








生物技术通报 ›› 2021, Vol. 37 ›› Issue (4): 293-302.doi: 10.13560/j.cnki.biotech.bull.1985.2020-1015
收稿日期:2020-08-13
出版日期:2021-04-26
发布日期:2021-05-13
作者简介:钱虹萍,女,硕士研究生,研究方向:植物细胞分子生物学;E-mail:基金资助:
QIAN Hong-ping( ), CHEN Bo, LIN Jin-xing, CUI Ya-ning(
), CHEN Bo, LIN Jin-xing, CUI Ya-ning( )
)
Received:2020-08-13
Published:2021-04-26
Online:2021-05-13
摘要:
RNA聚合酶II(RNA polymerase II,RNAP II 或Pol II)在调节生物生长发育和响应环境变化中发挥着重要的作用。转录工厂是细胞核RNA聚合酶II集合的位点,多个基因被招募到转录工厂发生共转录,形成一个高度动态协调的过程。尽管真核生物转录过程中RNA聚合酶II的重要功能已经被广泛研究,但是植物RNAP II 在不同生理条件下的活体动态特征仍然知之甚少。活细胞单分子成像技术和分子标记技术的发展,将有助于提升人们对植物RNAP II 的活体动态特征的新认识。主要阐述了真核生物RNAP II 的结构和功能特点,并介绍了细胞核内RNAP II 的动态调控转录机制,重点总结了活细胞内RNAP II 的动态标记和成像技术的研究进展,以期为今后开展植物RNAP II 的动态调控研究提供理论依据。
钱虹萍, 陈博, 林金星, 崔亚宁. RNA聚合酶II动态调控及其成像技术的研究进展[J]. 生物技术通报, 2021, 37(4): 293-302.
QIAN Hong-ping, CHEN Bo, LIN Jin-xing, CUI Ya-ning. Recent Advances on Dynamic Regulation and Imaging Techniques of RNA Polymerase II[J]. Biotechnology Bulletin, 2021, 37(4): 293-302.

图1 RNAP II介导的转录过程 A:转录起始;B:转录延伸;C:转录终止
Fig. 1 RNAP II mediated transcription A:Transcription initiation. B:Transcription elongation. C:Transcription termination
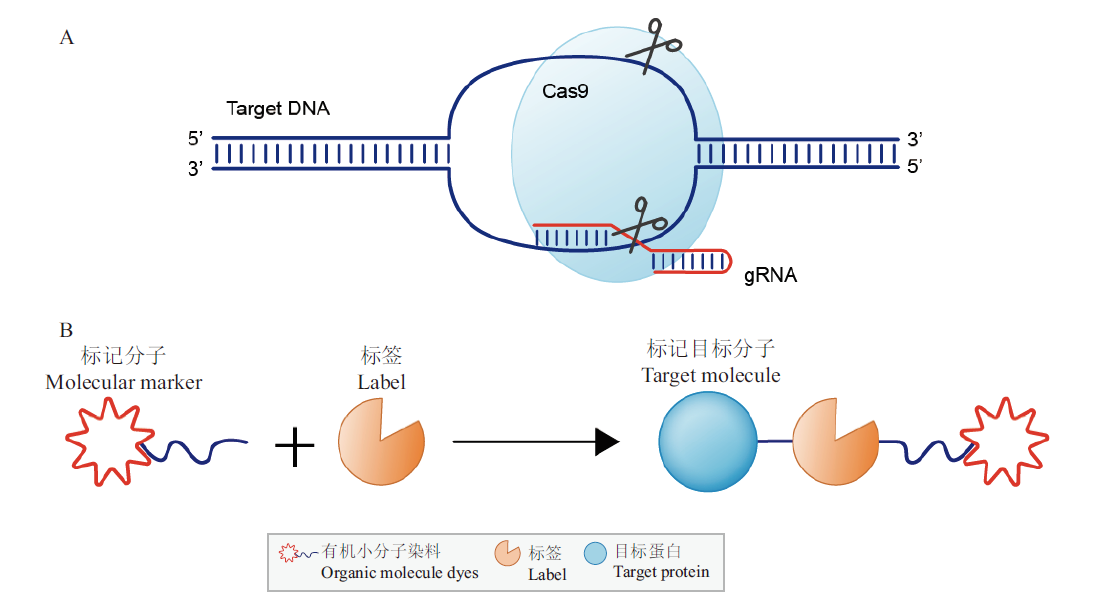
图2 CRISPR/Cas9和SLPs技术标记目标分子的示意图 A:CRISPR/Cas9技术标记目标分子的示意图;B:SLPs技术标记目标分子的示意图
Fig. 2 The schematic of CRISPR/Cas9 and SLPs A: The schematic of CRISPR/Cas9. B: The schematic of SLPs
| 参数Parameter | PALM | STORM | STED | LSFM | Bayesian nanoscopy |
|---|---|---|---|---|---|
| 分辨率Resolution/nm | <30 | <30 | 20-70 | 亚细胞分辨率 Subcellular resolution | 50 nm |
| 成像深度Imaging depth/μm | <10 | <10 | <100 | 通常<50 | 通常<50 |
| 成像速度Imaging speed | 慢Slow | 慢-低Slow to low | 中等Medium | 非常快Very fast | 非常快Very fast |
| 内源性Endogenous | 无No | 有Yes | 有Yes | 有Yes | 有Yes |
