶、传递转化信号等重要生理生化功能。在植物细胞内,类异戊二烯化合物是由不同数目的IPP与DMAPP作为初始物缩合而成,催化这一类过程的酶是异戊烯基转移酶,主要包括GPPS、FPPS、GGPPS和GFPPS[22]。
GPPS在植物中广泛存在,是萜烯化合物骨架代谢途径中的关键酶,主要参与薄荷醇、单萜吲哚生物碱和黄酮等物质的合成与调控,它的表达水平受到很多因素的影响,如亚细胞水平的特异分布、基因表达水平、蛋白翻译水平和蛋白聚合形式等[23,24,25,26]。Burke等[23]首先从荷兰薄荷(Mentha spicata)、胡椒薄荷(Mentha×piperita)中克隆获得了两个GPPS基因的全长cDNA序列,之后又从多种植物中分离出GPPS基因,如挪威云杉(Picea abies)[27]、长春花(Catharanthus roseus)[24]、薄荷(Mentha haplocalyx)[28]、金鱼草(Antirrhinum majus)[29]、拟南芥(Arabidopsis thaliana)[25]、啤酒花(Humulus lupulus)[26]、雷公藤(Tripterygium wilfordii)[30]和滇龙胆(Gentiana rigescens)[31]等。近年来,Adal等[32]在薰衣草(Lavandula angustifolia)中克隆出了GPPS基因并对其重组蛋白进行了功能鉴定,当小亚基LiGPPS.SSU1与大亚基LiGPPS.LSU在大肠杆菌中共表达时,得到的重组异源蛋白有GPPS的活性,可产生GPP;Ueoka等[33]利用EST和同源克隆的方法克隆了紫草(Lithospermum erythrorhizon)中43个胞质定位的GPPS基因LeGPPS,其重组蛋白主要产生GPP及少量的FPP;通过对12个蝴蝶兰属(Phalaenopsis)植物的GPPS基因上游启动子片段检测,发现双重复序列与单萜合成具有较强的相关性[34];Zhao等[35]研究了山鸡椒(Litsea cubeba)单萜生物合成途径中的GPPS小亚基1(LcGPPS.SSU1)的功能,发现在野生山鸡椒中瞬时表达和在烟草中过表达均能提高单萜的产量,甚至在烟草中引入LcGPPS.SSU1会影响GGPP的表达,从而提高二萜的产量;Kumar等[36]通过在长春花(Catharanthus roseus)中超表达GPPS基因与香叶醇合酶基因(Geraniol synthase,GES),提高了吲哚生物碱的水平。
FPPS参与一些植物激素与植物色素,如细胞分裂素、类胡萝卜素、叶绿素、脱落酸和赤霉素等类异戊二烯化合物的生物合成,这类有机化合物对维持植物生长发育具有重要作用[37],一些FPPS也有细胞增殖或信号转导等作用[38]。目前,植物FPPS基因已经分别从水稻(Oryza sativa)[39]、高粱(Sor-ghum bicolor)[40]、白木香(Aquilaria sinensis)[41]、洋常春藤(Hedera helix)[42]及香樟(Cinnamomum camphora)[43]等数十种植物中得到分离。研究表明,在拟南芥中同时敲除FPPS1和FPPS2两个法呢基焦磷酸合酶基因,会导致拟南芥死亡[44];Fei等[45]发现在灵芝(Ganoderma Lucidum)中过表达FPPS基因能导致角鲨烯合酶基因(Squalene synthase,SQS)和羊甾醇合酶(Lanosterol synthase)基因 LS的转录水平上调,从而提高灵芝的灵芝酸产量。
GGPPS能够催化IPP和DMAPP合成GGPP作为类维生素A、类胡萝卜素、二萜类化合物和天然橡胶等生物合成前体物质[46,47,48,49]。目前,已有多种植物的GGPPS基因得到研究,如南方红豆杉(Taxus wallichiana var. Mairei)[50]、银杏(Ginkgo biloba)[51]、拟南芥[20]等。在一部分植物中,GGPPS以基因家族的形式存在,例如在拟南芥中发现了12个GGPPS同源基因[20];Zhu等[52]在丹参(Salvia miltiorrhiza)中分离出3个GGPPS基因;李锋等[53]在烟草中也分离出4个GGPPS基因。近两年对于GGPPS基因的研究较多,Su等[54]在雷公藤中鉴定了8个可被调控的GGPPS基因,其蛋白功能鉴定的研究表明GGPPS1在合成与二萜、叶绿素及类胡萝卜素等相关的GGPP上发挥主要作用,而GGPPS8对雷公藤的根系生长有着重要的作用;Huang等[55]从雨生红球藻(Haematococcus pluvialis)中分离出8个预测的HpGGPPS候选基因,在大肠杆菌中异源表达鉴定其功能,结果表明其中两个蛋白具有GGPPS的活性,参与色素的形成,为虾青素产量的提高提供了一条生产途径;陈建荣等[56]从栀子(Gardenia jasminoides)中克隆出GGPPS基因小亚基并进行生物信息分析,但未对其蛋白进行功能鉴定。
GFPPS基因与二倍半萜类化合物合成密切相关,最初是从嗜盐碱杆菌(Natronobacterium pharaonis)中被分离[57],只有在古细菌(Aeropyrum pernix)和甲烷八叠球菌(Methanosarcina mazei)等原核生物中被克隆出来[58,59]。然而,对植物GFPPS基因和二倍半萜类化合物的生物合成研究甚少,Nagel等[60]在拟南芥和大肠杆菌中过表达GFPPS发现,产物大部分为GFPP;Wang等[61]纯化了拟南芥中的10个GGPPS,分析产物后发现其中4个为GFPP,随后利用模型在芸苔属(Brassica)植物中验证其正确性;Liu等[62]克隆了米团花(Leucosceptrum canum)基因LcGFPPS,在拟南芥中过表达GFPPS,验证其具有GFPP的活性,是二倍半萜类化合物合成的前体物质,为其他物种研究二倍半萜类化合物的生物合成提供了一个途径。
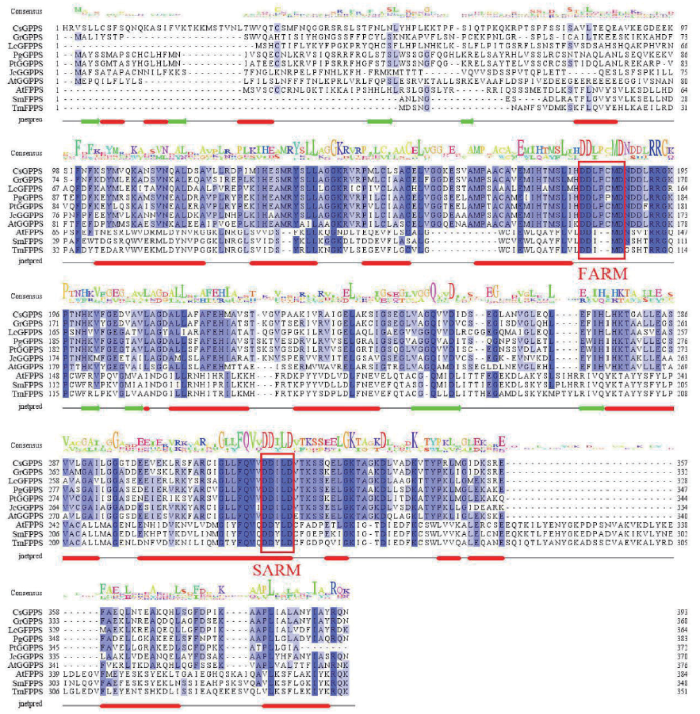
3 异戊烯基转移酶基因保守结构域分析
异戊烯类化合物的生物合成均是由IPP和DMAPP经异戊烯基转移酶催化缩合而成,有着不同的碳氢链长度且有特异性,是许多类异戊二烯化合物的前体,因为它们的氨基酸序列有高度的相似性,因此这些酶被认为具有相似的结构和相同的催化机制[63,64]。利用Clustral X软件我们对不同植物异戊烯基转移酶基因氨基酸序列的比对(图2),这些基因存在两个高度保守的富含天冬氨酸(Asp)的区域,第一个基序FARM(First aspartate-rich motif,DDX(2-4)D)和第二个基序SARM(Second aspartate-rich motif,DDXXD)对促进催化功能和底物结合至关重要(其中D表示Asp,X表示任意氨基酸)[65],这两个基序是典型的异戊烯基转移酶活性结构域,是缩合反应的结合位点。Burke等[23]分析了荷兰薄荷、胡椒薄荷与其他植物的GPPS氨基酸序列,发现其中存在FARM与SARM基序;Szkopiñska等[66]比对各个植物的FPPS氨基酸序列发现了两个天冬氨酸FARM和SARM基序;Huang等[55]比对了12个GGPPS氨基酸序列,发现两个保守区域FARM与SARM;Nagel等[60]认为GFPPS活性与特定的氨基酸残基有关,分析了拟南芥IDS部分氨基酸序列,发现存在天冬氨酸保守区域FARM;Liu等[62]比对了其他植物与5个米团花GFPPS氨基酸序列发现,这些序列中存在5个保守区,包括两个保守的天冬氨酸基序(DDLPCMD和DDILD)。
图2
图2
异戊烯基转移酶基因氨基酸序列比对
CsGPPS:大麻Cannabis sativa(ARE72269.1);GrGPPS:滇龙胆Gentiana rigescens(AHK06853.1);LcGFPPS:米团花Leucosceptrum canum(ALT16903.1);PgGPPS:白云杉Picea glauca(AHE15048.1);PtGGPPS:火炬松Pinus taeda(QBS32941.1);JcGGPPS:麻风树Jatropha curcas(ADD82422.1);AtGGPPS:拟南芥Arabidopsis thaliana(NP_179960.1);AtFPPS:拟南芥Arabidopsis thaliana(NP_199588.1);SmFPPS:丹参Salvia miltiorrhiza(ABV08819.1);TmFPPS:曼地亚红豆杉Taxus × media(AAS19931.1)
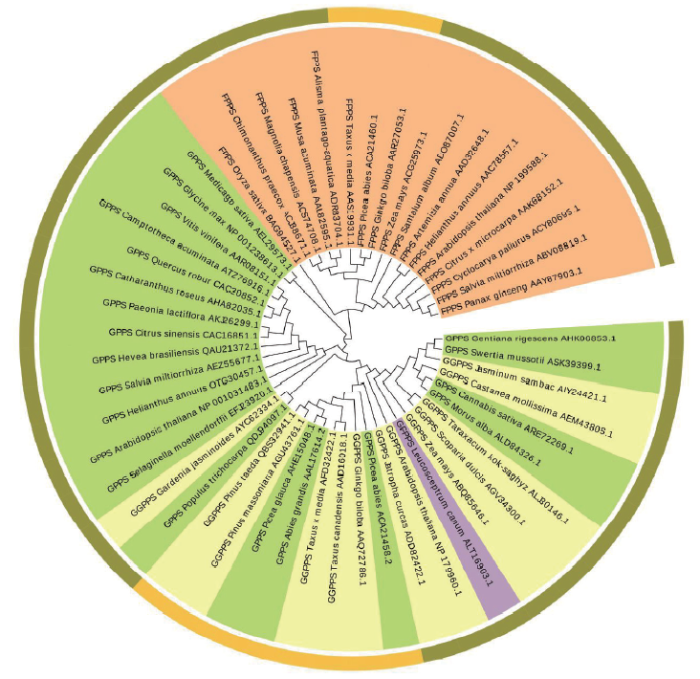
4 异戊烯基转移酶基因家族分子系统进化分析
近年来,为探讨植物异戊烯基转移酶基因家族的进化关系,Liu等[62]对植物的IDS基因用极大似然法构建了系统进化树,其结果主要分为A、B两簇,A簇包含两个亚簇 I 和 II,FPPS基因主要在B簇,而A簇中大部分是GPPS、GGPPS与GFPPS基因,表明其分类是依据生化功能的相近程度,而不是分类起源;Huang等[55]构建了GGPPS基因的进化树,8个雨生红球藻HPGGPPS基因被分到了3个不同的平行组,表明它们分别与各自组中GGPPS基因的功能相近;Su等[54]将雷公藤的8个GGPPS基因与其他植物异戊烯基转移酶基因家族成员一起构建进化树,结果表明这8个GGPPS基因依据功能相近程度被分散在各个分支上。
我们从NCBI上下载各个植物异戊烯基转移酶基因,利用分子进化遗传分析软件MEGA7构建了系统进化树(图3),结果表明,FPPS基因单独聚集成一大类,属于一个独立的分支,而其余3个基因GPPS、GGPPS和GFPPS聚集成另一大类,其中裸子植物的GPPS、GGPPS基因在同一分支上,表明其功能相近,这与上述研究结果一致。
图3
图3
不同植物异戊烯基转移酶基因系统进化树
外圈:裸子植物为黄色,被子植物为黄绿色,蕨类植物为紫色;内圈:GPPS以青色为背景,GGPPS以淡黄色为背景,FPPS以橙色为背景,GFPPS以淡紫色为背景。
Gentiana rigescens:滇龙胆;Swertia mussotii:川西獐牙菜;Jasminum sambac:茉莉;Castanea mollissima:板栗;Cannabis sativa:大麻;Morus alba:桑;Taraxacum kok-saghyz:橡胶草;Scoparia dulcis:野甘草;Zea mays:玉米;Leucosceptrum canum:米团花;Arabidopsis thaliana:拟南芥;Jatropha curcas:麻风树;Picea abies:欧洲云杉;Ginkgo biloba:银杏;Taxus canadensis:加拿大红豆杉;Taxus × media:曼地亚红豆杉;Abies grandis:冷杉;Picea glauca:白云杉;Pinus massoniana:马尾松;Pinus taeda:火炬松;Populus trichocarpa:毛果杨;Gardenia jasminoides:栀子;Selaginella moellendorffii:卷柏;Helianthus annuus:向日葵;Salvia miltiorrhiza:丹参;Hevea brasiliensis:橡胶树;Citrus sinensis:甜橙;Paeonia lactiflora:芍药;Catharanthus roseus:长春花;Quercus robur:夏栎;Camptotheca acuminata:喜树;Vitis vinifera:葡萄;Glycine max:大豆;Medicago sativa:紫花苜蓿;Oryza sativa:水稻;Chimonanthus praecox:腊梅;Magnolia chapensis:乐昌含笑;Musa acuminata:香蕉;Alisma plantago-aquatica:泽泻;Santalum album:檀香;Artemisia annua:黄花蒿;Citrus × microcarpa:柑橘;Cyclocarya paliurus:青钱柳;Panax ginseng:人参
5 针叶树异戊烯基转移酶基因的研究
裸子植物包括松杉纲(Coniferopsida)、苏铁纲(Cycadopsida)、银杏纲(Ginkgopsida)和买麻藤纲(Gnetopsida)是一种古老而广泛的胚珠裸露不被子房包被的植物,具有重要的经济和生态价值[67],。除买麻藤纲外,现已在银杏纲、松杉纲和苏铁纲发现了多个IDS[68]。裸子植物的针叶树种占世界森林39%,对北半球和南半球依赖林业的经济体具有巨大价值[69]。针叶树会产生复杂的单萜、倍半萜和二萜等次生代谢物,最显著的形式是松脂,是由松树树脂道中的泌脂细胞生成的天然树脂[70],可以抵御昆虫和病原菌攻击的物理和化学防御[71,72,73,74,75]。通过加热蒸馏等简单步骤去除杂质后可制成松节油(单萜和倍半萜)、松香(二萜树脂酸)和其他重要的工业原料,广泛应用于造纸、油漆、合成橡胶、涂料、纺织、医药、食品、肥皂、印染等多个领域[76,77],加工后又可作为高品质的生物燃料[78]。在受到生物或非生物刺激后,针叶树会从树脂道中释放松脂,并诱导松脂合成,因此,松脂在针叶树的防御系统中也起重要的作用[79,80,81,82,83]。
相较于被子植物,裸子植物异戊烯基转移酶基因的研究较少,目前已从银杏中克隆到的有GbIDS1、GbIDS2-1、GbIDS2(登陆号分别为:DQ251631、DQ251632和DQ251633)[68],和GbFPPS(登陆号:AY389818)[84];表1是从部分针叶树中克隆到的异戊烯基转移酶基因,对白云杉过表达IDS基因,研究发现单萜与二萜酸的含量无显著变化,而产生大量的香叶基香叶基脂肪酸酯(Geranylgeranyl fatty acid esters)用于抵御模毒蛾(Lymantria monacha)[85];从挪威云杉中克隆出3个IDS基因,其纯化的重组蛋白分析表明,其中一个基因编码FPPS,另外两个编码GGPPS,而这3个基因中,只有一个参与松脂的生物合成[86];Liao等[87]在曼地亚红豆杉中分离出FPPS基因,并发现重组FPPS蛋白能催化IPP底物缩合生成FPP;Burke等[88,89]在冷杉中分离出GPPS与GGPPS基因,发现重组GPPS与GGPPS蛋白可以催化IPP和DMAPP底物分别生成GPP与GGPP;从加拿大红豆杉中克隆了GGPPS基因,并在茉莉酸甲酯(Methyl Jasmonate)处理后检测到GGPPS与紫杉烯合酶(Taxadiene synthase)的活性有较大提高[90];陈博雯等[91]从马尾松中分离出GGPPS基因,仅对其进行生物信息学分析,未对酶功能作鉴定;钱丹等[92]从海南粗榧(Cephalotaxus mannii)克隆了GGPPS基因,用真菌和细菌的激发子以及茉莉酸甲酯处理海南粗榧,均发现GGPPS基因的表达量上升,但程度和持续时间有所不同。
表1 针叶树中已克隆的部分异戊烯基转移酶基因
| 基因名称 | 物种 | 登录号 | 文献 |
|---|---|---|---|
| PgIDS | Picea glauca | AHE15048.1 | [85] |
| PtIDS1/PtIDS2 | Pinus taeda | EF095154/EF095155 | [68] |
| AgGPPS | Abies grandis | AAN01133-5 | [88] |
| PaGPPS | Picea abies | ACA21458-9 | [27] |
| PaFPPS | Picea abies | ACA21460 | [86] |
| TmFPPS | Taxus media | AY461811 | [87] |
| AgGGPPS | Abies grandis | AAL17614 | [89] |
| TcaGGPPS | Taxus canadensis | AAD16018 | [90] |
| PaGGPPS | Picea abies | ACA21461-2 | [86] |
| PaGPPS/GGPPS | Picea abies | ACZ57571 | [27] |
| PmGGPPS | Pinus massoniana | AGU43761.1 | [91] |
| CmGGPPs | Cephalotaxus mannii | AGM53487.1 | [92] |
6 展望
类异戊二烯化合物及其衍生物萜烯类物质功能复杂,在药物、农林业等各个行业有着极大的应用前景,因此了解它们的生物合成方式并研究其合成途径中的关键酶及其基因,是推动这些化合物应用的基础。同时,由于很多针叶树种含有以类异戊二烯的衍生物萜类为主要成分的松脂,不仅能为产脂的林业化学工业带来可观的经济效益,而且是针叶树抵御许多有害生物的重要自源物质[93],如松树萎蔫病是由松材线虫引起的一种严重威胁森林安全的毁灭性松树病害,据国家林业和草原局2019年第4号公告显示,松材线虫病在全国18个省(直辖市、自治区)588个县级行政区发生危害面积64.93万hm2;自我国1982年首次发现30多年来累计造成数十亿株松树死亡,经济损失上千亿元,我国包括绝大部分马尾松林在内的广大面积松林正面临着松材线虫病爆发的严重威胁,当下对之仍束手无策。但有研究表明α-异松油烯和β-水芹烯对松材线虫繁殖有较强抑制,并且β-水芹烯与莰烯具有杀线虫活性[94]。而目前对于类异戊二烯生物合成的相关报道相对少,因此研究其合成的分子机制可为树木高产脂和抗性遗传育种提供理论依据。
近年来,研究者已在多种植物中分离出类异戊二烯化合物生物合成的关键酶基因GPPS、GGPPS、FPPS与GFPPS,并分析其功能,但大多数研究只针对单个基因的克隆及表达,缺乏对通路上相关基因的综合深入研究,而事实上相关多基因互作对于其整体的表达水平影响更为广泛。此外,对于类异戊二烯化合物生物合成酶的功能鉴定,大部分研究仍停留于对模式植物拟南芥和烟草的分析,很少在同源植物上进行过表达及功能鉴定,也未能将研究成果运用到实际生产中。可见今后极有必要通过前沿性的组学研究手段,进一步阐明重要造林树种(特别是主产松脂的松类树种)类异戊二烯化合物生物合成的相关机制。
目前,这些基因的研究主要集中于被子植物,裸子植物中的相关研究较少,且对于异戊烯基转移酶在裸子植物中进行功能方面的鉴定鲜见报道,可能是由于多数裸子植物的遗传转化体系还未成功建立,离体培养植株再生是其组织培养中难度较大的一个领域,还有待于进一步深入研究,并实现技术的熟化。此外,类异戊二烯化合物生物合成酶的相关研究(包括分离提纯、理化性质分析、催化机制研究等)也有待深入,以便能为人类在模拟生物合成领域拓展新思路,为进一步开发利用类异戊二烯化合物创造条件。
参考文献
Plant isoprenoids:a general overview
[J].DOI:10.1007/978-1-4939-0606-2_1 URL PMID:24777786 [本文引用: 1]
Terpenes and isoprenoids:a wealth of compounds for global use
[J].
植物萜类生物合成中的后修饰酶
[J].
Post-modification enzymes involved in the biosynjournal of plant terpenoids
[J].
芳香族异戊烯转移酶的研究进展
[J].
Research progress of aromatic prenyltransferase
[J].
Reaction kinetics, catalytic mechanisms, conformational changes, and inhibitor design for prenyltransferases
[J].
DOI:10.1021/bi900371p
URL
PMID:19537817
[本文引用: 1]

