








生物技术通报 ›› 2026, Vol. 42 ›› Issue (2): 77-88.doi: 10.13560/j.cnki.biotech.bull.1985.2025-1382
• 厌氧微生物专题(专题主编:承磊 研究员) • 上一篇 下一篇
收稿日期:2025-12-17
出版日期:2026-02-26
发布日期:2026-03-17
通讯作者:
李凌燕,博士,副研究员,研究方向 :产甲烷古菌资源、生理代谢及环境适应机制;E-mail: lilingyan@im.ac.cn作者简介:王雪萌,博士研究生,研究方向 :土壤微生物生态;E-mail: wangxuemeng22@mails.ucas.ac.cn
基金资助:
WANG Xue-meng1( ), DONG Xiu-zhu2, XUE Kai1(
), DONG Xiu-zhu2, XUE Kai1( ), LI Ling-yan2(
), LI Ling-yan2( )
)
Received:2025-12-17
Published:2026-02-26
Online:2026-03-17
摘要:
目的 探究直接种间电子转移(direct interspecies electron transfer, DIET)机制在增温下青藏高原若尔盖泥炭地甲烷排放中的作用,探索全球变暖背景下基于DIET机制的甲烷减排措施。 方法 以若尔盖泥炭地3种优势植物根部土壤为研究对象,构建室内土壤果胶和纤维素厌氧富集物(18 ℃),通过添加电子传输介质——生物炭及电化学实验,探究直接种间电子转移对植物有机碳来源产甲烷过程的作用及增温(28 ℃)对甲烷产生的效果。 结果 添加生物炭提高了18 ℃培养物从纤维素和果胶来源的甲烷产量1.1-9.8倍,提高了产甲烷速率1.4-12.3倍;添加生物炭后温度敏感性提高了2倍。计时电流法检测到电化学培养物中从细菌产生的胞外电子,其电流密度为33.7 μA/cm2,及产甲烷古菌介导的电流消耗(21.2 μA/cm2)。16S rRNA基因多样性分析发现,生物炭添加显著富集了已知的电活性细菌——地杆菌科(Geobacteraceae)和产甲烷古菌——甲烷八叠球菌科(Methanosarcinaceae)、嗜甲基甲烷菌科(Methanomethylophilaceae)及未培养的产甲烷古菌类群Rice Cluster Ⅱ。泥炭地中的Geobacteraceae和纤维素单胞菌科(Cellulomonadaceae)均与Rice Cluster Ⅱ具有显著共现性,推测它们可能通过直接种间电子转移介导植物有机碳产甲烷的过程。 结论 直接种间电子转移机制在若尔盖泥炭地植物有机碳产甲烷过程中发挥重要作用,尤其在增温下该作用更显著。因此抑制种间电子转移是泥炭地甲烷减排的措施之一。
王雪萌, 东秀珠, 薛凯, 李凌燕. 增温下直接种间电子转移对泥炭地甲烷产生的作用[J]. 生物技术通报, 2026, 42(2): 77-88.
WANG Xue-meng, DONG Xiu-zhu, XUE Kai, LI Ling-yan. Role of Direct Interspecies Electron Transfer in the Methane Production of the Peatland under Warming[J]. Biotechnology Bulletin, 2026, 42(2): 77-88.
优势植物 Dominating plant | 经纬度 Longitude and latitude | 土壤温度 Soil temperature (℃) | 静水深度 standing water depth (cm) |
|---|---|---|---|
| 刚毛荸荠 E. valleculosa | 33°55′12″ N, 102°49′12″ E | 17.7 | 28.7 |
| 西藏嵩草 K. tibetica | 32°20′37″ N, 102°26′36″ E | 18.3 | 28.7 |
| 木里苔草 C. muliensis | 32°33′6″ N, 102°21′15″ E | 15.5 | 30.4 |
表1 本研究采集的青藏高原若尔盖泥炭地土壤地理特征
Table 1 Geographic characteristics of the soils sampled from Zoige peatland of the Tibetan Plateau in this study
优势植物 Dominating plant | 经纬度 Longitude and latitude | 土壤温度 Soil temperature (℃) | 静水深度 standing water depth (cm) |
|---|---|---|---|
| 刚毛荸荠 E. valleculosa | 33°55′12″ N, 102°49′12″ E | 17.7 | 28.7 |
| 西藏嵩草 K. tibetica | 32°20′37″ N, 102°26′36″ E | 18.3 | 28.7 |
| 木里苔草 C. muliensis | 32°33′6″ N, 102°21′15″ E | 15.5 | 30.4 |
| 无机元素储液 Inorganic element solution | 维生素储液 Vitamin solution | 微量元素储液 Trace element solution | |||
|---|---|---|---|---|---|
试剂 Reagent | 浓度 Concentration (g/L) | 试剂 Reagent | 浓度 Concentration (g/L) | 试剂 Reagent | 浓度 Concentration (g/L) |
| NH4Cl | 6 | 生物素 | 0.002 | FeCl2·4H2O | 2 |
| NaCl | 6 | 叶酸 | 0.002 | ZnCl2 | 0.05 |
