








Biotechnology Bulletin ›› 2025, Vol. 41 ›› Issue (12): 304-312.doi: 10.13560/j.cnki.biotech.bull.1985.2025-0580
Previous Articles Next Articles
XUE Chao1( ), DUAN Ai-ling1,2, WANG Ai-yu1, YANG Yuan-xue1(
), DUAN Ai-ling1,2, WANG Ai-yu1, YANG Yuan-xue1( )
)
Received:2025-06-06
Online:2025-12-26
Published:2026-01-06
Contact:
YANG Yuan-xue
E-mail:xuechao943@163.com;yangyuanxue1989@163.com
XUE Chao, DUAN Ai-ling, WANG Ai-yu, YANG Yuan-xue. The Role of GSTs in the Detoxification Metabolism of Aphis craccivora to Afidopyropen[J]. Biotechnology Bulletin, 2025, 41(12): 304-312.
引物名称 Primer name | 引物序列 Primer sequence (5′-3′) |
|---|---|
| RT-qPCR | |
| GSTD1-F | CAGAAGAGCATCCGATTATTTCGGC |
| GSTD1-R | CGGCGAGAGTTACTTCAGGTCCAG |
| GSTD2-F | GGTTGCATTGGAAATCGACGAAG |
| GSTD2-R | TTCATTTTTGCATCTTCGGGGTTAG |
| GSTT1-F | ACTGGCCAGGCAGCAAATGTTG |
| GSTT1-R | CCACTTCACATGCAGCAACAAGATC |
| GSTT2-F | GGCGAGGTGGTTCTGTTGGATTT |
| GSTT2-R | CTTCCCGTTGTGGATGAGAACTGG |
| GSTS1-F | CCTGCTATGAATGGAAAACCAGCAA |
| GSTS1-R | GGACGATTGGCATATGGATCGAAA |
| GSTS2-F | CTCGAGCCGAACAGATCCGATTC |
| GSTS2-R | CCGACAAATGGCTACGGACTGATTA |
| GSTS3-F | CTTCAATTTTCCTGCACTGGCAGAA |
| GSTS3-R | GGAACTTTTCCAAATGGCATTGTGG |
| GSTO1-F | CGACATCACTGCTCTGGCTGAACC |
| GSTO1-R | CAATTTCCAATGTTGGAACTTTGCC |
| GSTM1-F | TCCACCACGGTGAACACATGAAAC |
| GSTM1-R | AGCGCTTGCTTCTTCGGGTCTT |
| GSTM2-F | GGTTGACCTCACCCAGGTAATGGA |
| GSTM2-R | TGCCAAATGCTACACAGTCTTCTGG |
| RPS8-F | ACTAAGCTTGGAGCTCGCCGTGTAC |
| RPS8-R | TCTGACCAACTCATTGTTTGATGCG |
| RNAi | |
| dsGSTT2-F | TAATACGACTCACTATAGGGCAGATACCAGTTCTCATCCACAA |
| dsGSTT2-R | TAATACGACTCACTATAGGGCAGATACCAGTTCTCATCCACAA |
| dsGSTT2-F | TAATACGACTCACTATAGGGAAGTATGAGTCCAAGGAAGAG |
| dsGSTT2-R | TAATACGACTCACTATAGGGAAGTATGAGTCCAAGGAAGAG |
| dsEGFP-F | TAATGTCTATAGGGAAGTTCAGCGTGTCCG |
| dsEGFP-R | TAATGTCTATAGGGCCTTGATGCCGTTC |
Table 1 Primers used in this study
引物名称 Primer name | 引物序列 Primer sequence (5′-3′) |
|---|---|
| RT-qPCR | |
| GSTD1-F | CAGAAGAGCATCCGATTATTTCGGC |
| GSTD1-R | CGGCGAGAGTTACTTCAGGTCCAG |
| GSTD2-F | GGTTGCATTGGAAATCGACGAAG |