Isoprenoids comprise a family of more than 55000 natural products with great structural variety derived from five-carbon isopentenyl diphosphate (IPP) and its isomer dimethylallyl diphosphate (DMAPP). Allylic diphosphates such as farnesyl diphosphate (FPP) synthesized from DMAPP and IPP serve as outlet points for a great variety of products. A group of prenyltransferases catalyzing chain elongation of FPP to designated lengths by consecutive condensation reactions with specific numbers of IPP are classified as cis and trans types according to the stereochemistry of the double bonds formed by IPP condensation. The complete kinetics of the multistep IPP condensation reactions by both types of enzymes has been determined using steady-state and pre-steady-state approaches. Because their crystal structures were determined in conjunction with biochemical studies, a more thorough understanding of their catalytic mechanisms, protein conformational changes, and product chain-length determination mechanisms has been gained recently. Since these prenyltransferases play important roles, potent inhibitors have been identified and their cocrystal structures have been determined for drug development. In this review, the current knowledge of these prenyltransferases that synthesize prenyl oligomers or polymers is summarized.
Polyisoprenoids:structure, biosynjournal and function
[J].
Polyisoprenoids - secondary metabolites or physiologically important superlipids?
[J].
DOI:10.1016/j.bbrc.2011.03.059
URL
PMID:21419101
[本文引用: 1]