| CaCl2·2H2O | 0.2 | VB6 | 0.01 | MnCl2·4H2O | 0.05 |
| MgCl2·6H2O | 2 | VB2 | 0.005 | CuCl2·2H2O | 0.03 |
| VB1 | 0.005 | (NH4)6Mo7O24·4H2O | 0.05 | ||
| VB3 | 0.005 | AlCl3 | 0.05 | ||
| VB12 | 0.005 | CoCl2·6H2O | 0.2 | ||
| 泛酸 | 0.005 | H3BO3饱和溶液 | 1 mL | ||
| 对氨基苯甲酸 | 0.005 | 浓HCl | 1 mL | ||
表2 无机元素-维生素-微量元素混合溶液
Table 2 Inorganic element-vitamin-trace element solution
| 无机元素储液 Inorganic element solution | 维生素储液 Vitamin solution | 微量元素储液 Trace element solution | |||
|---|---|---|---|---|---|
试剂 Reagent | 浓度 Concentration (g/L) | 试剂 Reagent | 浓度 Concentration (g/L) | 试剂 Reagent | 浓度 Concentration (g/L) |
| NH4Cl | 6 | 生物素 | 0.002 | FeCl2·4H2O | 2 |
| NaCl | 6 | 叶酸 | 0.002 | ZnCl2 | 0.05 |
| CaCl2·2H2O | 0.2 | VB6 | 0.01 | MnCl2·4H2O | 0.05 |
| MgCl2·6H2O | 2 | VB2 | 0.005 | CuCl2·2H2O | 0.03 |
| VB1 | 0.005 | (NH4)6Mo7O24·4H2O | 0.05 | ||
| VB3 | 0.005 | AlCl3 | 0.05 | ||
| VB12 | 0.005 | CoCl2·6H2O | 0.2 | ||
| 泛酸 | 0.005 | H3BO3饱和溶液 | 1 mL | ||
| 对氨基苯甲酸 | 0.005 | 浓HCl | 1 mL | ||
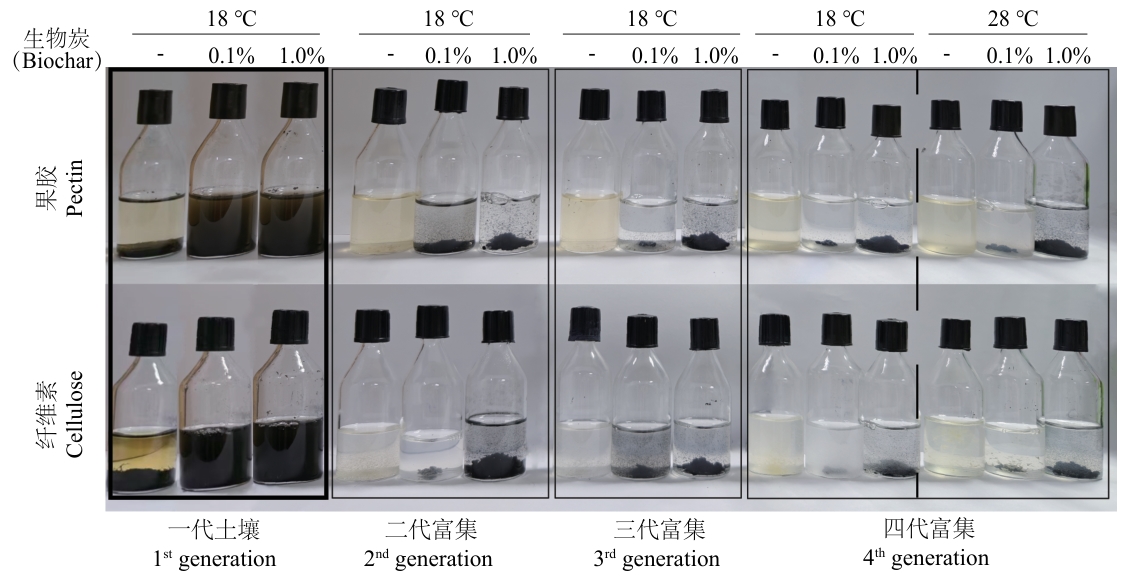
图1 若尔盖泥炭地土壤厌氧产甲烷富集培养分别将2.5 g的刚毛荸荠、西藏嵩草和木里苔草土壤接种至25 mL预还原的基础培养基(-)以及添加果胶或纤维素的生物炭(0.1%和1.0%,W/V)厌氧培养基中,于18 ℃静置厌氧培养180 d,检测甲烷,图中以刚毛荸荠土为例进行展示。从每代富集物中提取~10%(V/V)的炭粒进行传代培养,无生物炭(0.0%)的对照组以10%富集液传代,第4代培养物分别培养于18 ℃和28 ℃,各代均培养至180 d时拍照
Fig. 1 Anaerobic methanogenesis enrichment and cultures of the soils from Zoige peatlandA total of 2.5 g soils sampled from E. valleculosa, K. tibetica, and C. muliensis were inoculated into 25 mL pre-reduced basic medium (-) and amended with pectin or cellulose plus biochar (0.1% and 1.0%, W/V). The soil enrichments were incubated anaerobically at 18 ℃ for 180 d, CH4 in the headspace was measured. Data presented in the figure was acquired from E. valleculosa soil. About 10% (V/V) of the biochar was transferred from the enrichments of each generation for subculture. The group without biochar amended (0.0%) was subcultured with 10% (V/V) of the enrichment. The 4th generation soil enrichments were incubated anaerobically at 18 ℃ and 28 ℃ respectively. Soil enrichments from each generation were photographed at the 180-day incubation
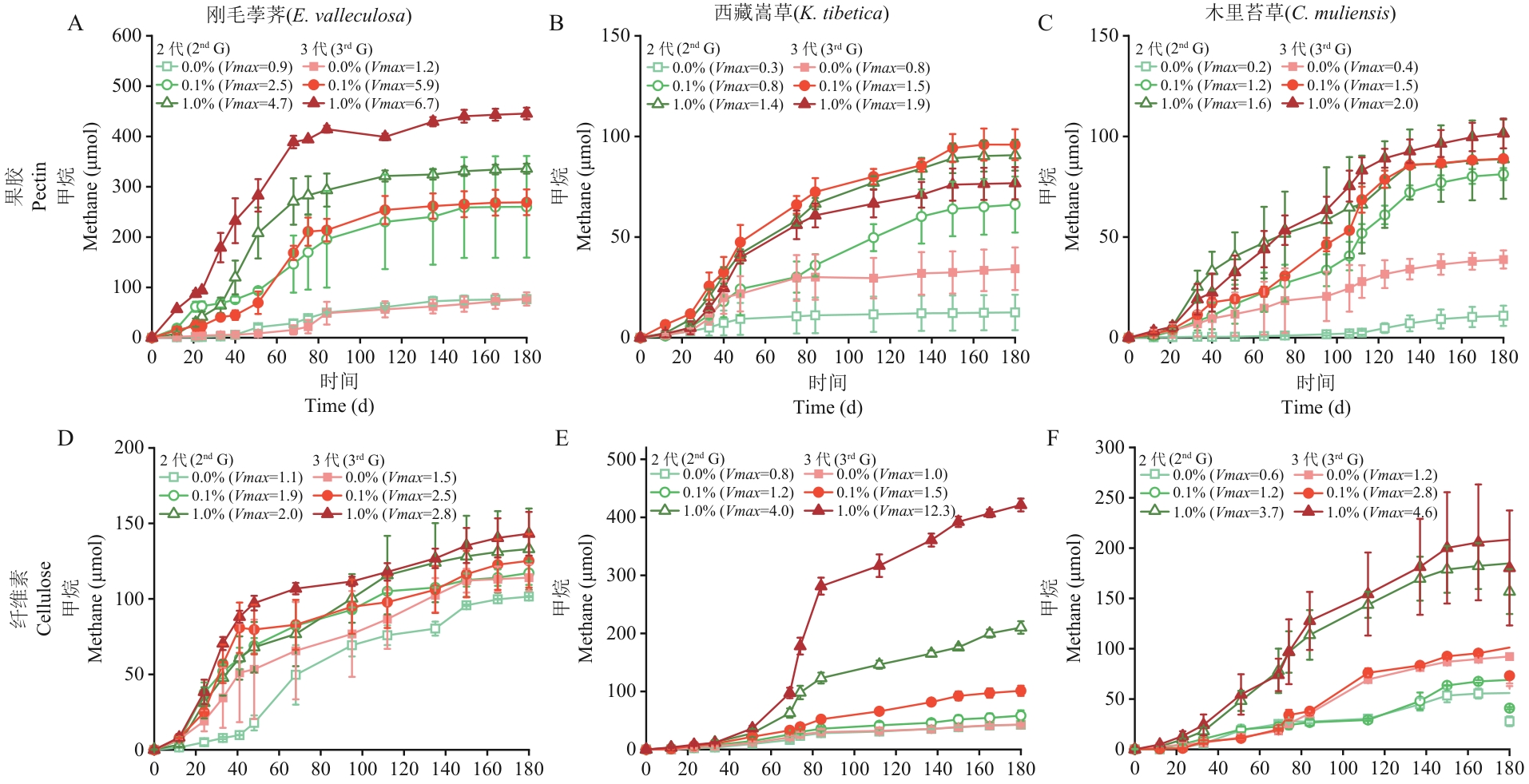
图2 生物炭对泥炭地土壤果胶或纤维素富集物产甲烷的作用添加生物炭的果胶或纤维素富集物以~10%(V/V)的炭粒比例接种至新鲜培养基,无生物炭(0.0%)的富集液以10%接种设置为对照组。在18 ℃条件下培养,定期监测刚毛荸荠(A,D)、西藏嵩草(B,E)和木里苔草(C,F)土壤富集物的顶空甲烷含量;最大产甲烷速率由公式1计算(μmol/d)。数据呈现为平均值±标准误(n=3)
