








生物技术通报 ›› 2025, Vol. 41 ›› Issue (1): 62-73.doi: 10.13560/j.cnki.biotech.bull.1985.2024-0554
收稿日期:2024-06-11
出版日期:2025-01-26
发布日期:2025-01-22
通讯作者:
许文涛,男,博士,教授,研究方向:功能食品、功能核酸与生物安全;E-mail: xuwentao@cau.edu.cn作者简介:高欣茹,女,研究方向:纳米材料与药物递送; E-mail: gao1587697334@qq.com
基金资助:
GAO Xin-ru1( ), XU Wen-tao1,2(
), XU Wen-tao1,2( )
)
Received:2024-06-11
Published:2025-01-26
Online:2025-01-22
摘要:
萝卜硫素是一种植物化学物质,具备抗氧化、抗癌、抗炎等多种生物活性。然而,萝卜硫素稳定差、热敏性强等因素限制了它在不同领域的应用。基于纳米技术的载药系统为萝卜硫素的多维应用提供了可能。通过与纳米技术结合,可以提高萝卜硫素的生物利用率,促进其临床试验的发展。基于以上特点,本文围绕萝卜硫素不同生物活性的具体作用机制、萝卜硫素纳米颗粒的特点及其生物学应用展开详细的介绍。其中,重点综述了应用于萝卜硫素递送的无机纳米颗粒、有机纳米颗粒及无机-有机复合纳米颗粒的特点,并根据萝卜硫素纳米颗粒的治疗多样性对其具体应用进行归纳总结。此外,对当前萝卜硫素纳米颗粒面临的主要问题与发展前景作出展望,以期扩展萝卜硫素的应用领域,为未来食品、保健、医疗科技的发展注入新的动力。
高欣茹, 许文涛. 萝卜硫素纳米颗粒及其生物学应用[J]. 生物技术通报, 2025, 41(1): 62-73.
GAO Xin-ru, XU Wen-tao. Sulforaphane Nanoparticles and Biological Applications[J]. Biotechnology Bulletin, 2025, 41(1): 62-73.
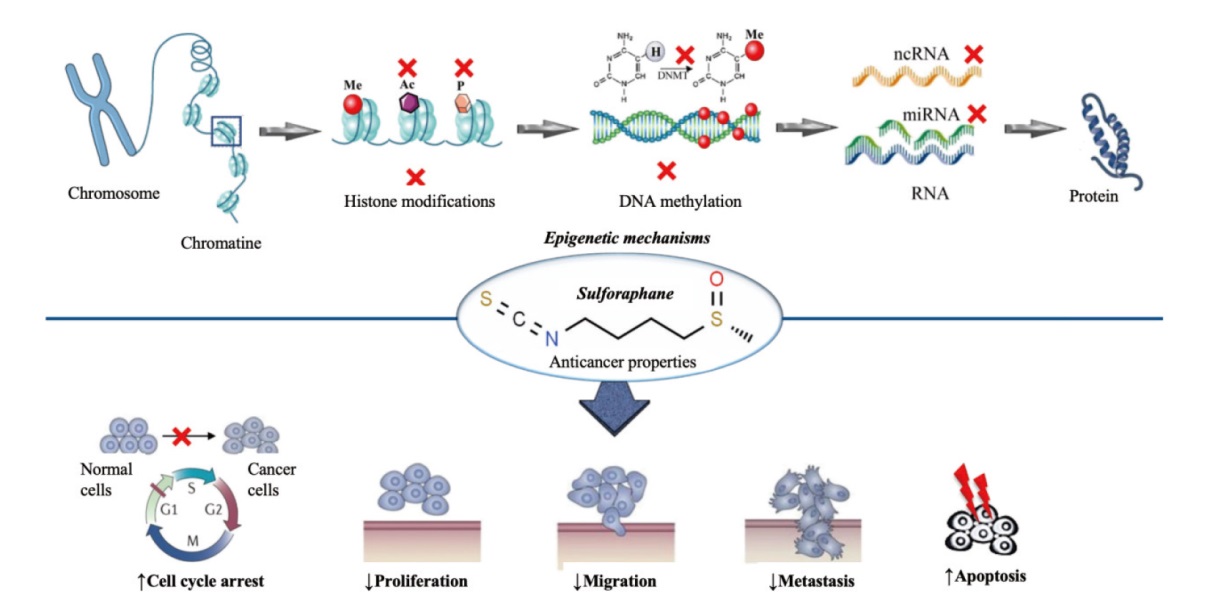
图2 关于萝卜硫素在癌症中的表观遗传修饰的汇总方案 X:抑制;Ac:乙酰化;P:磷酸化;Me:甲基化;DNMT:DNA 甲基转移酶
Fig. 2 Summarized scheme regarding epigenetic modifications of sulforaphane in cancer X: Iinhibition; Ac: acetylation; P: phosphorylation; Me: methylation; DNMT: DNA methyltransferase
| [1] | Sikdar S, Papadopoulou M, Dubois J. What do we know about sulforaphane protection against photoaging?[J]. J Cosmet Dermatol, 2016, 15(1): 72-77. |
| [2] | Greaney AJ, Maier NK, Leppla SH, et al. Sulforaphane inhibits multiple inflammasomes through an Nrf2-independent mechanism[J]. J Leukoc Biol, 2016, 99(1): 189-199. |
| [3] |
Johansson NL, Pavia CS, Chiao JW. Growth inhibition of a spectrum of bacterial and fungal pathogens by sulforaphane, an isothiocyanate product found in broccoli and other cruciferous vegetables[J]. Planta Med, 2008, 74(7): 747-750.