| GSTD2-R | TTCATTTTTGCATCTTCGGGGTTAG |
| GSTT1-F | ACTGGCCAGGCAGCAAATGTTG |
| GSTT1-R | CCACTTCACATGCAGCAACAAGATC |
| GSTT2-F | GGCGAGGTGGTTCTGTTGGATTT |
| GSTT2-R | CTTCCCGTTGTGGATGAGAACTGG |
| GSTS1-F | CCTGCTATGAATGGAAAACCAGCAA |
| GSTS1-R | GGACGATTGGCATATGGATCGAAA |
| GSTS2-F | CTCGAGCCGAACAGATCCGATTC |
| GSTS2-R | CCGACAAATGGCTACGGACTGATTA |
| GSTS3-F | CTTCAATTTTCCTGCACTGGCAGAA |
| GSTS3-R | GGAACTTTTCCAAATGGCATTGTGG |
| GSTO1-F | CGACATCACTGCTCTGGCTGAACC |
| GSTO1-R | CAATTTCCAATGTTGGAACTTTGCC |
| GSTM1-F | TCCACCACGGTGAACACATGAAAC |
| GSTM1-R | AGCGCTTGCTTCTTCGGGTCTT |
| GSTM2-F | GGTTGACCTCACCCAGGTAATGGA |
| GSTM2-R | TGCCAAATGCTACACAGTCTTCTGG |
| RPS8-F | ACTAAGCTTGGAGCTCGCCGTGTAC |
| RPS8-R | TCTGACCAACTCATTGTTTGATGCG |
| RNAi | |
| dsGSTT2-F | TAATACGACTCACTATAGGGCAGATACCAGTTCTCATCCACAA |
| dsGSTT2-R | TAATACGACTCACTATAGGGCAGATACCAGTTCTCATCCACAA |
| dsGSTT2-F | TAATACGACTCACTATAGGGAAGTATGAGTCCAAGGAAGAG |
| dsGSTT2-R | TAATACGACTCACTATAGGGAAGTATGAGTCCAAGGAAGAG |
| dsEGFP-F | TAATGTCTATAGGGAAGTTCAGCGTGTCCG |
| dsEGFP-R | TAATGTCTATAGGGCCTTGATGCCGTTC |
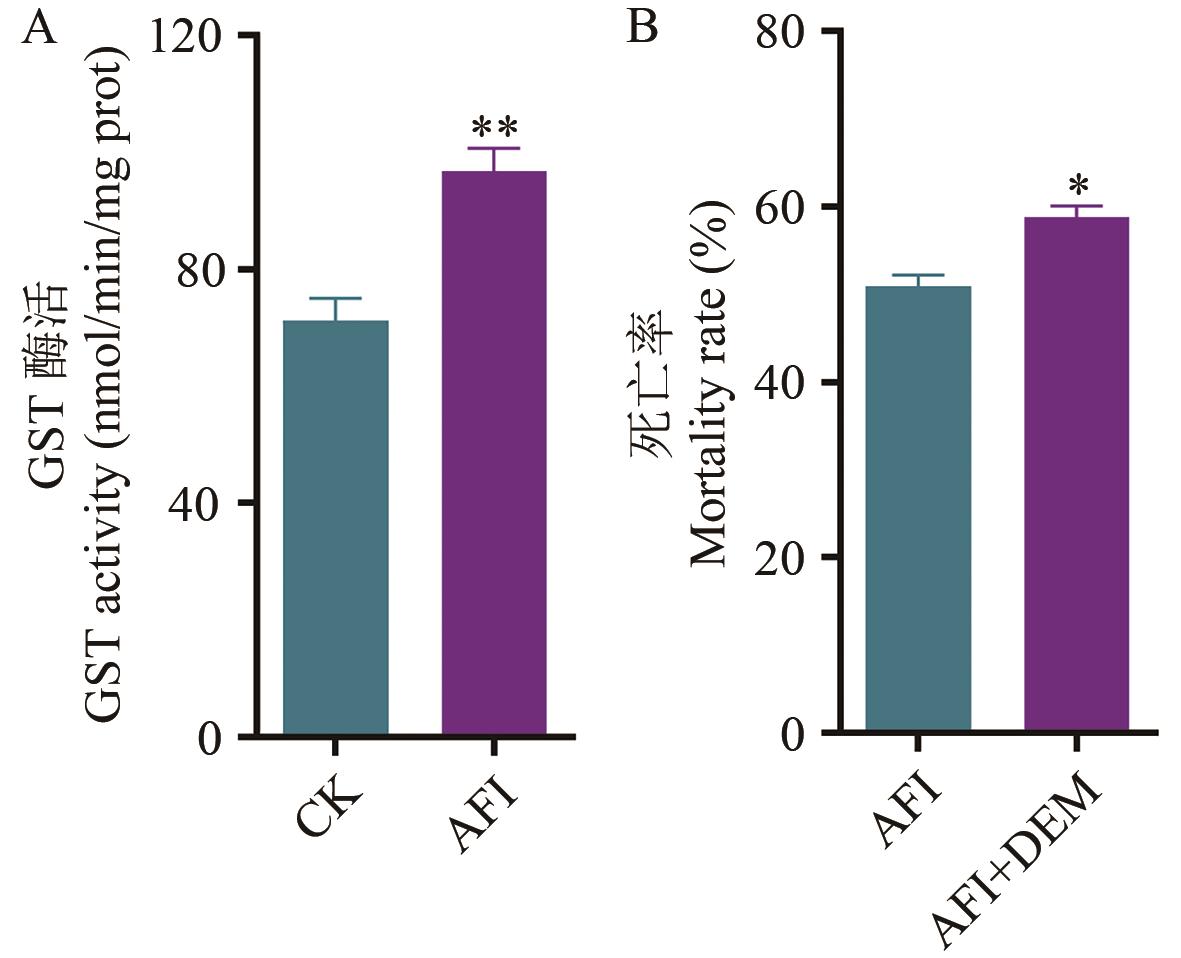
Fig. 1 Role of GST in the detoxification metabolism of A. craccivora to afidopyropenA: GST activity in A. craccivora after treatment with afidopyropen. B: Enhanced effect of DEM on afidopyropen. **P<0.01. *P<0.05. ns refers to no significant difference. The same below

Fig. 2 Expression patterns of GSTs at different tissues of A. craccivoraThe different letters indicate significant differences (P<0.05), the same below
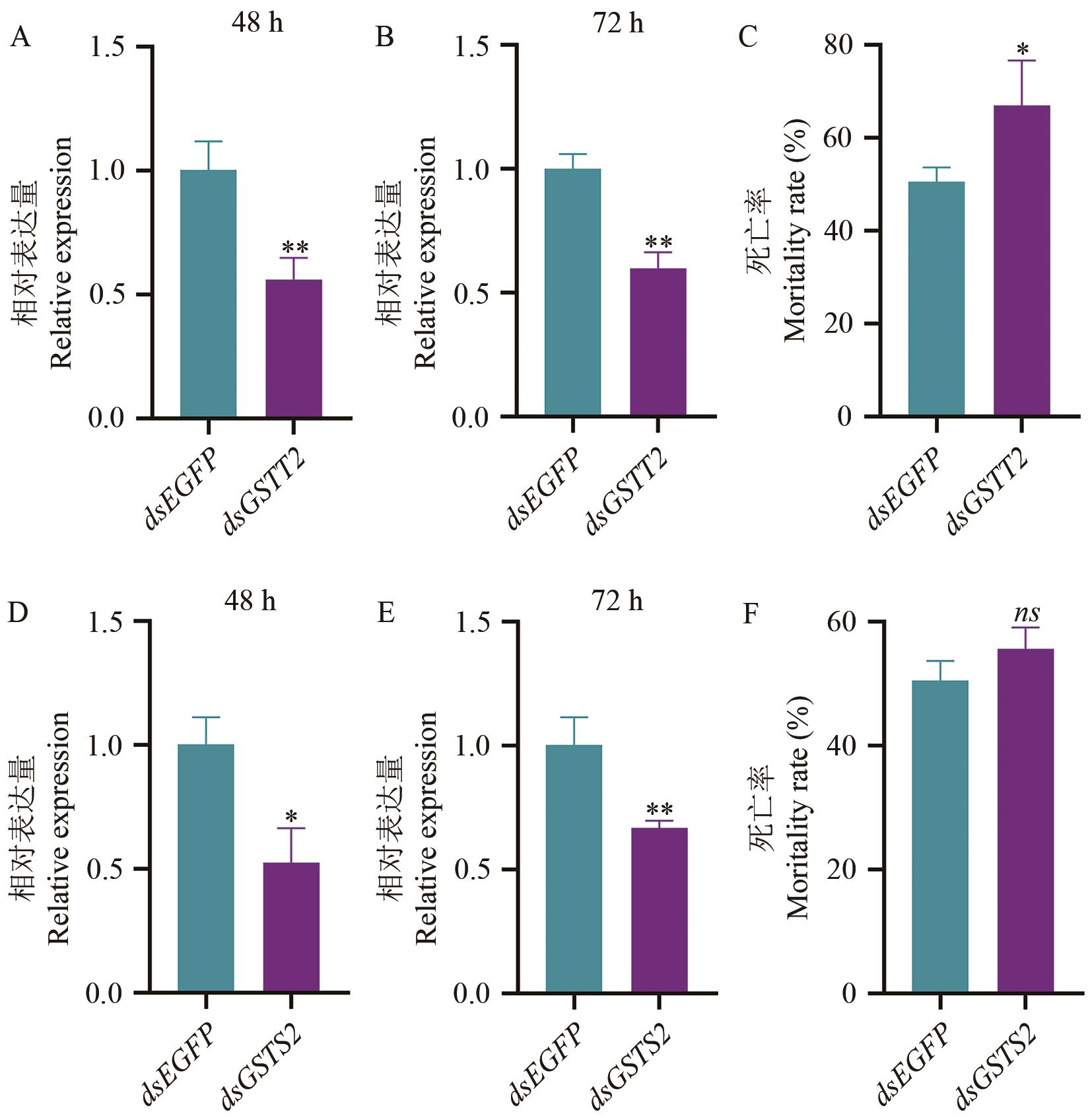
Fig. 5 Role of GSTT2 and GSTS1 in the detoxification to afidopyropen in A. craccivoraA-B: Expressions of GSTT2 at 48 and 72 h after feeding dsGSTT2. C: Changes in the sensitivity of A. craccivora after feeding dsGSTT2 to afidopyropen. D-E: Expressions of GSTS2 at 48 and 72 h after feeding dsGSTS2. F: Changes in the sensitivity of A. craccivora after feeding dsGSTT1 to afidopyropen
| [1] | Annan IB, Tingey WM, Schaefers GA, et al. Stylet penetration activities by Aphis craccivora (Homoptera: Aphididae) on plants and excised plant parts of resistant and susceptible cultivars of cowpea (Leguminosae) [J]. Ann Entomol Soc Am, 2000, 93(1): 133-140. |