Fig. 2 Effects of biochar on methane production from pectin or cellulose enrichments of peatland soilsAbout 10% (V/V) of the biochar was transferred into fresh medium from the pectin- or cellulose-amended microcosms for subculture. The group without biochar amendment (0.0%) was subcultured with10% (V/V) of the enrichment as the control group. The soil enrichments were incubated anaerobically at 18 ℃ and CH4 in headspace of the soil enrichments from under E. valleculosa (A, D), K. tibetica (B, E), or C. muliensis (C, F) was measured during incubation. The maximum methanogenic rates (Vmax, μmol/d) were calculated from Equation 1. Triplicate cultures were assayed, and the average and standard errors (n=3) are shown
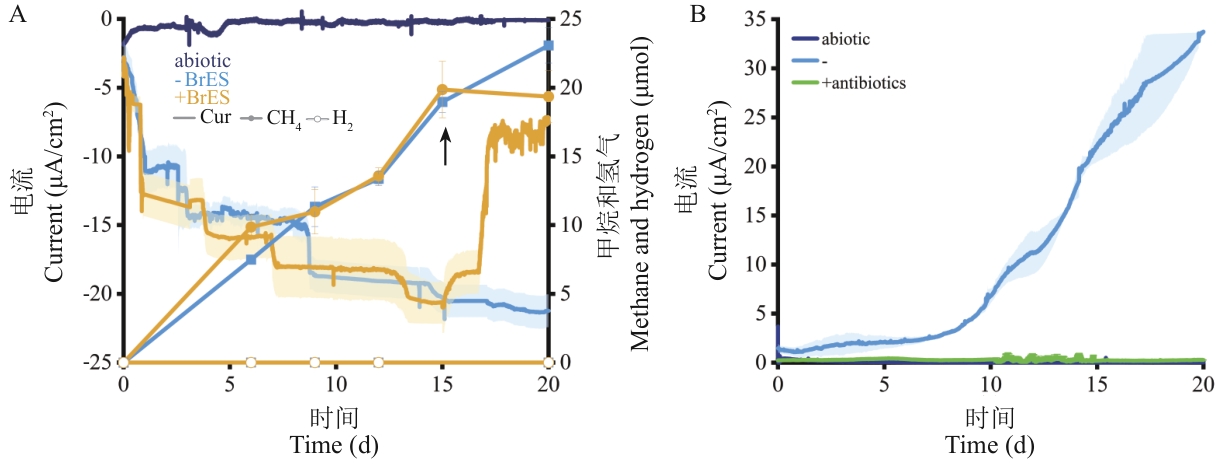
图3 泥炭地土壤富集物电化学实验将9 g刚毛荸荠土壤接种至含有果胶的90 mL预还原基础培养基的H-cell中构建土壤电化学培养物,未接种土壤的电化学室设为非生物对照(abiotic),所有样品均于18 ℃培养。A:阴极室电位设为-0.4 V (vs. Ag/AgCl),在培养第15天时添加10 mmol/L BrES(黑色箭头所示),未添加BrES组为对照组。B:阳极室电压设为+0.4 V (vs. Ag/AgCl),培养期间,每6 d添加1 mg/mL的氨苄霉素和2 mg/mL的卡那霉素,未添加抗生素组(-)的为对照组。定期监测培养物顶空中甲烷和氢气浓度,并实时记录电流,数据呈现为平均值±标准误(n=3)
Fig. 3 Electrochemical experiments from peatland soil enrichmentsElectrochemical soil cultures were constructed by inoculating 9 g soil of E. valleculosa into 90 mL pre-reduced basic medium amended with pectin inside H-cells. The electrochemical chambers without soil inoculation were included as an abiotic control, and anaerobically incubated at 18 ℃. A: The cathodic chambers were set at a potential of -0.4 V (vs. Ag/AgCl), 10 mmol/L BrES was added at day 15 of the incubation (black arrow indicated), and none amendment was included as a blank control. B: The anodic chambers were set at a potential of +0.4 V (vs. Ag/AgCl), 1 mg/mL of ampicillin and 2 mg/mL of kanamycin were amended every six days during incubation, and none amendment (-) was included as a blank control. The contents of CH4 and H2 in headspace of enrichments, and current were monitored during the incubation. Triplicate cultures were assayed, and the average and standard errors (n=3) are presentverifying the effects of DIET on methanogenesis
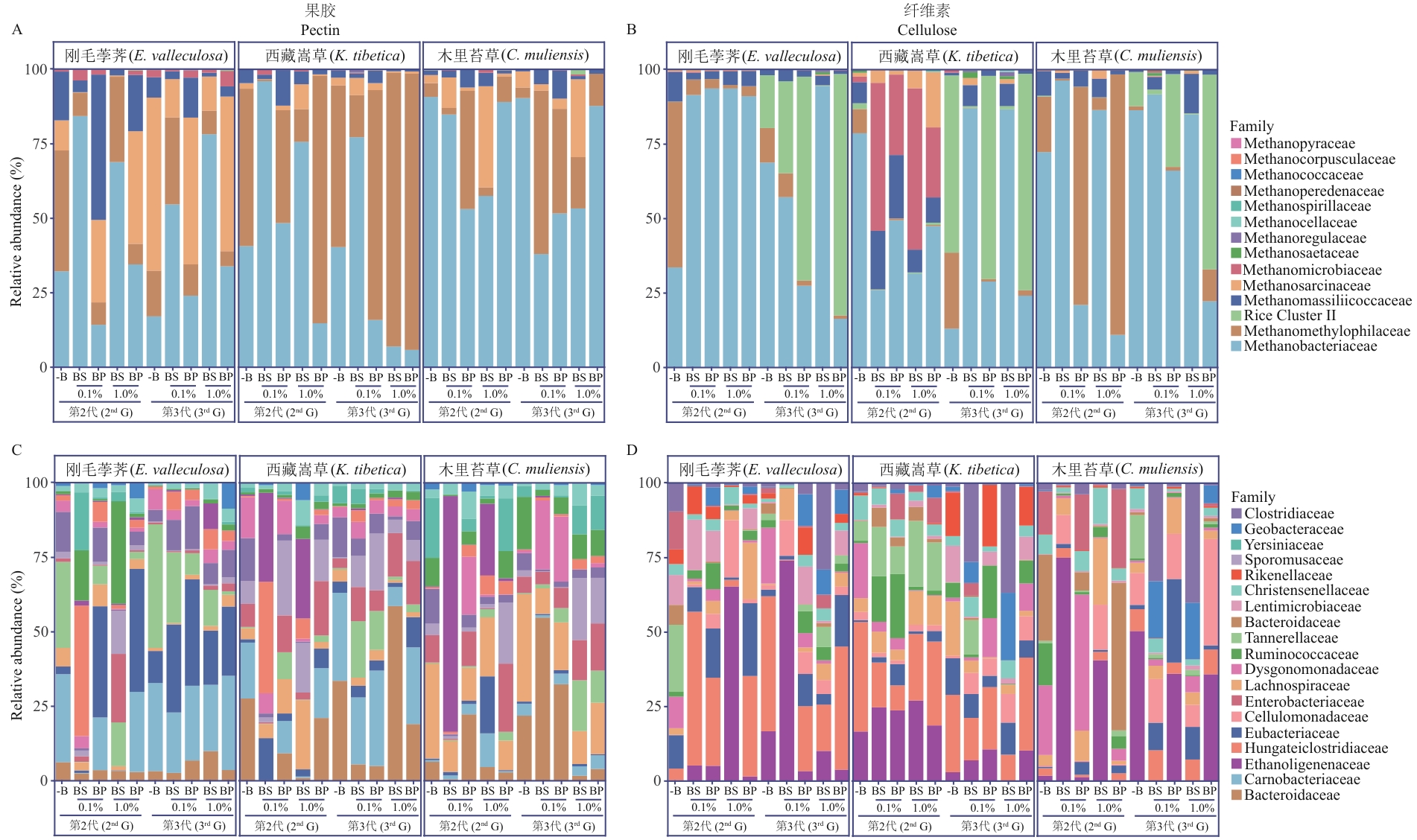
图4 泥炭地土壤果胶及纤维素富集物中产甲烷古菌与细菌相对丰度以图2中第2代和第3代添加生物炭组甲烷产量最高时的果胶及纤维素富集物为研究对象,提取炭粒表面的微生物总DNA进行16S rRNA基因扩增子测序,分析产甲烷古菌(A,B)和细菌(C,D)科水平的物种组成及相对丰度。图中展示了刚毛荸荠、西藏嵩草和木里苔草土壤富集物的所有产甲烷古菌和相对丰度前15的细菌。-B:无生物炭;BS:生物炭培养物的上清液;BP:生物炭培养物的炭粒。数据基于3次生物学重复(n=3)计算相对丰度的平均值
Fig. 4 Relative abundances of methanogenic archaea and bacteria in soil pectin- and cellulose-enrichments of peatland methanogenic archaeaTotal DNA was extracted from the biochar in pectin- and cellulose-amended enrichments with the highest methane production, which were from the 2nd and 3rd generations in Figure 2. 16S rRNA gene amplicons were sequenced, and the relative abundances of methanogens and bacteria were analyzed. The methanogens family (A, B) and top 15 of the most abundant bacterial family (C, D) from the enrichments of E. valleculosa, K. tibetica, or C. muliensis are shown. -B: without biochar amendment; BS: the supernatant of biochar plus pectin soil enrichments; BP: the particle of biochar plus pectin soil enrichments. Triplicate cultures were assayed, and the average (n=3) is shown