The polyisoprenoid alcohols (dolichols and polyprenols) are found in all living organism, from bacteria to mammals. In animal and yeast cells polyisoprenoids are derived from the cytoplasmic mevalonate (MVA) pathway while in plants two biosynthetic pathways, the MVA and the plastidial methylerythritol phosphate (MEP) pathway provide precursors for polyisoprenoid biosynthesis. The key enzymes of polyisoprenoid synthesis are cis-prenyltransferases (CTPs), responsible for construction of the long hydrocarbon skeleton. CPTs elongate a short all-trans precursor, oligoprenyl diphosphate, by sequential addition of the desired number of isopentenyl diphosphate molecules which results in formation of a stretch of cis units. Several genes encoding CPT have been cloned from bacteria, plants and mammals. Interestingly, in Arabidopsis, the tissue-specific expression of ten putative cis-prenyltransferases was observed. In contrast to polyisoprenoid phosphates serving as cofactors in the biosynthesis of glycoproteins, glucosyl phosphatidyl inositol (GPI) anchor or bacterial peptidoglycan, the biological importance of polyprenols and dolichols still remains a question of debate besides their function of reservoir of substrates for kinase. These extremely hydrophobic superlipids are postulated to be involved in intracellular traffic of proteins and in cellular defense against adverse environmental conditions. Recent publications show a direct link between the dolichol biosynthetic pathway and congenital disorders of glycosylation (CDG). These discoveries highlighting the cellular significance of polyisoprenoids simultaneously establish the background for future pharmacological interventions. Our mini-review summarizes the results of recent studies on polyisoprenoids.
Polyprenols are synthesized by a plastidial cis-prenyltransferase and influence photosynthetic performance
[J].
DOI:10.1105/tpc.16.00796
URL
PMID:28655749
[本文引用: 1]

Plants accumulate a family of hydrophobic polymers known as polyprenols, yet how they are synthesized, where they reside in the cell, and what role they serve is largely unknown. Using Arabidopsis thaliana as a model, we present evidence for the involvement of a plastidial cis-prenyltransferase (AtCPT7) in polyprenol synthesis. Gene inactivation and RNAi-mediated knockdown of AtCPT7 eliminated leaf polyprenols, while its overexpression increased their content. Complementation tests in the polyprenol-deficient yeast rer2 mutant and enzyme assays with recombinant AtCPT7 confirmed that the enzyme synthesizes polyprenols of approximately 55 carbons in length using geranylgeranyl diphosphate (GGPP) and isopentenyl diphosphate as substrates. Immunodetection and in vivo localization of AtCPT7 fluorescent protein fusions showed that AtCPT7 resides in the stroma of mesophyll chloroplasts. The enzymatic products of AtCPT7 accumulate in thylakoid membranes, and in their absence, thylakoids adopt an increasingly
New insights into short-chain prenyltransferases:structural features, evolutionary history and potential for selective inhibition
[J].
DOI:10.1007/s00018-009-0100-9
URL
PMID:19633972
[本文引用: 1]

Isoprenoids form an extensive group of natural products involved in a number of important biological processes. Their biosynthesis proceeds through sequential 1'-4 condensations of isopentenyl diphosphate (C5) with an allylic acceptor, the first of which is dimethylallyl diphosphate (C5). The reactions leading to the production of geranyl diphosphate (C10), farnesyl diphosphate (C15) and geranylgeranyl diphosphate (C20), which are the precursors of mono-, sesqui- and diterpenes, respectively, are catalyzed by a group of highly conserved enzymes known as short-chain isoprenyl diphosphate synthases, or prenyltransferases. In recent years, the sequences of many new prenyltransferases have become available, including those of several plant and animal geranyl diphosphate synthases, revealing novel mechanisms of product chain-length selectivity and an intricate evolutionary path from a putative common ancestor. Finally, there is considerable interest in designing inhibitors specific to short-chain prenyltransferases, for the purpose of developing new drugs or pesticides that target the isoprenoid biosynthetic pathway.
Functional gene network of prenyltransferases in Arabidopsis thaliana
[J].
植物中的异戊烯基转移酶
[J].
Prenyltransferase in plants
[J].
微生物来源芳香类化合物异戊烯基转移酶研究进展
[J].
Research progress in aromatic prenyltransferases originated from microorganisms
[J].
Chemoenzymatic syntheses of prenylated aromatic small molecules using Streptomyces prenyltransferases with relaxed substrate specificities
[J].
DOI:10.1016/j.bmc.2008.07.052
URL
PMID:18682327
[本文引用: 1]

NphB is a soluble prenyltransferase from Streptomyces sp. strain CL190 that attaches a geranyl group to a 1,3,6,8-tetrahydroxynaphthalene-derived polyketide during the biosynthesis of anti-oxidant naphterpin. Here we report multiple chemoenzymatic syntheses of various prenylated compounds from aromatic substrates including flavonoids using two prenyltransferases NphB and SCO7190, a NphB homolog from Streptomyces coelicolor A3(2), as biocatalysts. NphB catalyzes carbon-carbon-based and carbon-oxygen-based geranylation of a diverse collection of hydroxyl-containing aromatic acceptors. Thus, this simple method using the prenyltransferases can be used to explore novel prenylated aromatic compounds with biological activities. Kinetic studies with NphB reveal that the prenylation reaction follows a sequential ordered mechanism.
Biosynjournal and biological functions of terpenoids in plants
[J].
Multi-substrate terpene synthases:their occurrence and physiological significance
[J].
DOI:10.3389/fpls.2016.01019
URL
PMID:27462341
[本文引用: 4]

Terpene synthases are responsible for synthesis of a large number of terpenes in plants using substrates provided by two distinct metabolic pathways, the mevalonate-dependent pathway that is located in cytosol and has been suggested to be responsible for synthesis of sesquiterpenes (C15), and 2-C-methyl-D-erythritol-4-phosphate pathway located in plastids and suggested to be responsible for the synthesis of hemi- (C5), mono- (C10), and diterpenes (C20). Recent advances in characterization of genes and enzymes responsible for substrate and end product biosynthesis as well as efforts in metabolic engineering have demonstrated existence of a number of multi-substrate terpene synthases. This review summarizes the progress in the characterization of such multi-substrate terpene synthases and suggests that the presence of multi-substrate use might have been significantly underestimated. Multi-substrate use could lead to important changes in terpene product profiles upon substrate profile changes under perturbation of metabolism in stressed plants as well as under certain developmental stages. We therefore argue that multi-substrate use can be significant under physiological conditions and can result in complicate modifications in terpene profiles.
Volatile terpenoids:multiple functions, biosynjournal, modulation and manipulation by genetic engineering
[J].
Biochemistry and molecular biology of the isoprenoid biosynthetic pathway in plants
[J].
Terpene biosynjournal:modularity rules
[J].
DOI:10.1002/anie.201103110
URL
PMID:22105807
[本文引用: 1]

Terpenes are the largest class of small-molecule natural products on earth, and the most abundant by mass. Here, we summarize recent developments in elucidating the structure and function of the proteins involved in their biosynthesis. There are six main building blocks or modules (alpha, beta, gamma, delta, epsilon, and zeta) that make up the structures of these enzymes: the alphaalpha and alphadelta head-to-tail trans-prenyl transferases that produce trans-isoprenoid diphosphates from C(5) precursors; the epsilon head-to-head prenyl transferases that convert these diphosphates into the tri- and tetraterpene precursors of sterols, hopanoids, and carotenoids; the betagamma di- and triterpene synthases; the zeta head-to-tail cis-prenyl transferases that produce the cis-isoprenoid diphosphates involved in bacterial cell wall biosynthesis; and finally the alpha, alphabeta, and alphabetagamma terpene synthases that produce plant terpenes, with many of these modular enzymes having originated from ancestral alpha and beta domain proteins. We also review progress in determining the structure and function of the two 4Fe-4S reductases involved in formation of the C(5) diphosphates in many bacteria, where again, highly modular structures are found.
Elucidation of the methylerythritol phosphate pathway for isoprenoid biosynjournal in bacteria and plastids:a metabolic milestone achieved through genomics
[J].
Genome organization in Arabidopsis thaliana:a survey for genes involved in isoprenoid and chlorophyll metabolism
[J].
DOI:10.1023/a:1023005504702
URL
PMID:12777052
[本文引用: 3]