| [2] | Lu ZZ, Feng LK, Gao GZ, et al. Differences in the high-temperature tolerance of Aphis craccivora (Hemiptera: Aphididae) on cotton and soybean: implications for ecological niche switching among hosts [J]. Appl Entomol Zool, 2017, 52(1): 9-18. |
| [3] | Pavithran S, Murugan M, Mannu J, et al. Identification of salivary proteins of the cowpea aphid Aphis craccivora by transcriptome and LC-MS/MS analyses [J]. Insect Biochem Mol Biol, 2024, 165: 104060. |
| [4] | Fouad EA, Abou-Yousef HM, Abdallah IS, et al. Resistance monitoring and enzyme activity in three field populations of cowpea aphid (Aphis craccivora) from Egypt [J]. Crop Prot, 2016, 81: 163-167. |
| [5] | Yang YX, Duan AL, Zhang C, et al. Overexpression of ATP-binding cassette transporters ABCG10, ABCH3 and ABCH4 in Aphis craccivora (Koch) facilitates its tolerance to imidacloprid [J]. Pestic Biochem Physiol, 2022, 186: 105170. |
| [6] | Kandasamy R, London D, Stam L, et al. Afidopyropen: New and potent modulator of insect transient receptor potential channels [J]. Insect Biochem Mol Biol, 2017, 84: 32-39. |
| [7] | Gerwick BC, Sparks TC. Natural products for pest control: an analysis of their role, value and future [J]. Pest Manag Sci, 2014, 70(8): 1169-1185. |
| [8] | Koch RL, da Silva Queiroz O, Aita RC, et al. Efficacy of afidopyropen against soybean aphid (Hemiptera: Aphididae) and toxicity to natural enemies [J]. Pest Manag Sci, 2020, 76(1): 375-383. |
| [9] | Wang R, Gao BL, Che WN, et al. First report of field resistance to afidopyropen, the novel pyropene insecticide, on Bemisia tabaci Mediterranean (Q biotype) from China [J]. Agronomy, 2022, 12(3): 724. |
| [10] | Li R, Cheng SH, Liang PZ, et al. Status of the resistance of Aphis gossypii glover, 1877 (Hemiptera: Aphididae) to afidopyropen originating from microbial secondary metabolites in China [J]. Toxins, 2022, 14(11): 750. |
| [11] | Casida JE. Neonicotinoids and other insect nicotinic receptor competitive modulators: progress and prospects [J]. Annu Rev Entomol, 2018, 63: 125-144. |