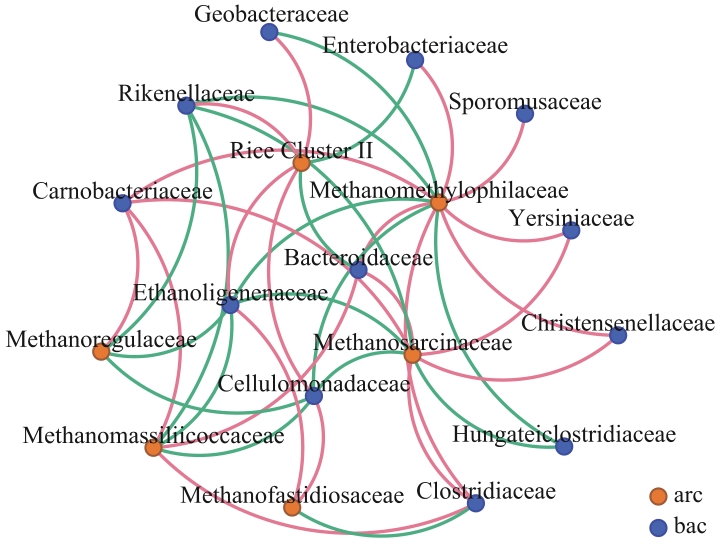
图5 泥炭地土壤产甲烷古菌和细菌的共现网络分析将果胶和纤维素来源的炭粒提取总DNA,获取16S rRNA扩增子测序数据,将所有的产甲烷古菌和相对丰度前15的细菌构建共现网络(n=96)。粉色线条表示物种间呈显著正相关,绿色线条表示物种间呈显著负相关
Fig. 5 Co-occurred network analysis of methanogens and bacteria in peatland soilsTotal DNA was extracted from the biochar of pectin or cellulose amended soil enrichments, respectively, and were used for 16S rRNA amplicon sequencing. Co-occurrence network was constructed incorporating the methanogens family and top 15 of the most abundant bacterial family (n=96). Pink lines indicate significant positive correlations, while green lines indicate significant negative correlations
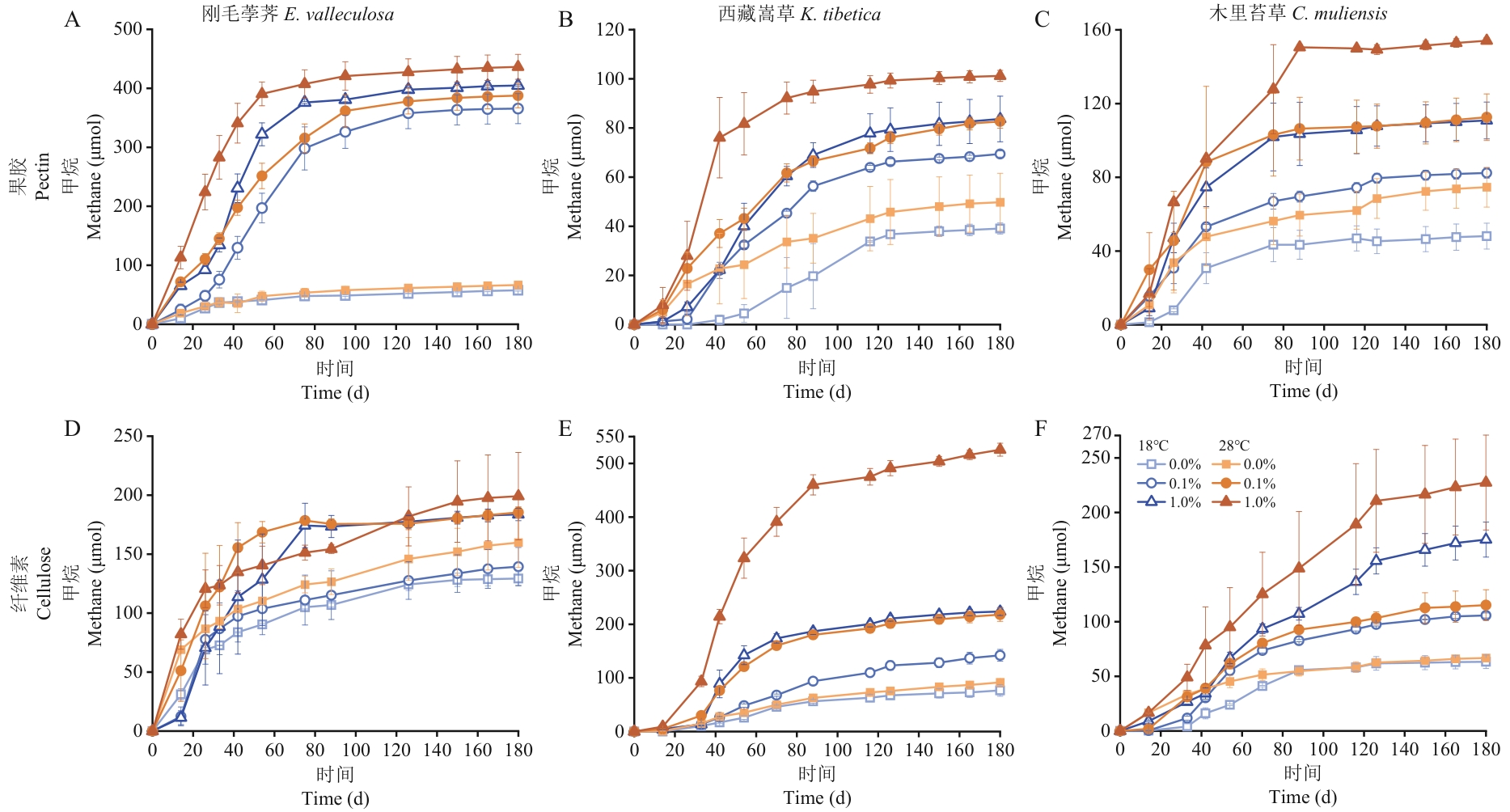
图6 升温对生物炭介导的泥炭地土壤微生物甲烷产生的影响从第3代刚毛荸荠(A,D)、西藏嵩草(B,E)和木里苔草(C,F)土壤培养物中提取~10%(V/V)炭粒比例接种至新鲜的产甲烷培养基,分别在18 ℃和28 ℃培养,定期检测富集物顶空中甲烷产量。数据呈现为平均值±标准误(n=3)
Fig. 6 Effects of temperature increase on biochar-mediated methane production by soil microorganisms in peatlandAbout 10% (V/V) of the biochar extracted from the third-generation soil enrichments of E. valleculosa (A, D), K. tibetica (B, E), and C. muliensis (C, F) were inoculated into fresh methane-producing culture, which then were incubated at 18 ℃ and 28 ℃, respectively. CH4 yield in the headspace of the soil enrichments was measured at given time. The data are present as Mean ± SE (n=3)
土壤富集物 Soil enrichments | 18 ℃ 产甲烷速率 Methanogenic rate at 18 ℃ | 28 ℃ 产甲烷速率 Methanogenic rate at 28 ℃ | ||||
|---|---|---|---|---|---|---|
| - | 0.1% | 1.0% | - | 0.1% | 1.0% | |
| 刚毛荸荠 E. valleculosa | 1.11 | 2.95 | 4.07 | 1.13 | 5.85 | 9.24 |
| 西藏嵩草 K. tibetica | 0.47 | 0.74 | 1.09 | 0.54 | 1.39 | 2.43 |
| 木里苔草 C. muliensis | 1.05 | 1.27 | 2.34 | 1.32 | 2.66 | 4.97 |
表3 生物炭对果胶富集物产甲烷速率的作用
Table 3 Effects of biochar on methanogenic rate from pectin amended soil enrichments (μmol/d)
土壤富集物 Soil enrichments | 18 ℃ 产甲烷速率 Methanogenic rate at 18 ℃ | 28 ℃ 产甲烷速率 Methanogenic rate at 28 ℃ | ||||
|---|---|---|---|---|---|---|
| - | 0.1% | 1.0% | - | 0.1% | 1.0% | |
| 刚毛荸荠 E. valleculosa | 1.11 | 2.95 | 4.07 | 1.13 | 5.85 | 9.24 |
| 西藏嵩草 K. tibetica | 0.47 | 0.74 | 1.09 | 0.54 | 1.39 | 2.43 |
| 木里苔草 C. muliensis | 1.05 | 1.27 | 2.34 | 1.32 | 2.66 | 4.97 |
土壤富集物 Soil enrichments | 18 ℃产甲烷速率 Methanogenic rate at 18 ℃ | 28 ℃ 产甲烷速率 Methanogenic rate at 28 ℃ | ||||
|---|---|---|---|---|---|---|