| 光毒性Phototoxicity | 中到高Medium to high | 中等Medium | 高High | 低Low | 低 Low |
| 蛇币价格Equipment price | 中等Medium | 中等Medium | 非常昂贵Very expensive | 非常昂贵Very expensive | 非常昂贵Very expensive |
表1 不同显微技术的主要优缺点
Table 1 Major merits and demerits of different microscopic technique
| 参数Parameter | PALM | STORM | STED | LSFM | Bayesian nanoscopy |
|---|---|---|---|---|---|
| 分辨率Resolution/nm | <30 | <30 | 20-70 | 亚细胞分辨率 Subcellular resolution | 50 nm |
| 成像深度Imaging depth/μm | <10 | <10 | <100 | 通常<50 | 通常<50 |
| 成像速度Imaging speed | 慢Slow | 慢-低Slow to low | 中等Medium | 非常快Very fast | 非常快Very fast |
| 内源性Endogenous | 无No | 有Yes | 有Yes | 有Yes | 有Yes |
| 光毒性Phototoxicity | 中到高Medium to high | 中等Medium | 高High | 低Low | 低 Low |
| 蛇币价格Equipment price | 中等Medium | 中等Medium | 非常昂贵Very expensive | 非常昂贵Very expensive | 非常昂贵Very expensive |
| [1] | 于福先, 朱志伟, 陈晓宇, 等. 真核生物RNA聚合酶Ⅱ核心启动子[J]. 生命的化学, 2011,31(6):822-826. |
| Yu FX, Zhu ZW, Chen XY, et al. Eukaryotic RNA polymerase Ⅱ core promoter[J]. Chemistry of Life, 2011,31(6):822-826. | |
| [2] |
Zhou M, Law JA. RNA Pol IV and V in gene silencing:rebel polymerases evolving away from Pol II’s rules[J]. Current Opinion in Plant Biology, 2015,27:154-164.
doi: 10.1016/j.pbi.2015.07.005 URL |
| [3] |
Xi MS, Anthony B, Miles P, et al. Size-Dependent increase in RNA Polymerase II initiation rates mediates gene expression scaling with cell size[J]. Current Biology, 2020,30(7):1217-1230.
doi: 10.1016/j.cub.2020.01.053 URL |
| [4] | Louder RK, He Y, Lopez-Blanco JR, et al. Corrigendum:structure of promoter-bound TFIID and model of human pre-initiation complex assembly[J]. Nature, 2016,536(7614):112. |
| [5] |
Zhang Z, Tjian R. Measuring dynamics of eukaryotic transcription initiation:challenges, insights and opportunities[J]. Transcription, 2018,9(3):159-165.