The isoprenoid biosynthetic pathway provides intermediates for the synthesis of a multitude of natural products which serve numerous biochemical functions in plants: sterols (isoprenoids with a C30 backbone) are essential components of membranes; carotenoids (C40) and chlorophylls (which contain a C20 isoprenoid side-chain) act as photosynthetic pigments; plastoquinone, phylloquinone and ubiquinone (all of which contain long isoprenoid side-chains) participate in electron transport chains; gibberellins (C20), brassinosteroids (C30) and abscisic acid (C15) are phytohormones derived from isoprenoid intermediates; prenylation of proteins (with C15 or C20 isoprenoid moieties) may mediate subcellular targeting and regulation of activity; and several monoterpenes (C10), sesquiterpenes (C15) and diterpenes (C20) have been demonstrated to be involved in plant defense. Here we present a comprehensive analysis of genes coding for enzymes involved in the metabolism of isoprenoid-derived compounds in Arabidopsis thaliana. By combining homology and sequence motif searches with knowledge regarding the phylogenetic distribution of pathways of isoprenoid metabolism across species, candidate genes for these pathways in A. thaliana were obtained. A detailed analysis of the vicinity of chromosome loci for genes of isoprenoid metabolism in A. thaliana provided evidence for the clustering of genes involved in common pathways. Multiple sequence alignments were used to estimate the number of genes in gene families and sequence relationship trees were utilized to classify their individual members. The integration of all these datasets allows the generation of a knowledge-based metabolic map of isoprenoid metabolic pathways in A. thaliana and provides a substantial improvement of the currently available gene annotation.
The family of terpene synthases in plants:a mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom
[J].
DOI:10.1111/j.1365-313X.2011.04520.x
URL
PMID:21443633
[本文引用: 2]

Some plant terpenes such as sterols and carotenes are part of primary metabolism and found essentially in all plants. However, the majority of the terpenes found in plants are classified as 'secondary' compounds, those chemicals whose synthesis has evolved in plants as a result of selection for increased fitness via better adaptation to the local ecological niche of each species. Thousands of such terpenes have been found in the plant kingdom, but each species is capable of synthesizing only a small fraction of this total. In plants, a family of terpene synthases (TPSs) is responsible for the synthesis of the various terpene molecules from two isomeric 5-carbon precursor 'building blocks', leading to 5-carbon isoprene, 10-carbon monoterpenes, 15-carbon sesquiterpenes and 20-carbon diterpenes. The bryophyte Physcomitrella patens has a single TPS gene, copalyl synthase/kaurene synthase (CPS/KS), encoding a bifunctional enzyme producing ent-kaurene, which is a precursor of gibberellins. The genome of the lycophyte Selaginella moellendorffii contains 18 TPS genes, and the genomes of some model angiosperms and gymnosperms contain 40-152 TPS genes, not all of them functional and most of the functional ones having lost activity in either the CPS- or KS-type domains. TPS genes are generally divided into seven clades, with some plant lineages having a majority of their TPS genes in one or two clades, indicating lineage-specific expansion of specific types of genes. Evolutionary plasticity is evident in the TPS family, with closely related enzymes differing in their product profiles, subcellular localization, or the in planta substrates they use.
Chain elongation in the isoprenoid biosynthetic pathway
[J].
DOI:10.1016/s1367-5931(97)80054-3
URL
PMID:9667899
[本文引用: 1]

Isoprenyl diphosphate synthases catalyze addition of allylic diphosphate primers to the isoprene unit in isopentenyl diphosphate to produce polyisoprenoid diphosphates with well defined chain lengths. Phylogenetic correlations suggest that the synthases which catalyze formation of isoprenoid diphosphates with (E) double bonds have evolved from a common ancestor. X-ray crystallographic studies of farnesyl diphosphate synthase in conjunction with site-directed mutagenesis have provided important new information about the residues involved in binding and catalysis and the source of chain length selectivity for the enzymes that catalyze chain elongation.
Geranyl diphosphate synthase:cloning, expression, and characterization of this prenyltransferase as a heterodimer
[J].
Heteromeric and homomeric geranyl diphosphate synthases from Catharanthus roseus and their role in monoterpene indole alkaloid biosynjournal
[J].
DOI:10.1093/mp/sst058
URL
[本文引用: 2]

This work suggests that, in Catharanthus roseus, the inactive small subunit of heteromeric GPP synthase (GPPS) interacts with bifunctional G(G)PPS and redirects the metabolic flux, thus acting as primary regulator of monoterpene indole alkaloid biosynthesis, whereas homomeric GPPS could be involved in ubiquinone formation.Catharanthus roseus is the sole source of two most important monoterpene indole alkaloid (MIA) anti-cancer agents: vinblastine and vincristine. MIAs possess a terpene and an indole moiety derived from terpenoid and shikimate pathways, respectively. Geranyl diphosphate (GPP), the entry point to the formation of terpene moiety, is a product of the condensation of isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP) by GPP synthase (GPPS). Here, we report three genes encoding proteins with sequence similarity to large subunit (CrGPPS.LSU) and small subunit (CrGPPS.SSU) of heteromeric GPPSs, and a homomeric GPPSs. CrGPPS.LSU is a bifunctional enzyme producing both GPP and geranyl geranyl diphosphate (GGPP), CrGPPS.SSU is inactive, whereas CrGPPS is a homomeric enzyme forming GPP. Co-expression of both subunits in Escherichia coli resulted in heteromeric enzyme with enhanced activity producing only GPP. While CrGPPS.LSU and CrGPPS showed higher expression in older and younger leaves, respectively, CrGPPS.SSU showed an increasing trend and decreased gradually. Methyl jasmonate (MeJA) treatment of leaves significantly induced the expression of only CrGPPS.SSU. GFP localization indicated that CrGPPS.SSU is plastidial whereas CrGPPS is mitochondrial. Transient overexpression of AmGPPS.SSU in C. roseus leaves resulted in increased vindoline, immediate monomeric precursor of vinblastine and vincristine. Although C. roseus has both heteromeric and homomeric GPPS enzymes, our results implicate the involvement of only heteromeric GPPS with CrGPPS.SSU regulating the GPP flux for MIA biosynthesis.
Molecular cloning of geranyl diphosphate synthase and compartmentation of monoterpene synjournal in plant cells
[J].DOI:10.1046/j.1365-313x.2000.00875.x URL [本文引用: 2]
Heterodimeric geranyl(geranyl)diphosphate synthase from hop(Humulus lupulus)and the evolution of monoterpene biosynthesis
[J].
DOI:10.1073/pnas.0904069106
URL
PMID:19482937
[本文引用: 2]

Myrcene, which accounts for 30-50% of the essential oil in hop (Humulus lupulus L.) trichomes, derives from geranyl diphosphate (GPP), the common precursor of monoterpenes. Full-length sequences of heterodimeric GPP synthase small subunit (GPPS.SSU, belonging to the SSU I subfamily) and large subunit (LSU) cDNAs were mined from a hop trichome cDNA library. The SSU was inactive, whereas the LSU produced GPP, farnesyl diphosphate, and geranylgeranyl diphosphate (GGPP) from dimethylallyl diphosphate and isopentenyl diphosphate in vitro. Coexpression of both subunits in Escherichia coli yielded a heterodimeric enzyme exhibiting altered ratios of GPP and GGPP synthase activities and greatly enhanced catalytic efficiency. Transcript analysis suggested that the heterodimeric geranyl(geranyl)diphosphate synthase [G(G)PPS] is involved in myrcene biosynthesis in hop trichomes. The critical role of the conserved CxxxC motif (where
Cloning and characterization of two different types of geranyl diphosphate synthases from Norway spruce(Picea abies)
[J].
DOI:10.1016/j.phytochem.2007.06.022
URL
PMID:17673268
[本文引用: 3]

Geranyl diphosphate (GPP), the universal precursor of monoterpenes, is formed from isopentenyl diphosphate and dimethylallyl diphosphate by the action of geranyl diphosphate synthase, one of the key branchpoint enzymes of terpene biosynthesis. Three types of GPP synthase can be distinguished in plants based on sequence similarity and subunit architecture, but until now individual species have been reported to contain only one of these types. Here we show that the conifer, Norway spruce (Picea abies), contains two different types of GPP synthase belonging to two separate groups of homodimeric proteins. One enzyme, designated PaIDS2 (P. abies isoprenyl diphosphate synthase 2), has high sequence similarity to other gymnosperm GPP synthases. It produces solely GPP in in vitro assays after expression in Escherichia coli and likely participates in monoterpene biosynthesis accompanying induced oleoresin formation, based on dramatic increases in transcript level after methyl jasmonate application. The other enzyme, designated PaIDS3, has highest similarity to the previously reported Arabidopsis thaliana GPP synthase and several other angiosperm sequences, and is not associated with induced oleoresin formation in Norway spruce. In vitro assay of this protein and one encoded by a similar gene sequence from Quercus robur gave substantial amounts of the larger prenyl diphosphates, FPP and GGPP, in addition to GPP. Hence these proteins may not be involved in monoterpene formation and could conceivably form products in addition to GPP in planta.
薄荷GPPS基因原核表达及RNA干扰载体构建
[J].
Prokaryotic expreesion and RNA interference vector construction for geranyl diphosphate synthase of Mentha haplocalyx Briq.
[J].
Formation of monoterpenes in Antirrhinum majus and Clarkia breweri flowers involves heterodimeric geranyl diphosphate synthases
[J].
DOI:10.1105/tpc.020156
URL
PMID:15031409
[本文引用: 1]