doi: 10.1055/s-2008-1074520 pmid: 18484523 |
| [4] | Kaiser AE, Baniasadi M, Giansiracusa D, et al. Sulforaphane: a broccoli bioactive phytocompound with cancer preventive potential[J]. Cancers, 2021, 13(19): 4796. |
| [5] |
Giacoppo S, Galuppo M, Montaut S, et al. An overview on neuroprotective effects of isothiocyanates for the treatment of neurodegenerative diseases[J]. Fitoterapia, 2015, 106: 12-21.
doi: 10.1016/j.fitote.2015.08.001 pmid: 26254971 |
| [6] | Wu YF, Mao JW, Mei LH, et al. Kinetic studies of the thermal degradation of sulforaphane and its hydroxypropyl-β-cyclodextrin inclusion complex[J]. Food Res Int, 2013, 53(1): 529-533. |
| [7] | Oravczová V, Garaiová Z, Hianik T. Nanoparticles and nanomotors modified by nucleic acids aptamers for targeted drug delivery[J]. Russ J Bioorg Chem, 2021, 47(2): 344-366. |
| [8] |
Krissanaprasit A, Key CM, Pontula S, et al. Self-assembling nucleic acid nanostructures functionalized with aptamers[J]. Chem Rev, 2021, 121(22): 13797-13868.
doi: 10.1021/acs.chemrev.0c01332 pmid: 34157230 |
| [9] | Marques AC, Costa PJ, Velho S, et al. Functionalizing nanoparticles with cancer-targeting antibodies: a comparison of strategies[J]. J Control Release, 2020, 320: 180-200. |
| [10] | Chen L, Zhang WL, Xie DQ, et al. Sulforaphane alleviates hepatic ischemia-reperfusion injury through promoting the activation of Nrf-2/HO-1 signaling[J]. Transpl Immunol, 2021, 68: 101439. |
| [11] |
Peng N, Jin LP, He AZ, et al. Effect of sulphoraphane on newborn mouse cardiomyocytes undergoing ischaemia/reperfusion injury[J]. Pharm Biol, 2019, 57(1): 753-759.
doi: 10.1080/13880209.2019.1680705 pmid: 31686558 |
| [12] | Wu DM, Zheng ZH, Fan SH, et al. Sulforaphane administration alleviates diffuse axonal injury(DAI)via regulation signaling pathway of NRF2 and HO-1[J]. J Cell Biochem, 2020, 121(1): 430-442. |
| [13] | Yang SH, Li P, Yu LH, et al. Sulforaphane protect against cadmium-induced oxidative damage in mouse leydigs cells by activating Nrf2/ARE signaling pathway[J]. Int J Mol Sci, 2019, 20(3): 630. |
| [14] |
Yanaka A. Role of NRF2 in protection of the gastrointestinal tract against oxidative stress[J]. J Clin Biochem Nutr, 2018, 63(1): 18-25.
doi: 10.3164/jcbn.17-139 pmid: 30087539 |
| [15] |
Guerrero-Beltrán CE, Calderón-Oliver M, Martínez-Abundis E, et al. Protective effect of sulforaphane against cisplatin-induced mitochondrial alterations and impairment in the activity of NAD(P)H: quinone oxidoreductase 1 and γ glutamyl cysteine ligase: studies in mitochondria isolated from rat kidney and in LLC-PK1 cells[J]. Toxicol Lett, 2010, 199(1): 80-92.