| - | 0.1% | 1.0% | - | 0.1% | 1.0% | |
| 刚毛荸荠 E. valleculosa | 1.67 | 1.92 | 2.67 | 2.82 | 4.57 | 5.86 |
| 西藏嵩草 K. tibetica | 0.97 | 1.47 | 4.33 | 0.97 | 4.34 | 10.95 |
| 木里苔草 C. muliensis | 0.94 | 1.11 | 1.59 | 1.01 | 2.25 | 3.26 |
表4 生物炭对纤维素富集物产甲烷速率的作用
Table 4 Effects of biochar on methanogenic rate from cellulose amended soil enrichments (μmol/d)
土壤富集物 Soil enrichments | 18 ℃产甲烷速率 Methanogenic rate at 18 ℃ | 28 ℃ 产甲烷速率 Methanogenic rate at 28 ℃ | ||||
|---|---|---|---|---|---|---|
| - | 0.1% | 1.0% | - | 0.1% | 1.0% | |
| 刚毛荸荠 E. valleculosa | 1.67 | 1.92 | 2.67 | 2.82 | 4.57 | 5.86 |
| 西藏嵩草 K. tibetica | 0.97 | 1.47 | 4.33 | 0.97 | 4.34 | 10.95 |
| 木里苔草 C. muliensis | 0.94 | 1.11 | 1.59 | 1.01 | 2.25 | 3.26 |
| [1] | Malyan SK, Bhatia A, Kumar A, et al. Methane production, oxidation and mitigation: a mechanistic understanding and comprehensive evaluation of influencing factors [J]. Sci Total Environ, 2016, 572: 874-896. |
| [2] | Zhang Z, Poulter B, Feldman AF, et al. Recent intensification of wetland methane feedback [J]. Nat Clim Chang, 2023, 13(5): 430-433. |
| [3] | Chen HY, Xu X, Fang CM, et al. Differences in the temperature dependence of wetland CO2 and CH4 emissions vary with water table depth [J]. Nat Clim Chang, 2021, 11(9): 766-771. |
| [4] | Saunois M, Martinez A, Poulter B, et al. Global methane budget 2000-2020[J]. Earth System Science Data, 2025, 17(5):1873-1958. |
| [5] | Wang YF, Xue K. Cold-adaptative methanogens and methanotrophs drive ecosystem feedback [J]. Sci Bull, 2023, 68(13): 1359-1360. |
| [6] | Chen H, Yao SP, Wu N, et al. Determinants influencing seasonal variations of methane emissions from Alpine wetlands in Zoige Plateau and their implications [J]. J Geophys Res, 2008, 113(D12): 2006JD008072. |
| [7] | Chen H, Wu N, Wang YF, et al. Inter-annual variations of methane emission from an open Fen on the Qinghai-Tibetan Plateau: a three-year study [J]. PLoS One, 2013, 8(1): e53878. |
| [8] | Peng HJ, Guo Q, Ding HW, et al. Multi-scale temporal variation in methane emission from an Alpine peatland on the Eastern Qinghai-Tibetan Plateau and associated environmental controls [J]. Agric For Meteor, 2019, 276/277: 107616. |
| [9] | Conrad R. The global methane cycle: recent advances in understanding the microbial processes involved [J]. Environ Microbiol Rep, 2009, 1(5): 285-292. |
| [10] | Schink B. Energetics of syntrophic cooperation in methanogenic degradation [J]. Microbiol Mol Biol Rev MMBR, 1997, 61(2): 262-280. |
| [11] | Yu GL, Chen JD, Wang GL, et al. Recent advances in constructed wetlands methane reduction: Mechanisms and methods [J]. Front Microbiol, 2023, 14: 1106332. |