The precursor of all monoterpenes is the C10 acyclic intermediate geranyl diphosphate (GPP), which is formed from the C5 compounds isopentenyl diphosphate and dimethylallyl diphosphate by GPP synthase (GPPS). We have discovered that Antirrhinum majus (snapdragon) and Clarkia breweri, two species whose floral scent is rich in monoterpenes, both possess a heterodimeric GPPS like that previously reported from Mentha piperita (peppermint). The A. majus and C. breweri cDNAs encode proteins with 53% and 45% amino acid sequence identity, respectively, to the M. piperita GPPS small subunit (GPPS.SSU). Expression of these cDNAs in Escherichia coli yielded no detectable prenyltransferase activity. However, when each of these cDNAs was coexpressed with the M. piperita GPPS large subunit (GPPS.LSU), which shares functional motifs and a high level of amino acid sequence identity with geranylgeranyl diphosphate synthases (GGPPS), active GPPS was obtained. Using a homology-based cloning strategy, a GPPS.LSU cDNA also was isolated from A. majus. Its coexpression in E. coli with A. majus GPPS.SSU yielded a functional heterodimer that catalyzed the synthesis of GPP as a main product. The expression in E. coli of A. majus GPPS.LSU by itself yielded active GGPPS, indicating that in contrast with M. piperita GPPS.LSU, A. majus GPPS.LSU is a functional GGPPS on its own. Analyses of tissue-specific, developmental, and rhythmic changes in the mRNA and protein levels of GPPS.SSU in A. majus flowers revealed that these levels correlate closely with monoterpene emission, whereas GPPS.LSU mRNA levels did not, indicating that the levels of GPPS.SSU, but not GPPS.LSU, might play a key role in regulating the formation of GPPS and, thus, monoterpene biosynthesis.
雷公藤牻牛儿基焦磷酸合酶基因TwGPPS克隆与表达分析
[J].
Cloning and protein expression analysis of geranyl diphosphate synthase genes in Tripterygium wilfordii
[J].
滇龙胆GrGPPS基因的克隆及其序列分析与原核表达
[J].
Cloning, sequence analysis, and prokaryotic expression of GrGPPS gene in Gentiana rigescens
[J].
Short-chain isoprenyl diphosphate synthases of lavender(Lavandula)
[J].
A cytosol-localized geranyl diphosphate synthase from Lithospermum erythrorhizon and its molecular evolution
[J].
DOI:10.1104/pp.19.00999
URL
PMID:31974127
[本文引用: 1]

Geranyl diphosphate (GPP) is the direct precursor of all monoterpenoids and is the prenyl source of many meroterpenoids, such as geranylated coumarins. GPP synthase (GPPS) localized in plastids is responsible for providing the substrate for monoterpene synthases and prenyltransferases for synthesis of aromatic substances that are also present in plastids, but GPPS activity in Lithospermum erythrorhizon localizes to the cytosol, in which GPP is utilized for the biosynthesis of naphthoquinone pigments, which are shikonin derivatives. This study describes the identification of the cytosol-localized GPPS gene, LeGPPS, through EST- and homology-based approaches followed by functional analyses. The deduced amino acid sequence of the unique LeGPPS showed greater similarity to that of farnesyl diphosphate synthase (FPPS), which generally localizes to the cytosol, than to plastid-localized conventional GPPS. Biochemical characterization revealed that recombinant LeGPPS predominantly produces GPP along with a trace amount of FPP. LeGPPS expression was mainly detected in root bark, in which shikonin derivatives are produced, and in shikonin-producing cultured cells. The GFP fusion protein in onion (Allium cepa) cells localized to the cytosol. Site-directed mutagenesis of LeGPPS and another FPPS homolog identified in this study, LeFPPS1, showed that the His residue at position 100 of LeGPPS, adjacent to the first Asp-rich motif, contributes to substrate preference and product specificity, leading to GPP formation. These results suggest that LeGPPS, which is involved in shikonin biosynthesis, is recruited from cytosolic FPPS and that point mutation(s) result in the acquisition of GPPS activity.
A dual repeat cis-element determines expression of GERANYL DIPHOSPHATE SYNTHASE for monoterpene production in Phalaenopsis orchids
[J].
Overexpression of geranyl diphosphate synthase small subunit 1(LcGPPS. SSU1)enhances the monoterpene content and biomass
[J].
Terpene moiety enhancement by overexpression of geranyl(geranyl)diphosphate synthase and geraniol synthase elevates monomeric and dimeric monoterpene indole alkaloids in transgenic Catharanthus roseus
[J].
DOI:10.3389/fpls.2018.00942
URL
PMID:30034406
[本文引用: 1]

Catharanthus roseus is the sole source of two of the most important anticancer monoterpene indole alkaloids (MIAs), vinblastine and vincristine and their precursors, vindoline and catharanthine. The MIAs are produced from the condensation of precursors derived from indole and terpene secoiridoid pathways. It has been previously reported that the terpene moiety limits MIA biosynthesis in C. roseus. Here, to overcome this limitation and enhance MIAs levels in C. roseus, bifunctional geranyl(geranyl) diphosphate synthase [G(G)PPS] and geraniol synthase (GES) that provide precursors for early steps of terpene moiety (secologanin) formation, were overexpressed transiently by agroinfiltration and stably by Agrobacterium-mediated transformation. Both transient and stable overexpression of G(G)PPS and co-expression of G(G)PPS+GES significantly enhanced the accumulation of secologanin, which in turn elevated the levels of monomeric MIAs. In addition, transgenic C. roseus plants exhibited increased levels of root alkaloid ajmalicine. The dimeric alkaloid vinblastine was enhanced only in G(G)PPS but not in G(G)PPS+GES transgenic lines that correlated with transcript levels of peroxidase-1 (PRX1) involved in coupling of vindoline and catharanthine into 3',4'-anhydrovinblastine, the immediate precursor of vinblastine. Moreover, first generation (T1) lines exhibited comparable transcript and metabolite levels to that of T0 lines. In addition, transgenic lines displayed normal growth similar to wild-type plants indicating that the bifunctional G(G)PPS enhanced flux toward both primary and secondary metabolism. These results revealed that improved availability of early precursors for terpene moiety biosynthesis enhanced production of MIAs in C. roseus at the whole plant level. This is the first report demonstrating enhanced accumulation of monomeric and dimeric MIAs including root MIA ajmalicine in C. roseus through transgenic approaches.
In the photorespiratory pathway phosphoglycolate formed by the oxygenase activity of RubisCo is recycled-7
[M].
A novel role for farnesyl pyrophosphate synthase in fibroblast growth factor-mediated signal transduction
[J].
Collection, mapping, and annotation of over 28, 000 cDNA clones from japonica rice
[J].
DOI:10.1126/science.1081288
URL
PMID:12869764
[本文引用: 1]

We collected and completely sequenced 28,469 full-length complementary DNA clones from Oryza sativa L. ssp. japonica cv. Nipponbare. Through homology searches of publicly available sequence data, we assigned tentative protein functions to 21,596 clones (75.86%). Mapping of the cDNA clones to genomic DNA revealed that there are 19,000 to 20,500 transcription units in the rice genome. Protein informatics analysis against the InterPro database revealed the existence of proteins presented in rice but not in Arabidopsis. Sixty-four percent of our cDNAs are homologous to Arabidopsis proteins.
The Sorghum bicolor genome and the diversification of grasses
[J].
DOI:10.1038/nature07723
URL
PMID:19189423
[本文引用: 1]

Sorghum, an African grass related to sugar cane and maize, is grown for food, feed, fibre and fuel. We present an initial analysis of the approximately 730-megabase Sorghum bicolor (L.) Moench genome, placing approximately 98% of genes in their chromosomal context using whole-genome shotgun sequence validated by genetic, physical and syntenic information. Genetic recombination is largely confined to about one-third of the sorghum genome with gene order and density similar to those of rice. Retrotransposon accumulation in recombinationally recalcitrant heterochromatin explains the approximately 75% larger genome size of sorghum compared with rice. Although gene and repetitive DNA distributions have been preserved since palaeopolyploidization approximately 70 million years ago, most duplicated gene sets lost one member before the sorghum-rice divergence. Concerted evolution makes one duplicated chromosomal segment appear to be only a few million years old. About 24% of genes are grass-specific and 7% are sorghum-specific. Recent gene and microRNA duplications may contribute to sorghum's drought tolerance.
白木香法呢基焦磷合酶基因AsFPS1的克隆及表达分析
[J].
Cloning and expression analysis of farnesyl pyrophosphate synthase from Aquilaria sinensis
[J].
洋常春藤法呢基焦磷酸合酶基因的克隆与序列分析
[J].
Cloning and sequence analyzing of farnesyl pyrophosphate synthase from Hedera helix
[J].
香樟FPPS基因的克隆及生物信息学分析
[J].
Cloning and bioinformatic analysis of FPPS gene from Cinnamomum camphora
[J].
The Arabidopsis thaliana FPP synthase isozymes have overlapping and specific functions in isoprenoid biosynjournal, and complete loss of FPP synthase activity causes early developmental arrest
[J].
DOI:10.1111/j.1365-313X.2010.04253.x
URL
PMID:20497375
[本文引用: 1]

Farnesyl diphosphate (FPP) synthase (FPS) catalyses the synthesis of FPP, the major substrate used by cytosolic and mitochondrial branches of the isoprenoid pathway. Arabidopsis contains two farnesyl diphosphate synthase genes, FPS1 and FPS2, that encode isozymes FPS1L (mitochondrial), FPS1S and FPS2 (both cytosolic). Here we show that simultaneous knockout of both FPS genes is lethal for Arabidopsis, and embryo development is arrested at the pre-globular stage, demonstrating that FPP-derived isoprenoid metabolism is essential. In addition, lack of FPS enzyme activity severely impairs male genetic transmission. In contrast, no major developmental and metabolic defects were observed in fps1 and fps2 single knockout mutants, demonstrating the redundancy of the genes. The levels of sterols and ubiquinone, the major mitochondrial isoprenoid, are only slightly reduced in the single mutants. Although one functional FPS gene is sufficient to support isoprenoid biosynthesis for normal growth and development, the functions of FPS1 and FPS2 during development are not completely redundant. FPS1 activity has a predominant role during most of the plant life cycle, and FPS2 appears to have a major role in seeds and during the early stages of seedling development. Lack of FPS2 activity in seeds, but not of FPS1 activity, is associated with a marked reduction in sitosterol content and positive feedback regulation of 3-hydroxy-3-methylglutaryl CoA reductase activity that renders seeds hypersensitive to the 3-hydroxy-3-methylglutaryl CoA reductase inhibitor mevastatin.
Increased production of ganoderic acids by overexpression of homologous farnesyl diphosphate synthase and kinetic modeling of ganoderic acid production in Ganoderma lucidum
[J].
Identification and characterization of the geranylgeranyl diphosphate synthase in Deinococcus radiodurans
[J].
A bifunctional geranyl and geranylgeranyl diphosphate synthase is involved in terpene oleoresin formation in Picea abies
[J].
Production of natural rubber from Para rubber tree
[J].
Cloning, expression and characterization of a functional cDNA clone encoding geranylgeranyl diphosphate synthase of Hevea brasiliensis
[J].
南方红豆杉GGPP合酶基因的克隆与生物信息学分析
[J].
Molecular cloning and sequence analysis of geranyhlgerany pyrophosphate systhase from Taxus wallichiana var. Mairei
[J].
银杏GGPPS转运肽与GFP融合基因表达载体的构建
[J].
The construction of fusion expression vector carrying GFP and TP of GGPPS from Ginkgo biloba L
.[J].
Geranylgeranyl pyrophosphate synthase encoded by the newly isolated gene GGPS6 from Arabidopsis thaliana is localized in mitochondria
[J].
DOI:10.1023/a:1005898805326
URL
PMID:9349257
[本文引用: 1]