doi: 10.1016/j.toxlet.2010.08.009 pmid: 20732396 |
| [16] |
Kubo ER, Chhunchha B, Singh P, et al. Sulforaphane reactivates cellular antioxidant defense by inducing Nrf2/ARE/Prdx6 activity during aging and oxidative stress[J]. Sci Rep, 2017, 7(1): 14130.
doi: 10.1038/s41598-017-14520-8 pmid: 29074861 |
| [17] | Patel B, Mann GE, Chapple SJ. Concerted redox modulation by sulforaphane alleviates diabetes and cardiometabolic syndrome[J]. Free Radic Biol Med, 2018, 122: 150-160. |
| [18] | Ahmed SMU, Luo L, Namani A, et al. Nrf2 signaling pathway: Pivotal roles in inflammation[J]. Biochim Biophys Acta Mol Basis Dis, 2017, 1863(2): 585-597. |
| [19] |
Heiss E, Herhaus C, Klimo K, et al. Nuclear factor κB is a molecular target for sulforaphane-mediated anti-inflammatory mechanisms[J]. J Biol Chem, 2001, 276(34): 32008-32015.
doi: 10.1074/jbc.M104794200 pmid: 11410599 |
| [20] | Liu YC, Hsieh CW, Weng YC, et al. Sulforaphane inhibition of monocyte adhesion via the suppression of ICAM-1 and NF-kappaB is dependent upon glutathione depletion in endothelial cells[J]. Vascul Pharmacol, 2008, 48(1): 54-61. |
| [21] |
Xu CJ, Shen GX, Chen C, et al. Suppression of NF-kappaB and NF-kappaB-regulated gene expression by sulforaphane and PEITC through IkappaBalpha, IKK pathway in human prostate cancer PC-3 cells[J]. Oncogene, 2005, 24(28): 4486-4495.
doi: 10.1038/sj.onc.1208656 pmid: 15856023 |
| [22] | Hung CN, Huang HP, Wang CJ, et al. Sulforaphane inhibits TNF-α-induced adhesion molecule expression through the Rho A/ROCK/NF-κB signaling pathway[J]. J Med Food, 2014, 17(10): 1095-1102. |
| [23] | Jhang KA, Park JS, Kim HS, et al. Sulforaphane rescues amyloid-β peptide-mediated decrease in MerTK expression through its anti-inflammatory effect in human THP-1 macrophages[J]. J Neuroinflammation, 2018, 15(1): 75. |
| [24] | Heiss E, Gerhäuser C. Time-dependent modulation of thioredoxin reductase activity might contribute to sulforaphane-mediated inhibition of NF-kappaB binding to DNA[J]. Antioxid Redox Signal, 2005, 7(11-12): 1601-1611. |
| [25] |
Reddy SA, Shelar SB, Dang TM, et al. Sulforaphane and its methylcarbonyl analogs inhibit the LPS-stimulated inflammatory response in human monocytes through modulating cytokine production, suppressing chemotactic migration and phagocytosis in a NF-κB- and MAPK-dependent manner[J]. Int Immunopharmacol, 2015, 24(2): 440-450.
doi: S1567-5769(15)00003-X pmid: 25585231 |
| [26] |
Murata W, Yamaguchi Y, Fujita KI, et al. Enhancement of paraben-fungicidal activity by sulforaphane, a cruciferous vegetable-derived isothiocyanate, via membrane structural damage in Saccha-romyces cerevisiae[J]. Lett Appl Microbiol, 2019, 69(6): 403-410.
doi: 10.1111/lam.13230 pmid: 31596500 |
| [27] |
Ordonez AA, Korin Bullen C, Villabona-Rueda AF, et al. Sulforaphane exhibits antiviral activity against pandemic SARS-CoV-2 and seasonal HCoV-OC43 coronaviruses in vitro and in mice[J]. Commun Biol, 2022, 5(1): 242.
doi: 10.1038/s42003-022-03189-z pmid: 35304580 |
| [28] | Janczewski Ł. Sulforaphane and its bifunctional analogs: synthesis and biological activity[J]. Molecules, 2022, 27(5): 1750. |
| [29] | Moon JK, Kim JR, Ahn YJ, et al. Analysis and anti-Helicobacter activity of sulforaphane and related compounds present in broccoli(Brassica oleracea L.)sprouts[J]. J Agric Food Chem, 2010, 58(11): 6672-6677. |
| [30] |
Fahey JW, Haristoy X, Dolan PM, et al. Sulforaphane inhibits extracellular, intracellular, and antibiotic-resistant strains of Hel-icobacter pylori and prevents benzo[a]Pyrene-induced stomach tumors[J]. Proc Natl Acad Sci USA, 2002, 99(11): 7610-7615.