| [12] | Zhao YY, Bi JF, Yi JY, et al. Pectin and homogalacturonan with small molecular mass modulate microbial community and generate high SCFAs via in vitro gut fermentation [J]. Carbohydr Polym, 2021, 269: 118326. |
| [13] | Schmidt O, Horn MA, Kolb S, et al. Temperature impacts differentially on the methanogenic food web of cellulose-supplemented peatland soil [J]. Environ Microbiol, 2015, 17(3): 720-734. |
| [14] | Hijab Z, Sikander A, Ali RN, et al. From nature to industry: Harnessing the power of pectinase towards potential biotechnological applications[J]. International Journal of Biology and Biotechnology, 2024, 21(1):17-35. |
| [15] | Conrad R, Bak F, Seitz HJ, et al. Hydrogen turnover by psychrotrophic homoacetogenic and mesophilic methanogenic bacteria in anoxic paddy soil and lake sediment [J]. FEMS Microbiol Lett, 1989, 62(5): 285-293. |
| [16] | Sieber JR, McInerney MJ, Gunsalus RP. Genomic insights into syntrophy: the paradigm for anaerobic metabolic cooperation [J]. Annu Rev Microbiol, 2012, 66: 429-452. |
| [17] | Hsieh CC, Cannella D, Jørgensen H, et al. Cellulase inhibition by high concentrations of monosaccharides [J]. J Agric Food Chem, 2014, 62(17): 3800-3805. |
| [18] | Liu X, Tan X, Tang S, et al. Comparative study on the mechanisms of Citrus pectin and polygalacturonic acid in alleviating food allergies [J]. Carbohydr Polym, 2026, 373: 124651. |
| [19] | Rotaru AE, Shrestha PM, Liu FH, et al. Direct interspecies electron transfer between Geobacter metallireducens and Methanosarcina barkeri [J]. Appl Environ Microbiol, 2014, 80(15): 4599-4605. |
| [20] | Rotaru AE, Shrestha PM, Liu FH, et al. A new model for electron flow during anaerobic digestion: direct interspecies electron transfer to Methanosaeta for the reduction of carbon dioxide to methane [J]. Energy Environ Sci, 2014, 7(1): 408-415. |
| [21] | 黄玲艳, 刘星, 周顺桂. 微生物直接种间电子传递: 机制及应用 [J]. 土壤学报, 2018, 55(6): 1313-1324. |
| Huang LY, Liu X, Zhou SG. Direct interspecies electron transfer of microbes: mechanism and application [J]. Acta Pedol Sin, 2018, 55(6): 1313-1324. | |
| [22] | 姜杰, 冯旗, 贺鹏宸, 等. 微生物种间直接电子传递机理及应用研究进展 [J]. 微生物学通报, 2023, 50(10): 4694-4704. |
| Jiang J, Feng Q, He PC, et al. Mechanism and application of direct interspecies electron transfer [J]. Microbiol China, 2023, 50(10): 4694-4704. | |
| [23] | Kraaijenbrink PDA, Stigter EE, Yao TD, et al. Climate change decisive for Asia’s snow meltwater supply [J]. Nat Clim Chang, 2021, 11(7): 591-597. |