| [12] | Zhang LP, Lu H, Guo K, et al. Insecticide resistance status and detoxification enzymes of wheat aphids Sitobion avenae and Rhopalosiphum padi [J]. Sci China Life Sci, 2017, 60(8): 927-930. |
| [13] | Liu NN. Insecticide resistance in mosquitoes: impact, mechanisms, and research directions [J]. Annu Rev Entomol, 2015, 60: 537-559. |
| [14] | Ketterman AJ, Saisawang C, Wongsantichon J. Insect glutathione transferases [J]. Drug Metab Rev, 2011, 43(2): 253-265. |
| [15] | Gao HL, Lin XM, Yang BJ, et al. The roles of GSTs in fipronil resistance in Nilaparvata lugens: Over-expression and expression induction [J]. Pestic Biochem Physiol, 2021, 177: 104880. |
| [16] | Zhang BZ, Jiang YT, Cui LL, et al. Overexpression of SmUGGT1 confers imidacloprid resistance to Sitobion miscanthi (takahashi) [J]. J Agric Food Chem, 2024, 72(32): 17824-17833. |
| [17] | Liu JP, Liu Y, Wang W, et al. Characterizing three heat shock protein 70 genes of Aphis gossypii and their expression in response to temperature and insecticide stress [J]. J Agric Food Chem, 2025, 73(5): 2842-2852. |
| [18] | Yang YX, Wang AY, Fu XC, et al. microRNA-mediated detoxification network conferring afidopyropen tolerance in Aphis craccivora [J]. Pestic Biochem Physiol, 2025, 213: 106485. |
| [19] | Silva AX, Jander G, Samaniego H, et al. Insecticide resistance mechanisms in the green peach aphid Myzus persicae (Hemiptera: Aphididae) I: a transcriptomic survey [J]. PLoS One, 2012, 7(6): e36366. |
| [20] | Field LM, Bass C, Davies TGE, et al. Aphid genomics and its contribution to understanding aphids as crop pests [M]//Aphids as crop pests. UK: CABI, 2017: 37-49. |
| [21] | Ding WJ, Guo LZ, Xue YN, et al. Life parameters and physiological reactions of cotton aphids Aphis gossypii (Hemiptera: Aphididae) to sublethal concentrations of afidopyropen [J]. Agronomy, 2024, 14(2): 258. |
| [22] | Zhang JQ, Ge PT, Li DQ, et al. Two homologous carboxylesterase genes from Locusta migratoria with different tissue expression patterns and roles in insecticide detoxification [J]. J Insect Physiol, 2015, 77: 1-8. |