doi: 10.1073/pnas.112203099 pmid: 12032331 |
| [31] |
Deramaudt TB, Ali M, Vinit S, et al. Sulforaphane reduces intracellular survival of Staphylococcus aureus in macrophages through inhibition of JNK and p38 MAPK-induced inflammation[J]. Int J Mol Med, 2020, 45(6): 1927-1941.
doi: 10.3892/ijmm.2020.4563 pmid: 32323751 |
| [32] | Bendary MM, Ali MAM, Abdel Halim AS, et al. Investigating Sulforaphane's anti-virulence and anti-quorum sensing properties against Pseudomonas aeruginosa[J]. Front Pharmacol, 2024, 15: 1406653. |
| [33] |
Lenzi M, Fimognari C, Hrelia P. Sulforaphane as a promising molecule for fighting cancer[J]. Cancer Treat Res, 2014, 159: 207-223.
doi: 10.1007/978-3-642-38007-5_12 pmid: 24114482 |
| [34] | Su XL, Jiang X, Meng LB, et al. Anticancer activity of sulforaphane: the epigenetic mechanisms and the Nrf2 signaling pathway[J]. Oxid Med Cell Longev, 2018, 2018: 5438179. |
| [35] | Pradhan N, Kar S, Parbin S, et al. Epigenetic dietary interventions for prevention of cancer[M]// Epigenetics of Cancer Prevention. Amsterdam: Elsevier, 2019: 23-48. |
| [36] | Asif Ali M, Khan N, Kaleem N, et al. Anticancer properties of sulforaphane: current insights at the molecular level[J]. Front Oncol, 2023, 13: 1168321. |
| [37] |
Ho E, Clarke JD, Dashwood RH. Dietary sulforaphane, a histone deacetylase inhibitor for cancer prevention[J]. J Nutr, 2009, 139(12): 2393-2396.
doi: 10.3945/jn.109.113332 pmid: 19812222 |
| [38] | Kim HJ, Bae SC. Histone deacetylase inhibitors: molecular mechanisms of action and clinical trials as anti-cancer drugs[J]. Am J Transl Res, 2011, 3(2): 166-179. |
| [39] |
Atwell LL, Beaver LM, Shannon J, et al. Epigenetic regulation by sulforaphane: opportunities for breast and prostate cancer chemoprevention[J]. Curr Pharmacol Rep, 2015, 1(2): 102-111.
pmid: 26042194 |
| [40] | Jiang LL, Zhou SJ, Zhang XM, et al. Sulforaphane suppresses in vitro and in vivo lung tumorigenesis through downregulation of HDAC activity[J]. Biomed Pharmacother, 2016, 78: 74-80. |
| [41] |
da Silva dos Santos PW, Machado ART, De Grandis RA, et al. Transcriptome and DNA methylation changes modulated by sulforaphane induce cell cycle arrest, apoptosis, DNA damage, and suppression of proliferation in human liver cancer cells[J]. Food Chem Toxicol, 2020, 136: 111047.
doi: 10.1016/j.fct.2019.111047 pmid: 31838189 |
| [42] |
Abbaoui B, Telu KH, Lucas CR, et al. The impact of cruciferous vegetable isothiocyanates on histone acetylation and histone phosphorylation in bladder cancer[J]. J Proteomics, 2017, 156: 94-103.
doi: S1874-3919(17)30026-X pmid: 28132875 |
| [43] |
Denis H, Ndlovu MN, Fuks F. Regulation of mammalian DNA methyltransferases: a route to new mechanisms[J]. EMBO Rep, 2011, 12(7): 647-656.
doi: 10.1038/embor.2011.110 pmid: 21660058 |
| [44] |
Dawson MA, Kouzarides T. Cancer epigenetics: from mechanism to therapy[J]. Cell, 2012, 150(1): 12-27.