We have cloned a new geranylgeranyl pyrophosphate (GGPP) synthase gene, designated GGPS6, from Arabidopsis thaliana genomic DNA. Nucleotide sequence analysis revealed that the GGPS6 gene contains an open reading frame coding for a protein of 343 amino acid residues with a calculated molecular mass of 37,507 Da. Also, the gene is not interrupted by an intron. The predicted amino acid sequence of the GGPS6 gene shows significant homology (34.0-57.7%) with other GGPP synthases from Arabidopsis. The GGPS6 protein contains a N-terminal signal peptide which is thought to function as an organelle targeting sequence. In fact, the GGPS6-GFP fusion protein was found to be localized exclusively in mitochondria when expressed in tobacco BY-2 cells. In vitro analysis of the enzyme activity as well as genetic complementation analysis with Erwinia uredovora crt gene cluster expressed in Escherichia coli showed that the GGPS6 gene most certainly encodes a GGPP synthase catalyzing the conversion of farnesyl pyrophosphate to GGPP.
烟草牻牛儿基牻牛儿基焦磷酸合成酶基因的克隆及分析
[J].
Cloning and characterization of a new gene encoding geranylgeranyl pyrophosphate synthase from Nicotiana tabacum
[J].
Analysis of the role of geranylgeranyl diphosphate synthase 8 from Tripterygium wilfordii in diterpenoids biosynjournal
[J].
DOI:10.1016/j.plantsci.2019.05.013
URL
PMID:31203883
[本文引用: 2]

Tripterygium wilfordii is known to contain various types of bioactive diterpenoids that exhibit many remarkable activities. Many studies have recently been targeted toward the elucidation of the diterpenoids biosynthetic pathways in attempts to obtain these compounds with a view to solving the dilemma of low yield in plants. However, the short-chain prenyltransferases (SC-PTSs) responsible for the formation of geranylgeranyl diphosphate (GGPP), a crucial precursor for synthesizing the skeleton structures of diterpenoids, have not been characterized in depth. Here, T. wilfordii transcriptome data were used to identify eight putative GGPPSs, including two small subunits of geranyl diphosphate synthase (GPPS.SSU). Of them, GGPPS1, GGPPS7, GGPPS8, GPPS.SSU II and GPPS.SSU were translocated mainly into chloroplasts, and GGPPS8 exhibited the optimal catalytic efficiency with respect to catalyzing the formation of GGPP. In addition, the expression pattern of GGPPS8 was similar to that of downstream terpene synthase genes that are directly correlated with triptolide production in roots, indicating that GGPPS8 was most likely to participate in triptolide biosynthesis in roots among the studied enzymes. GPPS.SSU was inactive alone but interacted with GGPPS1, GGPPS7 and GGPPS8 to change the product from GGPP to GPP. These findings implicate that these candidate genes can be regulated to shift the metabolic flux toward diterpenoid formation, increasing the yields of bioactive diterpenoids in plants.
Discovery of geranylgeranyl pyrophosphate synthase(GGPPS)paralogs from Haematococcus pluvialis based on iso-seq analysis and their function on astaxanthin biosynjournal
[J].
栀子(Gardenia jasminoides)GGPPS基因小亚基的克隆及表达分析
[J].
Cloning and expression analysis of gardenia jasminoides GGPPS small subunit gene
[J].
A novel prenyltransferase, farnesylgeranyl diphosphate synthase, from the haloalkaliphilic archaeon, Natronobacterium pharaonis
[J].
DOI:10.1016/0014-5793(94)80475-3
URL
PMID:8137956
[本文引用: 1]

A novel prenyltransferase, farnesylgeranyl diphosphate (FGPP) synthase (EC 2.5.1.X), which synthesizes C25-prenyl diphosphate, was found in the haloalkaliphilic archaeon Natronobacterium pharaonis. It was separated from geranylgeranyl diphosphate (GGPP) synthase (EC 2.5.1.29), which synthesizes C20-prenyl diphosphate, a major prenyltransferase in this organism. The highest activity of FGPP synthase was observed when GGPP was used as the allylic substrate. FGPP synthase may synthesize a precursor for the C25 moiety of C20, C25 diether lipids using a longer allylic diphosphate, such as GGPP synthesized by GGPP synthase, rather than dimethylallyl diphosphate, which is the product of isopentenyl diphosphate isomerase.
Novel prenyltransferase gene encoding farnesylgeranyl diphosphate synthase from a hyperthermophilic archaeon, Aeropyrum pernix. Molecularevolution with alteration in product specificity
[J].
DOI:10.1046/j.1432-1327.2000.00967.x
URL
PMID:10632701
[本文引用: 1]

Prenyltransferases catalyse sequential condensations of isopentenyl diphosphate with allylic diphosphates. Previously, we reported the presence of farnesylgeranyl diphosphate (FGPP) synthase activity synthesizing C25 isoprenyl diphosphate in Natronobacterium pharaonis which is a haloalkaliphilic archaeon having C20-C25 diether lipids in addition to C20-C20 diether lipids commonly occurring in archaea [Tachibana, A. (1994) FEBS Lett. 341, 291-294]. Recently, it was found that a newly isolated aerobic hyperthermophilic archaeon, Aeropyrum pernix, had only C25-C25 diether lipids, not the usual C20-containing lipids [Morii, H., Yagi, H., Akutsu, H., Nomura, N., Sako, Y. & Koga, Y. (1999) Biochim. Biophys. Acta 1436, 426-436]. In this report, we describe the isoloation from A. pernix of the novel prenyltransferase gene, fgs, encoding FGPP synthase. The protein encoded by fgs was expressed in Escherichia coli as a glutathione S-transferase fusion protein and produced FGPP as a final product. Phylogenetic analysis of fgs with other prenyltransferases revealed that the short-chain prenyltransferase family is divided into three subfamilies: bacterial subfamily I, eukaryotic subfamily II, and archaeal subfamily III. fgs is clearly contained within the archaeal geranylgeranyl diphosphate (GGPP) synthase group (subfamily III), suggesting that FGPP synthase evolved from an archaeal GGPP synthase with an alteration in product specificity.
Geranylfarnesyl diphosphate synthase from Methanosarcina mazei:Different role, different evolution
[J].
DOI:10.1016/j.bbrc.2010.01.063
URL
PMID:20097171
[本文引用: 1]

The gene of (all-E) geranylfarnesyl diphosphate synthase that is responsible for the biosynthesis of methanophenazine, an electron carrier utilized for methanogenesis, was cloned from a methanogenic archaeon Methanosarcina mazei Go1. The properties of the recombinant enzyme and the results of phylogenetic analysis suggest that the enzyme is closely related to (all-E) prenyl diphosphate synthases that are responsible for the biosynthesis of respiratory quinones, rather than to the enzymes involved in the biosynthesis of archaeal membrane lipids, including (all-E) geranylfarnesyl diphosphate synthase from a thermophilic archaeon.
Arabidopsis thaliana isoprenyl diphosphate synthases produce the C25 intermediate geranylfarnesyl diphosphate
[J].
DOI:10.1111/tpj.13064
URL
PMID:26505977
[本文引用: 2]

Isoprenyl diphosphate synthases (IDSs) catalyze some of the most basic steps in terpene biosynthesis by producing the prenyl diphosphate precursors of each of the various terpenoid classes. Most plants investigated have distinct enzymes that produce the short-chain all-trans (E) prenyl diphosphates geranyl diphosphate (GDP, C10 ), farnesyl diphosphate (FDP, C15 ) or geranylgeranyl diphosphate (GGDP, C20 ). In the genome of Arabidopsis thaliana, 15 trans-product-forming IDSs are present. Ten of these have recently been shown to produce GGDP by genetic complementation of a carotenoid pathway engineered into Escherichia coli. When verifying the product pattern of IDSs producing GGDP by a new LC-MS/MS procedure, we found that five of these IDSs produce geranylfarnesyl diphosphate (GFDP, C25 ) instead of GGDP as their major product in enzyme assays performed in vitro. Over-expression of one of the GFDP synthases in A. thaliana confirmed the production of GFDP in vivo. Enzyme assays with A. thaliana protein extracts from roots but not other organs showed formation of GFDP. Furthermore, GFDP itself was detected in root extracts. Subcellular localization studies in leaves indicated that four of the GFDP synthases were targeted to the plastoglobules of the chloroplast and one was targeted to the mitochondria. Sequence comparison and mutational studies showed that the size of the R group of the 5th amino acid residue N-terminal to the first aspartate-rich motif is responsible for C25 versus C20 product formation, with smaller R groups (Ala and Ser) resulting in GGDP (C20 ) as a product and a larger R group (Met) resulting in GFDP (C25 ).
Structural analyses of short-chain prenyltransferases identify an evolutionarily conserved GFPPS clade in Brassicaceae plants
[J].
DOI:10.1016/j.molp.2015.10.010
URL
PMID:26537048
[本文引用: 1]

Terpenoids are the largest and most diverse class of plant-specialized metabolites, which function in diverse physiological processes during plant development. In the biosynthesis of plant terpenoids, short-chain prenyltransferases (SC-PTs), together with terpene synthases (TPSs), play critical roles in determining terpenoid diversity. SC-PTs biosynthesize prenyl pyrophosphates with different chain lengths, and these compounds are the direct precursors of terpenoids. Arabidopsis thaliana possesses a subgroup of SC-PTs whose functions are not clearly known. In this study, we focus on 10 geranylgeranyl pyrophosphate synthase-like [GGPPSL] proteins, which are commonly thought to produce GGPP [C20]. We found that a subset of members of the Arabidopsis GGPPSL gene family have undergone neo-functionalization: GGPPSL6, 7, 9, and 10 mainly have geranylfarnesyl pyrophosphate synthase activity (C25; renamed AtGFPPS1, 2, 3, and 4), and GGPPSL8 produces even longer chain prenyl pyrophosphate (>/= C30; renamed polyprenyl pyrophosphate synthase 2, AtPPPS2). By solving the crystal structures of AtGFPPS2, AtPPPS2, and AtGGPPS11, we reveal the product chain-length determination mechanism of SC-PTs and interpret it as a
A geranylfarnesyl diphosphate synthase provides the precursor for sesterterpenoid(C25)formation in the glandular trichomes of the mint species Leucosceptrum canum
[J].
DOI:10.1105/tpc.15.00715
URL
PMID:26941091
[本文引用: 3]