| [24] | Ge MY, Laiho R, Korrensalo A, et al. Morphological constraints on plant-mediated methane release and oxidation under experimental warming in a peatland and meadow on the Qinghai-Tibetan Plateau [J]. Funct Ecol, 2025, 39(12): 3571-3585. |
| [25] | Yang G, Chen H, Wu N, et al. Effects of soil warming, rainfall reduction and water table level on CH4 emissions from the Zoige peatland in China [J]. Soil Biol Biochem, 2014, 78: 83-89. |
| [26] | Chen H, Wu N, Gao Y, et al. Spatial variations on methane emissions from Zoige Alpine wetlands of Southwest China [J]. Sci Total Environ, 2009, 407(3): 1097-1104. |
| [27] | Zeng MX, Zhu C, Song YG, et al. Paleoenvironment change and its impact on carbon and nitrogen accumulation in the Zoige wetland, northeastern Qinghai-Tibetan Plateau over the past 14, 000 years [J]. Geochem Geophys Geosyst, 2017, 18(4): 1775-1792. |
| [28] | Cui LJ, Kang XM, Li W, et al. Rewetting decreases carbon emissions from the zoige Alpine peatland on the Tibetan Plateau [J]. Sustainability, 2017, 9(6): 948. |
| [29] | Schwab L, Rago L, Koch C, et al. Identification of Clostridium cochlearium as an electroactive microorganism from the mouse gut microbiome [J]. Bioelectrochemistry, 2019, 130: 107334. |
| [30] | Prévoteau A, Geirnaert A, Arends JBA, et al. Hydrodynamic chronoamperometry for probing kinetics of anaerobic microbial metabolism-case study of Faecalibacterium prausnitzii [J]. Sci Rep, 2015, 5: 11484. |
| [31] | Zhang GS, Jiang N, Liu XL, et al. Methanogenesis from methanol at low temperatures by a novel psychrophilic methanogen, “Methanolobus psychrophilus” sp. nov., prevalent in zoige wetland of the Tibetan Plateau [J]. Appl Environ Microbiol, 2008, 74(19): 6114-6120. |
| [32] | Kayoumu M, Wang HL, Duan GL. Interactions between microbial extracellular polymeric substances and biochar, and their potential applications: a review [J]. Biochar, 2025, 7: 62. |
| [33] | Chen SS, Rotaru AE, Shrestha PM, et al. Promoting interspecies electron transfer with biochar [J]. Sci Rep, 2014, 4: 5019. |
| [34] | Yang Z, Sun TR, Kleindienst S, et al. A coupled function of biochar as geobattery and geoconductor leads to stimulation of microbial Fe(Ⅲ) reduction and methanogenesis in a paddy soil enrichment culture [J]. Soil Biol Biochem, 2021, 163: 108446. |
| [35] | Yu LQ, Tang J, Zhang RD, et al. Effects of biochar application on soil methane emission at different soil moisture levels [J]. Biol Fertil Soils, 2013, 49(2): 119-128. |
| [36] | Biffinger JC, Byrd JN, Dudley BL, et al. Oxygen exposure promotes fuel diversity for Shewanella oneidensis microbial fuel cells [J]. Biosens Bioelectron, 2008, 23(6): 820-826. |
