








生物技术通报 ›› 2026, Vol. 42 ›› Issue (4): 161-169.doi: 10.13560/j.cnki.biotech.bull.1985.2025-0692
徐玉娇1( ), 孙玉帅1, 刘道奇1, 张丽2, 张志昌2, 姚玉新1(
), 孙玉帅1, 刘道奇1, 张丽2, 张志昌2, 姚玉新1( )
)
收稿日期:2025-06-30
出版日期:2026-04-26
发布日期:2026-04-30
通讯作者:
姚玉新,男,博士,教授,研究方向 :葡萄盐碱抗性、果实次生代谢机制;E-mail: yaoyx@sdau.edu.cn作者简介:徐玉娇,女,硕士,研究方向 :葡萄抗逆栽培;E-mail: 15966391881@163.com
基金资助:
XU Yu-jiao1( ), SUN Yu-shuai1, LIU Dao-qi1, ZHANG Li2, ZHANG Zhi-chang2, YAO Yu-xin1(
), SUN Yu-shuai1, LIU Dao-qi1, ZHANG Li2, ZHANG Zhi-chang2, YAO Yu-xin1( )
)
Received:2025-06-30
Published:2026-04-26
Online:2026-04-30
摘要:
目的 盐碱胁迫严重影响葡萄生长、发育和品质形成,评价VvHSP18.2在调节葡萄盐碱抗性上的功能,鉴定葡萄抗盐碱关键基因,为葡萄抗逆遗传改良奠定理论基础。 方法 运用生物信息学技术构建VvHSP18.2进化树,分析其启动子,利用RT-qPCR技术分析VvHSP18.2在NaHCO3等各种胁迫处理后的表达水平,将VvHSP18.2在拟南芥和葡萄中过表达,通过表型分析和生理指标鉴定其功能。 结果 VvHSP18.2属于热激蛋白CI亚家族,主要在葡萄愈伤组织、果实和根系中表达。其启动子区含有多个响应干旱、盐、高温胁迫的顺式作用元件。VvHSP18.2快速响应多个非生物胁迫;在处理后6和12 h,VvHSP18.2的表达受NaCl,尤其是NaHCO3显著诱导;在处理后1 h,其表达受PEG6000显著诱导;在AlCl3处理后3和6 h,VvHSP18.2的表达显著降低。在葡萄根系和拟南芥中,VvHSP18.2过表达能缓解NaHCO3处理导致的表型伤害,降低电导率和MDA含量,提高NaHCO3胁迫抗性。并且,VvHSP18.2过表达提高了拟南芥SOD、CAT、POD酶活性,降低O2-和H2O2积累水平,表明其通过提高活性氧清除能力来提高胁迫抗性。相比之下,VvHSP18.2过表达对拟南芥NaCl抗性无显著影响。 结论 VvHSP18.2参与调控葡萄胁迫应激反应,VvHSP18.2过表达通过提高植物抗氧化酶活性和活性氧清除能力来调控活性氧代谢平衡,进而提高葡萄和拟南芥的NaHCO3胁迫抗性。
徐玉娇, 孙玉帅, 刘道奇, 张丽, 张志昌, 姚玉新. VvHSP18.2过表达调节葡萄盐碱抗性的功能分析[J]. 生物技术通报, 2026, 42(4): 161-169.
XU Yu-jiao, SUN Yu-shuai, LIU Dao-qi, ZHANG Li, ZHANG Zhi-chang, YAO Yu-xin. Functional Analysis of VvHSP18.2 Overexpression in Regulating Salt-alkali Resistance in Grapevines[J]. Biotechnology Bulletin, 2026, 42(4): 161-169.