doi: 10.1080/21541264.2017.1363017 URL |
| [6] |
Cho WK, Spille JH, Hech TM, et al. Mediator and RNA polymerase II clusters associate in transcription-dependent condensates[J]. Science, 2018,361(64):412-415.
doi: 10.1126/science.aar4199 URL |
| [7] |
Chen X, Wei M, Zheng M, et al. Study of RNA polymerase II clustering inside live-cell nuclei using bayesian nanoscopy[J]. ACS Nano, 2016,10(2):2447-54.
doi: 10.1021/acsnano.5b07257 URL |
| [8] | 杨宁, 肖桂林. 真核生物RNA聚合酶Ⅱ的研究进展[J]. 临床与病理杂志, 2005,25(4):333-335. |
| Yang N, Xiao GL. Research progress of eukaryotic RNA polymerase Ⅱ[J]. Journal of Clinical and Pathological Research, 2005,25(4):333-335. | |
| [9] | 陈亮. 拟南芥RNA聚合酶Ⅱ第三大亚基NRPB3调控气孔图式发育和分化[D]. 兰州:兰州大学, 2016. |
| Chen L. NRPB3, the third largest subunit of RNA polymerase II, is essential for stomatal patterning and differentiation in Arabidopsis[D]. Lanzhou:Lanzhou University, 2016. | |
| [10] | 李慧. HECT家族泛素连接酶Wwp2泛素化修饰RNA聚合酶II大亚基Rpb1[D]. 北京:中国科学院大学, 2006. |
| Li H. HECT domain ubiquitin-protein ligase Wwp2 ubiquitinates the large subunit of RNA polymerase II[D]. Beijing:University of Chinese Academy of Sciences, 2006. | |
| [11] |
Pavel B, Magdalena K, Aiste K, et al. Yeast Spt6 reads multiple phosphorylation patterns of RNA polymerase II C-terminal domain in vitro[J]. Journal of Molecular Biology, 2020,432(14):4092-4107.
doi: 10.1016/j.jmb.2020.05.007 URL |
| [12] |
Vasiljeva L, Kim M, Mutschler H, et al. The Nrd1-Nab3-Sen1 termination complex interacts with the Ser5-phosphorylated RNA polymerase II C-terminal domain[J]. Nature Structural & Molecular Biology, 2008,15(8):795-804.
doi: 10.1038/nsmb.1468 URL |
| [13] | 韩玉波, 张飞雄. RNA聚合酶II中的CTD结构在mRNA转录和加工偶联过程中的重要作用[J]. 遗传, 2003(1):102-106. |
| Han YB, Zhang FX. The Role of the carboxy-terminal domain of the RNA polymerase II on coupling mRNA transcription with processing[J]. Hereditas, 2003(1):102-106. | |
| [14] | Li B, Meng X, Shan L, et al. Transcriptional regulation of pattern-triggered immunity in plants[J]. Cell Host & Microbe, 2016,19(5):641-650. |
| [15] | 张倩倩. RNA聚合酶Ⅱ最大亚基RPB1的全长CTD通过控制细胞周期影响根尖干细胞池的命运[D]. 泰安:山东农业大学, 2017. |
| Zhang QQ. Intact CTD of RNA polymerse II maintains the root stem cell niches mediated by cell cycling control in arabidopsis[D]. Tai’an:Shandong Agricultural University, 2017. | |
| [16] |
Liu Z, Merkurjev D, Yang F, et al. Enhancer activation requires trans-recruitment of a mega transcription factor complex[J]. Cell, 2014,159(2):358-373.
doi: 10.1016/j.cell.2014.08.027 URL |
| [17] | 李宛莹. 真核生物转录因子结合位点聚集区间的演化规律研究[D]. 北京:军事科学院, 2019. |
| Li WY. The evolution of transcription factors binding sites clustered regions in eukaryote[D]. Beijing:Academy of Military Sciences PLA China, 2019. | |
| [18] |
Natalia P, Yi J, Koon HW, et al. Mediator undergoes a compositional change during transcriptional activation[J]. Mol Cell, 2016,64(3):443-454.