doi: 10.1016/j.cell.2012.06.013 pmid: 22770212 |
| [45] | Ali Khan M, Kedhari Sundaram M, Hamza A, et al. Sulforaphane reverses the expression of various tumor suppressor genes by targeting DNMT3B and HDAC1 in human cervical cancer cells[J]. Evid Based Complement Alternat Med, 2015, 2015: 412149. |
| [46] | Hsu A, Wong CP, Yu Z, et al. Promoter de-methylation of cyclin D2 by sulforaphane in prostate cancer cells[J]. Clin Epigenetics, 2011, 3(1): 3. |
| [47] |
Hyun TK. A recent overview on sulforaphane as a dietary epigenetic modulator[J]. EXCLI J, 2020, 19: 131-134.
doi: 10.17179/excli2019-2039 pmid: 32194360 |
| [48] | Mattick JS, Makunin IV. Non-coding RNA[J]. Hum Mol Genet, 2006, 15(suppl_1): R17-R29. |
| [49] |
Rafiei H, Ashrafizadeh M, Ahmadi Z. microRNAs as novel targets of sulforaphane in cancer therapy: The beginning of a new tale?[J]. Phytother Res, 2020, 34(4): 721-728.
doi: 10.1002/ptr.6572 pmid: 31972874 |
| [50] | Kalhori MR, Khodayari H, Khodayari S, et al. Regulation of long non-coding RNAs by plant secondary metabolites: a novel anticancer therapeutic approach[J]. Cancers, 2021, 13(6): 1274. |
| [51] | Liu CM, Peng CY, Liao YW, et al. Sulforaphane targets cancer stemness and tumor initiating properties in oral squamous cell carcinomas via miR-200c induction[J]. J Formos Med Assoc, 2017, 116(1): 41-48. |
| [52] | Martin SL, Kala R, Tollefsbol TO. Mechanisms for the inhibition of colon cancer cells by sulforaphane through epigenetic modulation of microRNA-21 and human telomerase reverse transcriptase(hTERT)down-regulation[J]. Curr Cancer Drug Targets, 2018, 18(1): 97-106. |
| [53] | Wang DX, Zou YJ, Zhuang XB, et al. Sulforaphane suppresses EMT and metastasis in human lung cancer through miR-616-5p-mediated GSK3β/β-catenin signaling pathways[J]. Acta Pharmacol Sin, 2017, 38(2): 241-251. |
| [54] |
Beaver LM, Kuintzle R, Buchanan A, et al. Long noncoding RNAs and sulforaphane: a target for chemoprevention and suppression of prostate cancer[J]. J Nutr Biochem, 2017, 42: 72-83.
doi: S0955-2863(16)30565-4 pmid: 28131897 |
| [55] |
Bhat SA, Kamal MA, Yarla NS, et al. Synopsis on managment strategies for neurodegenerative disorders: challenges from bench to bedside in successful drug discovery and development[J]. Curr Top Med Chem, 2017, 17(12): 1371-1378.
doi: 10.2174/1568026616666161222121229 pmid: 28017151 |
| [56] | Ladak Z, Garcia E, Yoon J, et al. Sulforaphane(SFA)protects neuronal cells from oxygen & glucose deprivation(OGD)[J]. PLoS One, 2021, 16(3): e0248777. |
| [57] |
Kraft AD, Johnson DA, Johnson JA. Nuclear factor E2-related factor 2-dependent antioxidant response element activation by tert-butylhydroquinone and sulforaphane occurring preferentially in astrocytes conditions neurons against oxidative insult[J]. J Neurosci, 2004, 24(5): 1101-1112.
doi: 10.1523/JNEUROSCI.3817-03.2004 pmid: 14762128 |
| [58] |
Mizuno K, Kume T, Muto C, et al. Glutathione biosynthesis via activation of the nuclear factor E2-related factor 2(Nrf2)—antioxidant-response element(ARE)pathway is essential for neuroprotective effects of sulforaphane and 6-(methylsulfinyl)hexyl isothiocyanate[J]. J Pharmacol Sci, 2011, 115(3): 320-328.
pmid: 21358121 |
| [59] |
Hong Y, Yan W, Chen S, et al. The role of Nrf2 signaling in the regulation of antioxidants and detoxifying enzymes after traumatic brain injury in rats and mice[J]. Acta Pharmacol Sin, 2010, 31(11): 1421-1430.
doi: 10.1038/aps.2010.101 pmid: 20953205 |
| [60] | Flint Beal M. Therapeutic approaches to mitochondrial dysfunction in Parkinson's disease[J]. Parkinsonism Relat Disord, 2009, 15(Suppl 3): S189-S194. |
| [61] |
Denzer I, Münch G, Friedland K. Modulation of mitochondrial dysfunction in neurodegenerative diseases via activation of nuclear factor erythroid-2-related factor 2 by food-derived compounds[J]. Pharmacol Res, 2016, 103: 80-94.