引物 Primer | 正向引物 Forward primer (5′‒3′) | 反向引物 Reverse primer (5′‒3′) |
|---|---|---|
| VvHSP18.2 | GCAGAGGTCAAGAAACCCGA | CCTCAAAGCATGCTCTTCCT |
| β-Actin | GAGATTCCGTTGTCCAGAAGTC | CAATGTTGCCATAGAGGTCCTT |
表1 RT-qPCR引物序列
Table 1 Primer sequences for RT-qPCR
引物 Primer | 正向引物 Forward primer (5′‒3′) | 反向引物 Reverse primer (5′‒3′) |
|---|---|---|
| VvHSP18.2 | GCAGAGGTCAAGAAACCCGA | CCTCAAAGCATGCTCTTCCT |
| β-Actin | GAGATTCCGTTGTCCAGAAGTC | CAATGTTGCCATAGAGGTCCTT |
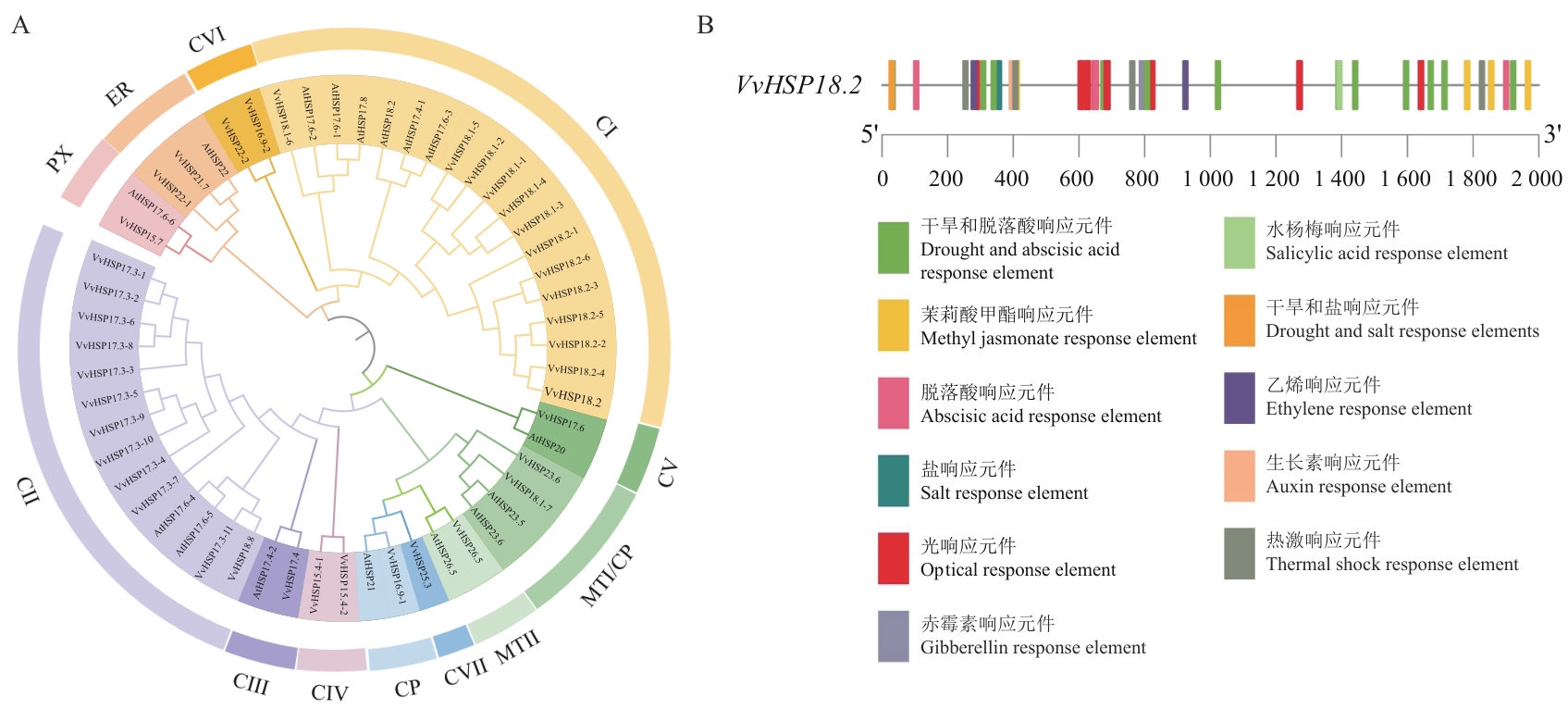
图1 VvHSP18.2系统进化及表达分析A:拟南芥和葡萄HSP蛋白的系统进化树;B:VvHSP18.2上游2 000 bp启动子分析
Fig. 1 Phylogenetic evolution and expression analysis of VvHSP18.2A: Phylogenetic tree of HSP proteins in Arabidopsis thaliana (At) and Vitis vinifera (Vv). B: Analysis of the 2 000 bp promoter before VvHSP18.2
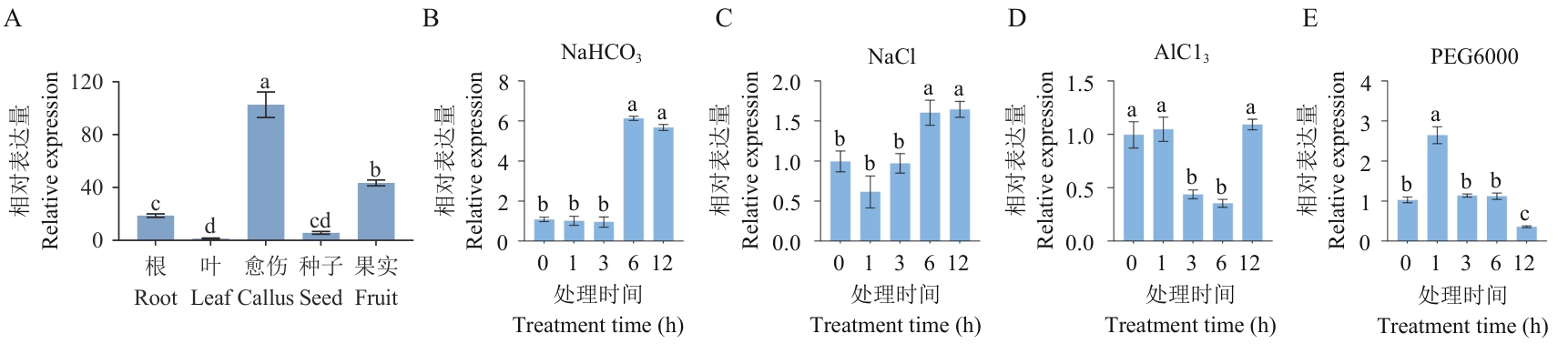
图2 VvHSP18.2组织特异性分析和非生物胁迫响应分析A:VvHSP18.2组织特异性分析;B:NaHCO3处理;C:NaCl处理;D:AlCl3处理;E:PEG6000处理。不同的小写字母表示显著差异(P<0.05)。下同