doi: 10.1016/j.molcel.2016.09.015 URL |
| [19] | 窦悦, 刘美彤, 卢安娜, 等. 中介体亚基MED25调控植物激素信号转导的研究进展[J]. 生物技术通报, 2018,34(7):40-47. |
| Dou Y, Liu MT, Lu AN, et al. Regulatory mechanism of mediator subunit MED25 on multi-phytohormone signaling pathways[J]. Biotechnology Bulletin, 2018,34(7):40-47. | |
| [20] | Peil K, Jurgens H, Luige J, et al. Taf14 is required for the stabilization of transcription pre-initiation complex in saccharomyces cerevisiae[J]. Epigenetics & Chromatin, 2020,13(1):24. |
| [21] | 梅坤荣. CREPT和p15RS结合RNA聚合酶II的结构生物学研究[D]. 北京:清华大学, 2014. |
| Mei KR. Structural study of the interaction between CREPT/p15RS and RNA polymerase II[D]. Beijing:Tsinghua University, 2014. | |
| [22] |
Paakinaho V, Presman DM, Ball DA, et al. Single-molecule analysis of steroid receptor and cofactor action in living cells[J]. Nature Communications, 2017,8(8):15896.
doi: 10.1038/ncomms15896 URL |
| [23] |
Harlen KM, Churchman LS. The code and beyond:transcription regulation by the RNA polymerase II carboxy-terminal domain[J]. Nature Reviews Molecular Cell Biology, 2017,18(4):263-273.
doi: 10.1038/nrm.2017.10 URL |
| [24] |
Hajheidari M, Farrona S, Huettel B, et al. CDKF;1 and CDKD protein kinases regulate phosphorylation of serine residues in the C-terminal domain of Arabidopsis RNA polymerase II[J]. The Plant Cell, 2012,24(4):1626-1642.
doi: 10.1105/tpc.112.096834 pmid: 22547781 |
| [25] |
Ly E, Powell AE, Goodrich JA, et al. Release of human TFIIB from actively transcribing complexes is triggered upon synjournal of 7- and 9-nt RNAs[J]. Journal of Molecular Biology, 2020,432(14):4049-4060.
doi: 10.1016/j.jmb.2020.05.005 URL |
| [26] |
Alan G, Keiichi I, Chi SC, et al. Gene-specific control of tRNA expression by RNA polymerase II[J]. Molecular Cell, 2020,78(4):765-778.
doi: 10.1016/j.molcel.2020.03.023 URL |
| [27] |
Peterlin B. Controlling the elongation phase of transcription with P-TEFb[J]. Molecular Cell, 2006,23(3):297-305.
doi: 10.1016/j.molcel.2006.06.014 URL |
| [28] |
Marion G, Klaus DG. The plant RNA polymerase II elongation complex:A hub coordinating transcript elongation and mRNA processing[J]. Transcription, 2018,9(2):117-122.
doi: 10.1080/21541264.2017.1356902 URL |
| [29] | 王娟. 转录因子ⅡA及其识别元件在RNA聚合酶II指导的基因转录中的作用和调节机制的研究[D]. 武汉:武汉科技大学, 2019. |
| Wang J. The roles of transcription factor IIA and its recognition element in gene transcription directed by RNA polymerase II and their regulatory mechanisms[D]. Wuhan:Wuhan University of Science and Technology, 2019. | |
| [30] |
Eaton JD, Weat S. Termination of transcription by RNA polymerase II:BOOM![J]. Trends in Genetics, 2020,36(9):664-675.
doi: 10.1016/j.tig.2020.05.008 URL |
| [31] | 夏珺, 李建军, 王梁华. 转录终止及其调控[J]. 生命的化学, 2012,32(6):550-555. |
| Xia J, Li JJ, Wang LH. Transcriptional termination and its regulation[J]. Chemistry of Life, 2012,32(6):550-555. | |
| [32] |
Sanchez AM, Garg A, Shuman S, et al. Genetic interactions and transcriptomics implicate fission yeast CTD prolyl isomerase pin1 as an agent of RNA 3' processing and transcription termination that functions via its effects on CTD phosphatase Ssu72[J]. Nucleic Acids Research, 2020,48(9):4811-4826.