| [23] | Gonis E, Fraichard S, Chertemps T, et al. Expression patterns of Drosophila melanogaster glutathione transferases [J]. Insects, 2022, 13(7): 612. |
| [24] | Zhu YC, Guo ZB, Chen MS, et al. Major putative pesticide receptors, detoxification enzymes, and transcriptional profile of the midgut of the tobacco budworm, Heliothis virescens (Lepidoptera: Noctuidae) [J]. J Invertebr Pathol, 2011, 106(2): 296-307. |
| [25] | Maiwald F, Haas J, Hertlein G, et al. Expression profile of the entire detoxification gene inventory of the western honeybee, Apis mellifera across life stages [J]. Pestic Biochem Physiol, 2023, 192: 105410. |
| [26] | Xu ZB, Zou XP, Zhang N, et al. Detoxification of insecticides, allechemicals and heavy metals by glutathione S-transferase SlGSTE1 in the gut of Spodoptera litura [J]. Insect Sci, 2015, 22(4): 503-511. |
| [27] | Yang BJ, Lin XM, Yu N, et al. Contribution of glutathione S-transferases to imidacloprid resistance in Nilaparvata lugens [J]. J Agric Food Chem, 2020, 68(52): 15403-15408. |
| [28] | Li XX, Shi HY, Gao XW, et al. Characterization of UDP-glucuronosyltransferase genes and their possible roles in multi-insecticide resistance in Plutella xylostella (L.) [J]. Pest Manag Sci, 2018, 74(3): 695-704. |
| [29] | Du TH, Fu BL, Wei XG, et al. Knockdown of UGT352A5 decreases the thiamethoxam resistance in Bemisia tabaci (Hemiptera: Gennadius) [J]. Int J Biol Macromol, 2021, 186: 100-108. |
| [30] | Nair PMG, Choi J. Identification, characterization and expression profiles of Chironomus riparius glutathione S-transferase (GST) genes in response to cadmium and silver nanoparticles exposure [J]. Aquat Toxicol, 2011, 101(3-4): 550-560. |
| [31] | Yang YX, Wang AY, Zhang Y, et al. Activating pathway of three metabolic detoxification phases via down-regulated endogenous microRNAs, modulates triflumezopyrim tolerance in the small brown planthopper, Laodelphax striatellus (Fallén) [J]. Int J Biol Macromol, 2022, 222: 2439-2451. |