doi: 10.1016/j.phrs.2015.11.019 pmid: 26626189 |
| [62] | Luis-García ER, Limón-Pacheco JH, Serrano-García N, et al. Sulforaphane prevents quinolinic acid-induced mitochondrial dysfunction in rat striatum[J]. J Biochem Mol Toxicol, 2017, 31(2). DOI: 10.1002/jbt.21837. |
| [63] | Hernandez-Rabaza V, Cabrera-Pastor A, Taoro-Gonzalez L, et al. Neuroinflammation increases GABAergic tone and impairs cognitive and motor function in hyperammonemia by increasing GAT-3 membrane expression. Reversal by sulforaphane by promoting M2 polarization of microglia[J]. J Neuroinflammation, 2016, 13(1): 83. |
| [64] |
Qin SS, Yang CH, Huang WH, et al. Sulforaphane attenuates microglia-mediated neuronal necroptosis through down-regulation of MAPK/NF-κB signaling pathways in LPS-activated BV-2 microglia[J]. Pharmacol Res, 2018, 133: 218-235.
doi: S1043-6618(17)31137-4 pmid: 29391237 |
| [65] |
Zhao FF, Zhang JL, Chang N. Epigenetic modification of Nrf2 by sulforaphane increases the antioxidative and anti-inflammatory capacity in a cellular model of Alzheimer's disease[J]. Eur J Pharmacol, 2018, 824: 1-10.
doi: S0014-2999(18)30059-1 pmid: 29382536 |
| [66] | Kim J, Lee S, Choi BR, et al. Sulforaphane epigenetically enhances neuronal BDNF expression and TrkB signaling pathways[J]. Mol Nutr Food Res, 2017, 61(2). DOI: 10.1002/mnfr.201600194. |
| [67] | Wu HH, Liang H, Yuan QP, et al. Preparation and stability investigation of the inclusion complex of sulforaphane with hydroxypropyl-β-cyclodextrin[J]. Carbohydr Polym, 2010, 82(3): 613-617. |
| [68] | Wu YF, Zou LG, Mao JW, et al. Stability and encapsulation efficiency of sulforaphane microencapsulated by spray drying[J]. Carbohydr Polym, 2014, 102: 497-503. |
| [69] | Lee S, Lee G, Jeon G, et al. Anti-aging and lightening effects of Au-decorated zeolite-based biocompatible nanocomposites in epidermal delivery systems[J]. J Funct Biomater, 2023, 14(2): 66. |
| [70] | Zhang J, Dong YY, Liu X, et al. Effective myocardial infarction treatment by targeted accumulation of Sulforaphane using porous magnetic silica nanoparticles[J]. Int J Pharm, 2023, 645: 123389. |
| [71] |
Ibrahim Fouad G, El-Sayed SAM, Mabrouk M, et al. Neuroprotective potential of intranasally delivered sulforaphane-loaded iron oxide nanoparticles against cisplatin-induced neurotoxicity[J]. Neurotox Res, 2022, 40(5): 1479-1498.
doi: 10.1007/s12640-022-00555-x pmid: 35969308 |
| [72] | Wu SQ, Li L, Liang QQ, et al. A DFT study of sulforaphane adsorbed on M12O12(M=Be, Mg and Ca)nanocages[J]. Mater Today Commun, 2024, 38: 107687. |
| [73] |
Danafar H, Sharafi A, Kheiri Manjili H, et al. Sulforaphane delivery using mPEG-PCL co-polymer nanoparticles to breast cancer cells[J]. Pharm Dev Technol, 2017, 22(5): 642-651.
doi: 10.3109/10837450.2016.1146296 pmid: 26916923 |
| [74] | Cristiano MC, Froiio F, Spaccapelo R, et al. Sulforaphane-loaded ultradeformable vesicles as a potential natural nanomedicine for the treatment of skin cancer diseases[J]. Pharmaceutics, 2019, 12(1): 6. |
| [75] | Azarashkan Z, Motamedzadegan A, Ghorbani-HasanSaraei A, et al. Improvement of the stability and release of sulforaphane-enriched broccoli sprout extract nanoliposomes by co-encapsulation into basil seed gum[J]. Food Bioprocess Technol, 2022, 15(7): 1573-1587. |
| [76] | Li RH, Hu XC, Shan SQ, et al. Study on the controlled release properties of modified multi-walled carbon nanotubes on sulforaphane[J]. Carbon Lett, 2024, 34(2): 757-765. |
| [77] |
Manjili HK, Ma'mani L, Naderi-Manesh H. Monodisperse rattle-structured gold nanorod-mesoporous silica nanoparticles core-shell as sulforaphane carrier and its sustained-release property[J]. Drug Res, 2018, 68(9): 504-513.