doi: 10.1093/nar/gkaa212 pmid: 32282918 |
| [33] | 唐文. 细胞核肌动蛋白参与基因转录调控的分子机制研究[D]. 长春:东北师范大学, 2009. |
| Tang W. Studies on the molecular mechanism of nuclear actin in gene transcription regulation[D]. Changchun:Northeast Normal University, 2009. | |
| [34] |
Cisse II, Izeddin I, Causse SZ, et al. Real-time dynamics of RNA polymerase II clustering in live human cells[J]. Science, 2013,341(6146):664-667.
doi: 10.1126/science.1239053 URL |
| [35] | 张茜. 可逆光开关荧光蛋白的开发及其在超高分辨率显微成像中的应用[D]. 武汉:华中师范大学, 2016. |
| Zhang Q. Development of reversibly switchable fluorescent proteins for super-resolution imaging[D]. Wuhan:Central China Normal University, 2016. | |
| [36] | 邢晶晶, 林金星. 活细胞单分子荧光标记——点亮生命微观世界的繁星[J]. 生命世界, 2015(12):48-53. |
| Xing JJ, Lin JX. Single molecule fluorescent labeling of living cells-stars that light up the microscopic world of life[J]. Life World, 2015(12):48-53. | |
| [37] | Prather RS. Green fluorescent protein(GFP)transgenic pig produced by somatic cell nuclear transfer[J]. Chinese Science Bulletin, 2008(7):1035-1039. |
| [38] | 张静. 荧光标记技术的研究进展分析[J]. 科技视界, 2018,247(25):90-91. |
| Zhang J. Research progress of fluorescent labeling technology[J]. Science & Technology Vision, 2018,247(25):90-91. | |
| [39] | 王飞, 杨海涛, 王泽方. 红色荧光蛋白的研究进展[J]. 生物技术通报, 2017,33(9):32-47. |
| Wang F, Yang HT, Wang ZF. Research progress on red fluorescent protein[J]. Biotechnology Bulletin, 2017,33(9):32-47. | |
| [40] |
Reelina B, Lan TL, Zhen YM, et al. Using amino-labeled nucleotide probes for simultaneous single molecule RNA-DNA fISH[J]. PLoS One, 2014,9(9):e107425.
doi: 10.1371/journal.pone.0107425 URL |
| [41] | 李和刚, 杨峰, 张宝珣, 等. CRISPR/Cas9技术研究进展[J]. 中国畜牧杂志, 2015,51(21):92-97. |
| Li HG, Yang F, Zhang BX, et al. Progress of CRISPR/Cas9 system[J]. Chinese Journal of Animal Science, 2015,51(21):92-97. | |
| [42] |
Cho WK, Jayanth N, Mullen S, et al. Super-resolution imaging of fluorescently labeled, endogenous RNA polymerase II in living cells with CRISPR/Cas9-mediated gene editing[J]. Scientific Reports, 2016,6(6):35949.
doi: 10.1038/srep35949 URL |
| [43] |
Ma XL, Zhang QY, Zhu QL, et al. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants[J]. Molecular Plant, 2015,8(8):1274-1284.
doi: 10.1016/j.molp.2015.04.007 URL |
| [44] | 张晨, 雷展, 李凯, 等. CRISPR/Cas9系统中的脱靶效应及检测技术研究进展[J]. 生物技术通报, 2020,36(3):78-87. |
| Zhang C, Lei Z, Li K, et al. Research progress on off-target effects and detection techniques in CRISPR/Cas9 systems[J]. Biotechnology Bulletin, 2020,36(3):78-87. | |
| [45] |
Liu Y, Miao K, Dunham NP, et al. The cation-π interaction enables a Halo-Tag fluorogenic probe for fast no-wash live cell imaging and gel-free protein quantification[J]. Biochemistry, 2017,56(11):1585-1595.
doi: 10.1021/acs.biochem.7b00056 pmid: 28221782 |
| [46] |
Roman SE, Stephanie WB, Jennifer HR, et al. Labeling strategies matter for super-resolution microscopy:a comparison between Halo-Tags and SNAP-tags[J]. Cell Chemical Biology, 2019,26(4):584-592.