Plant sesterterpenoids, an important class of terpenoids, are widely distributed in various plants, including food crops. However, little is known about their biosynthesis. Here, we cloned and functionally characterized a plant geranylfarnesyl diphosphate synthase (Lc-GFDPS), the enzyme producing the C25 prenyl diphosphate precursor to all sesterterpenoids, from the glandular trichomes of the woody plant Leucosceptrum canum. GFDPS catalyzed the formation of GFDP after expression in Escherichia coli. Overexpressing GFDPS in Arabidopsis thaliana also gave an extract catalyzing GFDP formation. GFDPS was strongly expressed in glandular trichomes, and its transcript profile was completely in accordance with the sesterterpenoid accumulation pattern. GFDPS is localized to the plastids, and inhibitor studies indicated its use of isoprenyl diphosphate substrates supplied by the 2-C-methyl-D-erythritol 4-phosphate pathway. Application of a jasmonate defense hormone induced GFDPS transcript and sesterterpenoid accumulation, while reducing feeding and growth of the generalist insect Spodoptera exigua, suggesting that these C25 terpenoids play a defensive role. Phylogenetic analysis suggested that GFDPS probably evolved from plant geranylgeranyl diphosphate synthase under the influence of positive selection. The isolation of GFDPS provides a model for investigating sesterterpenoid formation in other species and a tool for manipulating the formation of this group in plants and other organisms.
Thermostable farnesyl diphosphate synthase of Bacillus stearothermophilus:molecular cloning, sequence determination, overproduction, and purification
[J].
DOI:10.1093/oxfordjournals.jbchem.a124051
URL
PMID:8486607
[本文引用: 1]

The structural gene for thermostable farnesyl diphosphate synthase from Bacillus stearothermophilus was cloned, sequenced, and overexpressed in Escherichia coli cells. A 1,260-nucleotide sequence of the cloned fragment was determined. This sequence specifies an open reading frame of 891 nucleotides for farnesyl diphosphate synthase. The deduced amino acid sequence shows a 42% similarity with that of E. coli FPP synthase [Fujisaki et al. (1990) J. Biochem. 108, 995-1000]. Comparison with prenyltransferases from a wide range of organisms, from bacteria to human, revealed the presence of seven highly conserved regions. In contrast to thermolabile prenyltransferases, which have four to six cysteine residues, the thermostable farnesyl diphosphate synthase carries only two cysteine residues. This enzyme is also unique in that some of the amino acids that are fully conserved in equivalents from other sources are replaced by functionally different amino acids. Construction of an overproducing strain provided a sufficient supply of this enzyme and it was purified to homogeneity. The purified recombinant enzyme is immunochemically identical with the native B. stearothermophilus enzyme, and it is not inactivated even after treatment at 65 degrees C for 70 min.
Isoprenyl diphosphate synthases:protein sequence comparisons, a phylogenetic tree, and predictions of secondary structure
[J].
DOI:10.1002/pro.5560030408
URL
PMID:8003978
[本文引用: 1]

Isoprenyl diphosphate synthases are ubiquitous enzymes that catalyze the basic chain-elongation reaction in the isoprene biosynthetic pathway. Pairwise sequence comparisons were made for 6 farnesyl diphosphate synthases, 6 geranylgeranyl diphosphate synthases, and a hexaprenyl diphosphate synthase. Five regions with highly conserved residues, two of which contain aspartate-rich DDXX(XX)D motifs found in many prenyltransferases, were identified. A consensus secondary structure for the group, consisting mostly of alpha-helices, was predicted for the multiply aligned sequences from amino acid compositions, computer assignments of local structure, and hydropathy indices. Progressive sequence alignments suggest that the 13 isoprenyl diphosphate synthases evolved from a common ancestor into 3 distinct clusters. The most distant separation is between yeast hexaprenyl diphosphate synthetase and the other enzymes. Except for the chromoplastic geranylgeranyl diphosphate synthase from Capsicum annuum, the remaining farnesyl and geranylgeranyl diphosphate synthases segregate into prokaryotic/archaebacterial and eukaryotic families.
Identification of significant residues in the substrate binding site of Bacillus stearothermophilus farnesyl diphosphate synthase
[J].
DOI:10.1021/bi960137v
URL
PMID:8755734
[本文引用: 1]

Farnesyl diphosphate synthases have been shown to possess seven highly conserved regions (I-VII) in their amino acid sequences [Koyama et al. (1993) J. Biochem. (Tokyo) 113, 355-363]. Site-directed mutants of farnesyl diphosphate synthase from Bacillus stearothermophilus were made to evaluate the roles of the conserved aspartic acids in region VI and lysines in regions I, V, and VI. The aspartate at position 224 was changed to alanine or glutamate (mutants designated as D224A and D224E, respectively); aspartates at positions 225 and 228 were changed to isoleucine and alanine (D225I, D228A); lysine at position 238 was changed to either alanine or arginine (K238A, K238R). The lysines at positions 47 and 183 were changed to isoleucine and alanine (K471, K183A), respectively. Kinetic analyses of the wild-type and mutant enzymes indicated that the mutagenesis of Asp-224 and Asp-225 resulted in a decrease of Kcat values of approximately 10(4)- to 10(5)-fold compared to the wild type. On the other hand, D228A showed a Kcat value approximately one-tenth of that of the wild type, and the k(m) value for isopentenyl diphosphate increased approximately 10-fold. Both K471 and K183A showed k(m) values for isopentenyl diphosphate 20-fold larger and kcat values 70-fold smaller than the wild type. These results suggest that the two conserved lysines in regions I and V contribute to the binding of isopentenyl diphosphate and that the first and the second aspartates in region VI are involved in catalytic function. Aspartate-228 is also important for the binding of isopentenyl diphosphate rather than for catalytic reaction.
Farnesyl diphosphate synthase;regulation of product specificity
[J].
Evolution and biogeography of gymnosperms
[J].
DOI:10.1016/j.ympev.2014.02.005
URL
[本文引用: 1]

Living gymnosperms comprise only a little more than 1000 species, but represent four of the five main lineages of seed plants, including cycads, ginkgos, gnetophytes and conifers. This group has huge ecological and economic value, and has drawn great interest from the scientific community. Here we review recent advances in our understanding of gymnosperm evolution and biogeography, including phylogenetic relationships at different taxonomic levels, patterns of species diversification, roles of vicariance and dispersal in development of intercontinental disjunctions, modes of molecular evolution in different genomes and lineages, and mechanisms underlying the formation of large nuclear genomes. It is particularly interesting that increasing evidence supports a sister relationship between Gnetales and Pinaceae (the Gnepine hypothesis) and the contribution of recent radiations to present species diversity, and that expansion of retrotransposons is responsible for the large and complex nuclear genome of gymnosperms. In addition, multiple coniferous genera such as Picea very likely originated in North America and migrated into the Old World, further indicating that the center of diversity is not necessarily the place of origin. The Bering Land Bridge acted as an important pathway for dispersal of gymnosperms in the Northern Hemisphere. Moreover, the genome sequences of conifers provide an unprecedented opportunity and an important platform for the evolutionary studies of gymnosperms, and will also shed new light on evolution of many important gene families and biological pathways in seed plants. (c) 2014 Elsevier Inc.
1-Hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase(IDS)is encoded by multicopy genes in gymnosperms Ginkgo biloba and Pinus taeda
[J].
Validation of DNA barcoding as an efficient tool for taxon identification and detection of species diversity in Italian conifers
[J].
马尾松材性与产脂性状遗传改良研究
[D].
Genetic improvement of key wood and resin properties of Pinus massoniana Lamb
[D].
Resin-based defenses in conifers
[J].
DOI:10.1016/s1360-1385(99)01401-6
URL
PMID:10322558
[本文引用: 1]

Bark beetle infestation and associated fungal infection are a serious disease problem in conifer species. Conifers have evolved elaborate, constitutive and inducible, terpene-based defense mechanisms to deter insect pests and their symbiotic fungal pathogens. This process involves the secretion of oleoresin, a complex mixture of monoterpenes, sesquiterpenes and diterpenoid acids. Induced oleoresinosis in grand fir (Abies grandis) provides a model system for studying the regulation of defensive terpene biosynthesis and for identifying relevant genes. The ecological relationships between conifers, beetle pests, beetle predators and fungal pathogens present several possible avenues for manipulating oleoresin composition to improve tree resistance. Possible examples include chemically disguising the host, adding toxins and altering the levels of pheromone precursors, attractants for predators or hormone mimics to disrupt insect development. Strategies and prospects for generating transgenic conifers with increased defense capability are discussed.
Diterpene resin acids in conifers
[J].
Genes, enzymes and chemicals of terpenoid diversity in the constitutive and induced defence of conifers against insects and pathogens
[J].
DOI:10.1111/j.1469-8137.2006.01716.x
URL
PMID:16684230
[本文引用: 1]

Insects select their hosts, but trees cannot select which herbivores will feed upon them. Thus, as long-lived stationary organisms, conifers must resist the onslaught of varying and multiple attackers over their lifetime. Arguably, the greatest threats to conifers are herbivorous insects and their associated pathogens. Insects such as bark beetles, stem- and wood-boring insects, shoot-feeding weevils, and foliage-feeding budworms and sawflies are among the most devastating pests of conifer forests. Conifer trees produce a great diversity of compounds, such as an enormous array of terpenoids and phenolics, that may impart resistance to a variety of herbivores and microorganisms. Insects have evolved to specialize in resistance to these chemicals -- choosing, feeding upon, and colonizing hosts they perceive to be best suited to reproduction. This review focuses on the plant-insect interactions mediated by conifer-produced terpenoids. To understand the role of terpenoids in conifer-insect interactions, we must understand how conifers produce the wide diversity of terpenoids, as well as understand how these specific compounds affect insect behaviour and physiology. This review examines what chemicals are produced, the genes and proteins involved in their biosynthesis, how they work, and how they are regulated. It also examines how insects and their associated pathogens interact with, elicit, and are affected by conifer-produced terpenoids.
Terpenoid biosynjournal and specialized vascular cells of conifer defense
[J].
DOI:10.1111/j.1744-7909.2010.00910.x
URL
PMID:20074143
[本文引用: 1]