doi: 10.1055/a-0573-8966 pmid: 29660748 |
| [78] | Lu WJ, Du FF, Zhao XW, et al. Sulforaphane-conjugated carbon dots: a versatile nanosystem for targeted imaging and inhibition of EGFR-overexpressing cancer cells[J]. ACS Biomater Sci Eng, 2019, 5(9): 4692-4699. |
| [79] | Ibrahim Fouad G, Mabrouk M, El-Sayed SAM, et al. Neurotherapeutic efficacy of loaded sulforaphane on iron oxide nanoparticles against cuprizone-induced neurotoxicity: role of MMP-9 and S100β[J]. Toxicol Mech Methods, 2023, 33(6): 463-479. |
| [80] | Zhang P, Li TT, Liu C, et al. Nano-sulforaphane attenuates PhIP-induced early abnormal embryonic neuro-development[J]. Ann Anat, 2021, 233: 151617. |
| [81] | Danafar H, Shara\ufb 01 A, Kheiri S, et al. Co-delivery of sulforaphane and curcumin with PEGylated iron oxide-gold core shell nanoparticles for delivery to breast cancer cell line[J]. Iran J Pharm Res, 2018, 17(2): 480-494. |
| [82] | Gu HF, Ren FZ, Mao XY, et al. Mineralized and GSH-responsive hyaluronic acid based nano-carriers for potentiating repressive effects of sulforaphane on breast cancer stem cells-like properties[J]. Carbohydr Polym, 2021, 269: 118294. |
| [83] |
Xu Y, Han XX, Li YY, et al. Sulforaphane mediates glutathione depletion via polymeric nanoparticles to restore cisplatin chemosensitivity[J]. ACS Nano, 2019, 13(11): 13445-13455.
doi: 10.1021/acsnano.9b07032 pmid: 31670945 |
| [84] | Dutta D, Siddiqui L, Shah S, et al. Synergistic anticancer activity of Sulphoraphane and Teriflunomide co loaded lignin nanoparticles against triple negative breast cancer: Targeted nanoparticle delivery and drug repurposing[J]. Med Hypotheses, 2024, 189: 111404. |
| [85] | Zhang X, Peng X, Hui JQ, et al. Sulforaphane functionalized chitosan nanoparticles serve as an effective nano complex system for the treatment of human cervical cancer[J]. J Clust Sci, 2023, 34(6): 2963-2975. |
| [86] | Li SM, Xu ZK, Alrobaian M, et al. EGF-functionalized lipid-polymer hybrid nanoparticles of 5-fluorouracil and sulforaphane with enhanced bioavailability and anticancer activity against colon carcinoma[J]. Biotechnol Appl Biochem, 2022, 69(5): 2205-2221. |
| [87] |
Soni K, Mujtaba A, Akhter MH, et al. Optimisation of ethosomal nanogel for topical nano-CUR and sulphoraphane delivery in effective skin cancer therapy[J]. J Microencapsul, 2020, 37(2): 91-108.
doi: 10.1080/02652048.2019.1701114 pmid: 31810417 |
| [88] | Yepes-Molina L, Pérez-Jiménez MI, Martínez-Esparza M, et al. Membrane vesicles for nanoencapsulated sulforaphane increased their anti-inflammatory role on an in vitro human macrophage model[J]. Int J Mol Sci, 2022, 23(4): 1940. |
| [89] | Ramírez-Pavez T, García-Peñaranda A, Garcia-Ibañez P, et al. Potential of sulforaphane and broccoli membrane vesicles as regulators of M1/M2 human macrophage activity[J]. Int J Mol Sci, 2022, 23(19): 11141. |
| [90] | Essien EN, Revi N, Khatri V, et al. Methotrexate and sulforaphane loaded PBA-G5-PAMAM dendrimers as a combination therapy for anti-inflammatory response in an intra-articular joint arthritic animal model[J]. Int J Pharm, 2023, 642: 123150. |
| [91] |
黄心童, 耿宇昊, 刘恒源, 等. 微流控制备新型功能纳米粒子研究进展[J]. 化工学报, 2023, 74(1): 355-364.