doi: 10.1016/j.chembiol.2019.01.003 URL |
| [47] | Los GV, Wood K. The HaloTag:a novel technology for cell imaging and protein analysis[J]. Methods in Molecular Biology, 2007,356(356):195-208. |
| [48] | Yazaki J, Galli M, Kim AY, et al. Mapping transcription factor interactome networks using HaloTag protein arrays[J]. Proceedings of the National Academy of Sciences of the United States of America, 2016,113(29):E4238. |
| [49] | 夏鹏, 窦震, 姚雪彪. 超高分辨率显微技术研究进展[J]. 生命的化学, 2015,35(3):430-437. |
| Xia P, Dou Z, Yao XB. Progress of super-resolution microscopy[J]. Chemistry of Life, 2015,35(3):430-437. | |
| [50] | 胡春光, 查日东, 凌秋雨, 等. 超分辨显微技术在活细胞中的应用与发展[J]. 红外与激光工程, 2017,46(11):25-35. |
| Hu CG, Zha RD, Ling QY, et al. Super-resolution microscopy applications and development in living cell[J]. Infrared and Laser Engineering, 2017,46(11):25-35. | |
| [51] |
Scheermelleh , Ferrand A, Huser T, et al. Super-resolution microscopy demystified[J]. Nature Cell Biology, 2019,21(1):72-84.
doi: 10.1038/s41556-018-0251-8 URL |
| [52] | 马迪, 夏仁品, 冯志坚, 等. 随机光学重构显微镜在外泌体观察中的应用[J]. 分子影像学杂志, 2018,41(2):224-228. |
| Ma D, Xia RP, Feng ZJ, et al. Research progress on red fluorescent protein[J]. Journal of Molecular Imaging, 2018,41(2):224-228. | |
| [53] | 苏乾, 刘振, 薛博鑫, 等. 超高分辨率荧光显微成像:2014年诺贝尔化学奖解析[J]. 科学通报, 2014,59(34):3342-3343. |
| Su Q, Liu Z, Xue BX, et al. Super-resolution fluorescence microscopy:analysis of the 2014 Nobel Prize in Chemistry[J]. Chinese Science Bulletin, 2014,59(34):3342-3343. | |
| [54] |
Wildanger D, Medda R, Kastrup L, et al. A compact STED microscope providing 3D nanoscale resolution[J]. Journal of Microscopy, 2009,236(1):35-43.
doi: 10.1111/jmi.2009.236.issue-1 URL |
| [55] |
Li J, Dong A, Saydaminova K, et al. Single-molecule nanoscopy elucidates RNA polymerase II transcription at single genes in live cells[J]. Cell, 2019,178(2):491-506.
doi: 10.1016/j.cell.2019.05.029 URL |
| [56] |
Buglak NE, Jennifer L, Pablo A, et al. Light sheet fluorescence microscopy as a new method for unbiased three-dimensional analysis of vascular injury[J]. Cardiovascular Research, 2021,117(2):520-532.
doi: 10.1093/cvr/cvaa037 URL |
| [57] | 谢新林, 陈蓉, 赵宇轩, 等. 结合光片照明与超分辨的三维荧光显微成像[J]. 中国激光, 2018,45(3):56-63. |
| Xie XL, Chen R, Zhao YX, et al. Combination light-sheet illumination with super-resolution three-dimensional fluorescence microimaging[J]. Chinese Journal of Lasers, 2018,45(3):56-63. | |
| [58] | Zhao ZW, Roy R, Gebhard TC, et al. Spatial organization of RNA polymerase II inside a mammalian cell nucleus revealed by reflected light-sheet superresolution microscopy[J]. Proceedings of the National Academy of Sciences of the United States of America, 2014,111(2):681-686. |
| [59] | 于湘华, 刘超, 柏晨, 等. 光片荧光显微成像技术及应用进展[J]. 激光与光电子学进展, 2020,57(10):100001. |
| Yu XH, Liu C, Bo C, et al. Progress in light-sheet fluorescence microscopy and applications[J]. Laser & Optoelectronics Progress, 2020,57(10):100001. | |
| [60] |
Cox S, Rosten E, Monypenny J, et al. Bayesian localization microscopy reveals nanoscale podosome dynamics[J]. Nature Methods, 2012,9(2):195.