| [37] | Dumas C, Basseguy R, Bergel A. Microbial electrocatalysis with Geobacter sulfurreducens biofilm on stainless steel cathodes [J]. Electrochim Acta, 2008, 53(5): 2494-2500. |
| [38] | Zhao F, Rahunen N, Varcoe JR, et al. Activated carbon cloth as anode for sulfate removal in a microbial fuel cell [J]. Environ Sci Technol, 2008, 42(13): 4971-4976. |
| [39] | Bond DR, Holmes DE, Tender LM, et al. Electrode-reducing microorganisms that harvest energy from marine sediments [J]. Science, 2002, 295(5554): 483-485. |
| [40] | Zuo Y, Xing DF, Regan JM, et al. Isolation of the exoelectrogenic Bacterium Ochrobactrum anthropi YZ-1 by using a U-tube microbial fuel cell [J]. Appl Environ Microbiol, 2008, 74(10): 3130-3137. |
| [41] | Summers ZM, Fogarty HE, Leang C, et al. Direct exchange of electrons within aggregates of an evolved syntrophic coculture of anaerobic bacteria [J]. Science, 2010, 330(6009): 1413-1415. |
| [42] | Li LY, Tian XP, Wang XM, et al. Extracellular electron transfer based methylotrophic methanogenesis in paddy soil and the prevalent Methanomassiliicoccus [J]. Commun Earth Environ, 2025, 6: 297. |
| [43] | Großkopf R, Stubner S, Liesack W. Novel euryarchaeotal lineages detected on rice roots and in the anoxic bulk soil of flooded rice microcosms [J]. Appl Environ Microbiol, 1998, 64(12): 4983-4989. |
| [44] | Mondav R, Woodcroft BJ, Kim EH, et al. Discovery of a novel methanogen prevalent in thawing permafrost [J]. Nat Commun, 2014, 5: 3212. |
| [45] | Kotsyurbenko OR, Chin KJ, Glagolev MV, et al. Acetoclastic and hydrogenotrophic methane production and methanogenic populations in an acidic West-Siberian peat bog [J]. Environ Microbiol, 2004, 6(11): 1159-1173. |
| [46] | Lehmann-Richter, Großkopf, Liesack, et al. Methanogenic Archaea and CO2-dependent methanogenesis on washed rice roots [J]. Environ Microbiol, 1999, 1(2): 159-166. |
| [47] | Lovley DR. Syntrophy goes electric: direct interspecies electron transfer [J]. Annu Rev Microbiol, 2017, 71: 643-664. |
| [48] | Yates MD, Golden JP, Roy J, et al. Thermally activated long range electron transport in living biofilms [J]. Phys Chem Chem Phys, 2015, 17(48): 32564-32570. |
| [1] | 张岩峰, 叶丽丹, 于洪巍. 氧化还原伴侣工程:P450低效问题的解决方案之一[J]. 生物技术通报, 2023, 39(4): 10-23. |
| [2] | 贺文俊, 杨铁钊, 田培, 段杜薇, 李晓辉, 何佳, 周健飞. 平顶后增温对烤烟上部叶生长发育的影响[J]. 生物技术通报, 2019, 35(3): 19-23. |
| [3] | P.N.F.Thoneley. 对固氮酶的新见解[J]. , 1994, 0(01): 8-9. |
| [4] | . 能源上的应用[J]. , 1990, 0(08): 96-98. |
| [5] | 李思经;. 一种测定植物细胞存活力的新方法[J]. , 1988, 0(11): 9-9. |
| [6] | . 能源上的应用[J]. , 1988, 0(09): 91-93. |
| [7] | . 农药[J]. , 1985, 0(02): 67-71. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||