Defense-related terpenoid biosynthesis in conifers is a dynamic process closely associated with specialized anatomical structures that allows conifers to cope with attack from many potential pests and pathogens. The constitutive and inducible terpenoid defense of conifers involves several hundred different monoterpenes, sesquiterpenes and diterpenes. Changing arrays of these many compounds are formed from the general isoprenoid pathway by activities of large gene families for two classes of enzymes, the terpene synthases and the cytochrome P450-dependent monooxygenases of the CYP720B group. Extensive studies have been conducted on the genomics, proteomics and molecular biochemical characterization of these enzymes. Many of the conifer terpene synthases are multi-product enzymes, and the P450 enzymes of the CYP720B group are promiscuous in catalyzing multiple oxidations, along homologous series of diterpenoids, from a broad spectrum of substrates. The terpene synthases and CYP720B genes respond to authentic or simulated insect attack with increased transcript levels, protein abundance and enzyme activity. The constitutive and induced oleoresin terpenoids for conifer defense accumulate in preformed cortical resin ducts and in xylem trauma-associated resin ducts. Formation of these resin ducts de novo in the cambium zone and developing xylem, following insect attack or treatment of trees with methyl jasmonate, is a unique feature of the induced defense of long-lived conifer trees.
Efficacy of tree defense physiology varies with bark beetle population density:a basis for positive feedback in eruptive species
[J].
湿地松采脂负荷大小对树木生长影响试验
[J].
Test on the influence of slash pine resin load on tree growth
[J].
马尾松天然林采脂试验分析
[J].
Analysis of tapping resin trial in natural stands of masson’s pine
[J].
High-density renewable fuels based on the selective dimerization of pinenes
[J].
Oleoresin characteristics of progeny of loblolly pines that escaped attack by the southern pine beetle
[J].
Molecular biochemistry and genomics of terpenoid defenses in conifers
[J].
Insect-induced conifer defense. White pine weevil and methyl jasmonate induce traumatic resinosis, de novo formed volatile emissions, and accumulation of terpenoid synthase and putative octadecanoid pathway transcripts in Sitka spruce
[J].
Wound-induced traumatic resin duct development in stems of Norway spruce(Pinaceae):anatomy and cytochemical traits
[J].Wounding of Norway spruce by inoculation with sterile agar, or agar containing the pathogenic fungus Ceratocystis polonica, induced traumatic resin duct formation in the stem. Visible anatomical responses occurred in the cambium 6-9 d post-inoculation. Near the inoculation site cellular proliferation, polyphenolic accumulation, and lignification were induced as a wound reaction to seal the damaged area. Five centimetres from the inoculation site cells in the cambial zone swelled and divided to form clusters. By 18 d post-inoculation, these cells began to differentiate into resin duct epithelial cells surrounding incipient schizogenous lumens. Mature axial traumatic ducts appeared by 36 d as a row of ducts in the xylem centripetal to the cambium. The ducts formed an interconnected network continuous with radial resin ducts. Parenchyma cells surrounding the ducts accumulated polyphenols that disappeared as the cells differentiated into tracheids. These polyphenols appeared to contain fewer sugar residues compared to those accumulating in the secondary phloem, as indicated by the periodic acid-Schiff's staining. The epithelial cells did not accumulate polyphenols but contained immunologically detectable phenylalanine ammonia lyase (EC 4.3.1.5), indicating synthesis of phenolics as a possible resin component. These findings may represent a defense mechanism in Norway spruce against the pathogenic fungus Ceratocystis polonica.
Insect attack and wounding induce traumatic resin duct development and gene expression of(-)-pinene synthase in Sitka spruce
[J].
DOI:10.1104/pp.103.022723
URL
PMID:12970502
[本文引用: 1]

Conifers possess inducible terpenoid defense systems. These systems are associated with the formation of traumatic resin ducts (TRD) and are underpinned by enhanced gene expression and activity of terpene synthases (TPS), enzymes responsible for oleoresin formation. We first determined that Sitka spruce (Picea sitchensis [Bong.] Carriere) had the capacity for TRD formation by mechanically wounding representative trees. We then proceeded to investigate whether the white pine weevil (Pissodes strobi Peck.), a stem-boring insect, can influence the expression of genes encoding monoterpene synthases (mono-tps) in Sitka spruce. We went on to compare this response with the effects of a simulated insect attack by drill wounding. A significant increase in mono-tps transcript level was observed in the leaders of lateral branches of weevil-attacked and mechanically wounded trees. In this study, weevils induced a more rapid enhancement of mono-tps gene expression. A full-length Sitka spruce mono-tps cDNA (PsTPS2) was isolated, expressed in Escherichia coli, and functionally identified as (-)-pinene synthase. The recombinant (-)-pinene synthase catalyzes the formation of (-)-alpha-pinene and (-)-beta-pinene, both of which are known constituents of stem oleoresin in Sitka spruce and increase in abundance after weevil attack. These data suggest that increased (-)-pinene synthase gene expression is an important element of the direct defense system deployed in Sitka spruce after insect attack.
Cloning and functional analysis of a cDNA encoding Ginkgo biloba farnesyl diphosphate synthase
[J].Farnesyl diphosphate synthase (FPS; EC2.5.1.1/EC2. 5.1.10) catalyzes the synthesis of farnesyl diphosphate, and provides precursor for biosynthesis of sesquiterpene and isoprenoids containing more than 15 isoprene units in Ginkgo biloba. Here we report the cloning, characterization and functional analysis of a new cDNA encoding FPS from G. biloba. The full-length cDNA (designated GbFPS) had 1731 bp with an open reading frame of 1170 bp encoding a polypeptide of 390 amino acids. The deduced GbFPS was similar to other known FPSs and contained all the conserved regions of trans-prenyl chain-elongating enzymes. Structural modeling showed that GbFPS had the typical structure of FPS, the most prominent feature of which is the arrangement of 13 core helices around a large central cavity. Southern blot analysis revealed a small FPS gene family in G. biloba. Expression analysis indicated that GbFPS expression was high in roots and leaves, and low in stems. Functional complementation of GbFPS in an FPS-deficient strain confirmed that GbFPS mediates farnesyl diphosphate biosynthesis.
Overexpression of an isoprenyl diphosphate synthase in spruce leads to unexpected terpene diversion products that function in plant defense
[J].
DOI:10.1104/pp.113.228940
URL
PMID:24346420
[本文引用: 2]

Spruce (Picea spp.) and other conifers employ terpenoid-based oleoresin as part of their defense against herbivores and pathogens. The short-chain isoprenyl diphosphate synthases (IDS) are situated at critical branch points in terpene biosynthesis, producing the precursors of the different terpenoid classes. To determine the role of IDS and to create altered terpene phenotypes for assessing the defensive role of terpenoids, we overexpressed a bifunctional spruce IDS, a geranyl diphosphate and geranylgeranyl diphosphate synthase in white spruce (Picea glauca) saplings. While transcript level (350-fold), enzyme activity level (7-fold), and in planta geranyl diphosphate and geranylgeranyl diphosphate levels (4- to 8-fold) were significantly increased in the needles of transgenic plants, there was no increase in the major monoterpenes and diterpene acids of the resin and no change in primary isoprenoids, such as sterols, chlorophylls, and carotenoids. Instead, large amounts of geranylgeranyl fatty acid esters, known from various gymnosperm and angiosperm plant species, accumulated in needles and were shown to act defensively in reducing the performance of larvae of the nun moth (Lymantria monacha), a conifer pest in Eurasia. These results show the impact of overexpression of an IDS and the defensive role of an unexpected accumulation product of terpenoid biosynthesis with the potential for a broader function in plant protection.
Cloning and characterization of isoprenyl diphosphate synthases with farnesyl diphosphate and geranylgeranyl diphosphate synthase activity from Norway spruce(Picea abies)and their relation to induced oleoresin formation
[J].
A new farnesyl diphosphate synthase gene from Taxus media rehder:cloning, characterization and functional complementation
[J].
Geranyl diphosphate synthase from Abies grandis:cDNA isolation, functional expression, and characterization
[J].
DOI:10.1016/s0003-9861(02)00335-1
URL
PMID:12176066
[本文引用: 2]

Geranyl diphosphate synthase catalyzes the condensation of dimethylallyl diphosphate and isopentenyl diphosphate to generate geranyl diphosphate, the essential precursor of monoterpene biosynthesis. Using geranylgeranyl diphosphate synthase from Taxus canadensis as a hybridization probe, four full length cDNA clones, sharing high sequence identity to each other (>69%) and to the Taxus geranylgeranyl diphosphate synthase (>66%), were isolated from a grand fir (Abies grandis) cDNA library. When expressed in Escherichia coli, three of the recombinant enzymes produced geranyl diphosphate and one produced geranylgeranyl diphosphate as the dominant product when supplied with isopentenyl diphosphate and dimethylallyl diphosphate as cosubstrates. One enzyme (AgGPPS2) was confirmed as a specific geranyl diphosphate synthase, in that it accepted only dimethylallyl diphosphate as the allylic cosubstrate and it produced exclusively geranyl diphosphate as product, with a k(cat) of 1.8s(-1). Gel filtration experiments performed on the recombinant geranyl diphosphate synthases, in which the plastidial targeting sequences had been deleted, revealed that these enzymes are homodimers similar to other short-chain prenyltransferases but different from the heterotetrameric geranyl diphosphate synthase of mint.
Interaction with the small subunit of geranyl diphosphate synthase modifies the chain length specificity of geranylgeranyl diphosphate synthase to produce geranyl diphosphate
[J].
Cloning and functional expression of a cDNA encoding geranylgeranyl diphosphate synthase from Taxus canadensis and assessment of the role of this prenyltransferase in cells induced for taxol production
[J].
DOI:10.1006/abbi.1998.0926
URL
PMID:9826430
[本文引用: 2]

Geranylgeranyl diphosphate synthase supplies the essential acyclic precursor for Taxol biosynthesis in methyl jasmonate-induced Taxus canadensis suspension cell cultures. A cDNA encoding this prenyltransferase was cloned from an induced T. canadensis cell library. The recombinant enzyme expressed in yeast was confirmed by radiochromatographic analysis to produce geranylgeranyl diphosphate from farnesyl diphosphate and [4-14C]isopentenyl diphosphate and was subjected to preliminary kinetic characterization. The deduced amino acid sequence of this gymnosperm geranylgeranyl diphosphate synthase (393 residues) resembles those of geranylgeranyl diphosphate synthases of angiosperm origin, except for the 90-100 N-terminal residues that correspond to the plastidial transit peptide. The full-length preprotein (42.6 kDa) and two truncated versions, corresponding to putative
马尾松GGPPS基因克隆及序列分析
[J].
Cloning and bioinformatics analysis of GGPPS of Pinus massoniana
[J].
海南粗榧GGPPs基因克隆与诱导表达分析
[J].
Cloning and induced expression analysis of GGPP synthase gene from Cephalotaxus mannii
[J].
Oleoresin defenses in conifers:chemical diversity, terpene synthases and limitations of oleoresin defense under climate change
[J].