doi: 10.1038/nmeth.1812 URL |
| [61] |
Hu YS, Zhu Q, Elkins K, et al. Light-sheet bayesian microscopy enables deep-cell super-resolution imaging of heterochromatin in live human embryonic stem cells[J]. Optical Nanoscopy, 2013,2(1):1-12.
doi: 10.1186/2192-2853-2-1 URL |
| [1] | 位欣欣, 兰海燕. 植物MYB转录因子调控次生代谢及逆境响应的研究进展[J]. 生物技术通报, 2022, 38(8): 12-23. |
| [2] | 张婵, 吴友根, 于靖, 杨东梅, 姚广龙, 杨华庚, 张军锋, 陈萍. 光与茉莉酸信号介导的萜类化合物合成分子机制[J]. 生物技术通报, 2022, 38(8): 32-40. |
| [3] | 孙曼銮, 葛赛, 卜佳, 朱壮彦. 大肠杆菌核糖核酸酶调控机制研究[J]. 生物技术通报, 2022, 38(3): 234-245. |
| [4] | 张凤, 陈伟. 代谢组学在植物逆境生物学中的研究进展[J]. 生物技术通报, 2021, 37(8): 1-11. |
| [5] | 张婵, 姚广龙, 张军锋, 于靖, 杨东梅, 陈萍, 吴友根. 广藿香百秋李醇分子调控及合成生物学研究进展[J]. 生物技术通报, 2021, 37(8): 55-64. |
| [6] | 邹坤, 路丽丽, Collins Asiamah Amponsah, 薛缘, 张少伟, 苏瑛, 赵志辉. 家禽卵泡闭锁机制的研究进展[J]. 生物技术通报, 2020, 36(4): 185-191. |
| [7] | 李泽卿, 刘彩贤, 邢文, 文亚峰. miRNA在植物响应高温胁迫中的研究进展[J]. 生物技术通报, 2020, 36(2): 149-157. |
| [8] | 郑文清, 张倩, 杜亮. 短串联靶标模拟技术及其在植物miRNA功能研究中的应用[J]. 生物技术通报, 2020, 36(12): 256-264. |
| [9] | 位明明, 曾霞, 安泽伟, 胡彦师, 黄肖, 李维国. C类花器官特征基因AGAMOUS(AG)调控植物花分生组织维持与终止研究进展[J]. 生物技术通报, 2020, 36(1): 135-143. |
| [10] | 常永芳, 包鹏甲, 褚敏, 吴晓云, 梁春年, 阎萍. LncRNA在哺乳动物毛囊发育调控中的研究进展[J]. 生物技术通报, 2019, 35(8): 205-212. |
| [11] | 莫显兰, 史列琴, 陆秋利, 王小敏, 任振新. Sl-miR482在番茄果实中的表达分析及STTM沉默载体的构建[J]. 生物技术通报, 2019, 35(12): 50-56. |
| [12] | 黄幸, 丁峰, 彭宏祥, 潘介春, 何新华, 徐炯志, 李琳. 植物WRKY转录因子家族研究进展[J]. 生物技术通报, 2019, 35(12): 129-143. |
| [13] | 旷永洁, 柳浪, 严芳, 任斌, 闫大琦, 张大伟, 林宏辉, 周焕斌. 水稻与病原物互作中植物激素功能的研究进展[J]. 生物技术通报, 2018, 34(2): 74-86. |
| [14] | 谭玉荣, 王丹, 高璇, 刘进平. 植物长链非编码RNA研究进展[J]. 生物技术通报, 2018, 34(10): 1-10. |
| [15] | 刘晓威, 杨秀艳, 刘正祥, 武海雯, 张华新, 朱建峰. MicroRNA在植物抵御盐胁迫过程中的作用[J]. 生物技术通报, 2017, 33(12): 12-21. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||