| [32] | Bass C, Carvalho RA, Oliphant L, et al. Overexpression of a cytochrome P450 monooxygenase, CYP6ER1, is associated with resistance to imidacloprid in the brown planthopper, Nilaparvata lugens [J]. Insect Mol Biol, 2011, 20(6): 763-773. |
| [33] | Pym A, Umina PA, Reidy-Crofts J, et al. Overexpression of UDP-glucuronosyltransferase and cytochrome P450 enzymes confers resistance to sulfoxaflor in field populations of the aphid, Myzus persicae [J]. Insect Biochem Mol Biol, 2022, 143: 103743. |
| [34] | Balakrishnan B, Su S, Wang K, et al. Identification, expression, and regulation of an omega class glutathione S-transferase in Rhopalosiphum padi (L.) (Hemiptera: Aphididae) under insecticide stress [J]. Front Physiol, 2018, 9: 427. |
| [35] | Liu L, Tang HC, Huang BJ, et al. A novel Theta-class glutathione S-transferase gene in Rhopalosiphum padi (L.) (Hemiptera: Aphididae): Identification, recombinant protein expression and function analyses of RpGSTT1 [J]. J Asia Pac Entomol, 2022, 25(2): 101915. |
| [36] | Zhang YX, Yang BJ, Yu N, et al. Insecticide resistance associated overexpression of two sigma GST genes assists Nilaparvata lugens to remedy oxidative stress from feeding on resistant rice variety [J]. Pestic Biochem Physiol, 2022, 188: 105230. |
| [37] | Fu XC, Xue C, Wang X, et al. Two detoxification enzyme genes, CYP6DA2 and CarFE4, mediate the susceptibility to afidopyropen in Semiaphis heraclei d [J]. Front Physiol, 2024, 15: 1478869. |
| [38] | Pan YO, Tian FY, Wei X, et al. Thiamethoxam resistance in Aphis gossypii glover relies on multiple UDP-glucuronosyltransferases [J]. Front Physiol, 2018, 9: 322. |
| [39] | Du TH, Xue H, Zhou XM, et al. The UDP-glycosyltransferase UGT352A3 contributes to the detoxification of thiamethoxam and imidacloprid in resistant whitefly [J]. Pestic Biochem Physiol, 2025, 208: 106321. |
| [40] | Tao F, Si FL, Hong R, et al. Glutathione S-transferase (GST) genes and their function associated with pyrethroid resistance in the malaria vector Anopheles sinensis [J]. Pest Manag Sci, 2022, 78(10): 4127-4139. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||