doi: 10.11949/0438-1157.20220935 |
|
Huang XT, Geng YH, Liu HY, et al. Research progress on new functional nanoparticles prepared by microfluidic technology[J]. CIESC J, 2023, 74(1): 355-364.
doi: 10.11949/0438-1157.20220935 |
|
| [92] | 董翠芳, 刘树恒, 刘长霞. 环境响应壳聚糖纳米材料的制备及其应用研究进展[J]. 沧州师范学院学报, 2017, 33(2): 49-52, 57. |
| Dong CF, Liu SH, Liu CX. Research progress in preparation and application of environment-responsive chitosan nanomaterial[J]. J Cangzhou Norm Univ, 2017, 33(2): 49-52, 57. |
| [1] | 蔡楠, 方静平, 陈必链, 何勇锦. 高值化等鞭金藻固碳研究进展[J]. 生物技术通报, 2024, 40(6): 68-80. |
| [2] | 王楠, 廖永琴, 施竹凤, 申云鑫, 杨童雨, 冯路遥, 矣小鹏, 唐加菜, 陈齐斌, 杨佩文. 三株无量山森林土壤芽孢杆菌鉴定及其生物活性挖掘[J]. 生物技术通报, 2024, 40(2): 277-288. |
| [3] | 康晓博, 张璟汐, 卢甜甜, 刘亚月, 周龙建, 张翼. 不同盐度培养下海洋真菌Aspergillus unguis DLEP2008001生物活性及次生代谢组变化[J]. 生物技术通报, 2024, 40(11): 296-311. |
| [4] | 赵睿萌, 王梦雨, 吕国英, 宋婷婷, 张作法. 药用真菌桑黄中多酚类成分药用机理研究进展[J]. 生物技术通报, 2024, 40(11): 3-13. |
| [5] | 游子娟, 陈汉林, 邓辅财. 鱼皮生物活性肽的提取及功能活性研究进展[J]. 生物技术通报, 2023, 39(7): 91-104. |
| [6] | 汪巧菊, 胡雨萌, 温亚亚, 宋丽, 孟闯, 潘志明, 焦新安. 新型冠状病毒S1蛋白的表达及活性鉴定[J]. 生物技术通报, 2022, 38(3): 157-163. |
| [7] | 王楠, 苏誉, 刘文杰, 封明, 毛瑜, 张新国. 植物内生菌中抗耐药微生物活性成分的研究进展[J]. 生物技术通报, 2021, 37(8): 263-274. |
| [8] | 张秀民, 马绍英, 杨洁, 包金玉, 张晓玲, 田鹏, 路亚琦, 李胜. 以次生代谢物产量为目标的西兰花毛状根培养技术体系优化[J]. 生物技术通报, 2021, 37(8): 75-84. |
| [9] | 付志强, 熊艳. 便携式生物光学传感器的研究进展[J]. 生物技术通报, 2021, 37(3): 219-226. |
| [10] | 李雪, 李俊敏, 张雷, 李杉. 细胞穿膜肽M918偶联抗体的表达纯化及其内吞效率研究[J]. 生物技术通报, 2021, 37(12): 198-204. |
| [11] | 薛帆正, 黄海辰, 吴福泉, 李晓敏, 吴小平, 傅俊生. 真菌黑色素研究现状与产业应用[J]. 生物技术通报, 2021, 37(11): 32-41. |
| [12] | 徐重新, 张存政, 刘媛, 张霄, 仲建锋, 刘贤金. 食源性致病微生物危害风险及其防控用抗菌生物活性肽研究进展[J]. 生物技术通报, 2019, 35(7): 202-212. |
| [13] | 赵祥杰, 杨文君, 杨荣玲, 吴婷婷, 王朝宇, 许宁宁, 何佳美. 花色苷生物转化修饰的研究进展[J]. 生物技术通报, 2019, 35(10): 205-211. |
| [14] | 瞿佳, 赵玲侠, 陈锐, 路鹏鹏, 孙晓宇, 沈卫荣. 抗结核海洋放线菌的筛选及菌株HY286生物活性研究[J]. 生物技术通报, 2017, 33(11): 194-199. |
| [15] | 陈龙, 梁子宁, 朱华. 植物内生菌研究进展[J]. 生物技术通报, 2015, 31(8): 30-34. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||