Fig. 2 VvHSP18.2 tissue-specific analysis and abiotic stress response analysisA: VvHSP18.2 tissue-specific analysis. B: NaHCO3 treatment. C: NaCl treatment. D: AlCl3 treatment. E: PEG6000treatment. Different lowercase letters indicate significant differences (P<0.05). The same below
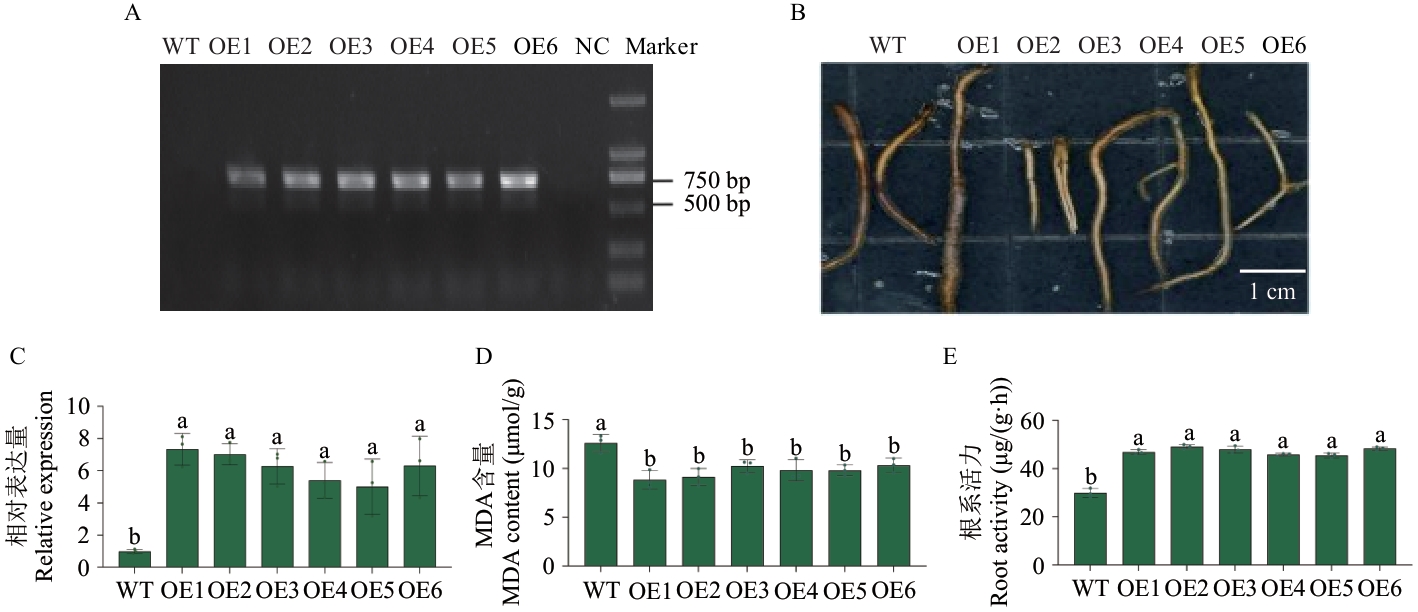
图3 过表达VvHSP18.2减轻葡萄根系碱性盐胁迫伤害A:VvHSP18.2转基因葡萄根系鉴定(NC:阴性对照);B:转基因葡萄根系受碱性盐胁迫伤害程度表型;C:转基因葡萄根系中VvHSP18.2的表达水平;D:MDA含量;E:根系活力
Fig. 3 Overexpression of VvHSP18.2 alleviates alkaline salt stress injury in grapevine rootsA: Root system identificationof VvHSP18.2 transgenic grape (NC: Negative control). B: Phenotype of damage to the root system of genetically modified grapes under alkaline salt stress. C: The expressions of VvHSP18.2 in the root system of genetically modified grapes. D: MDA content. E: Root activity
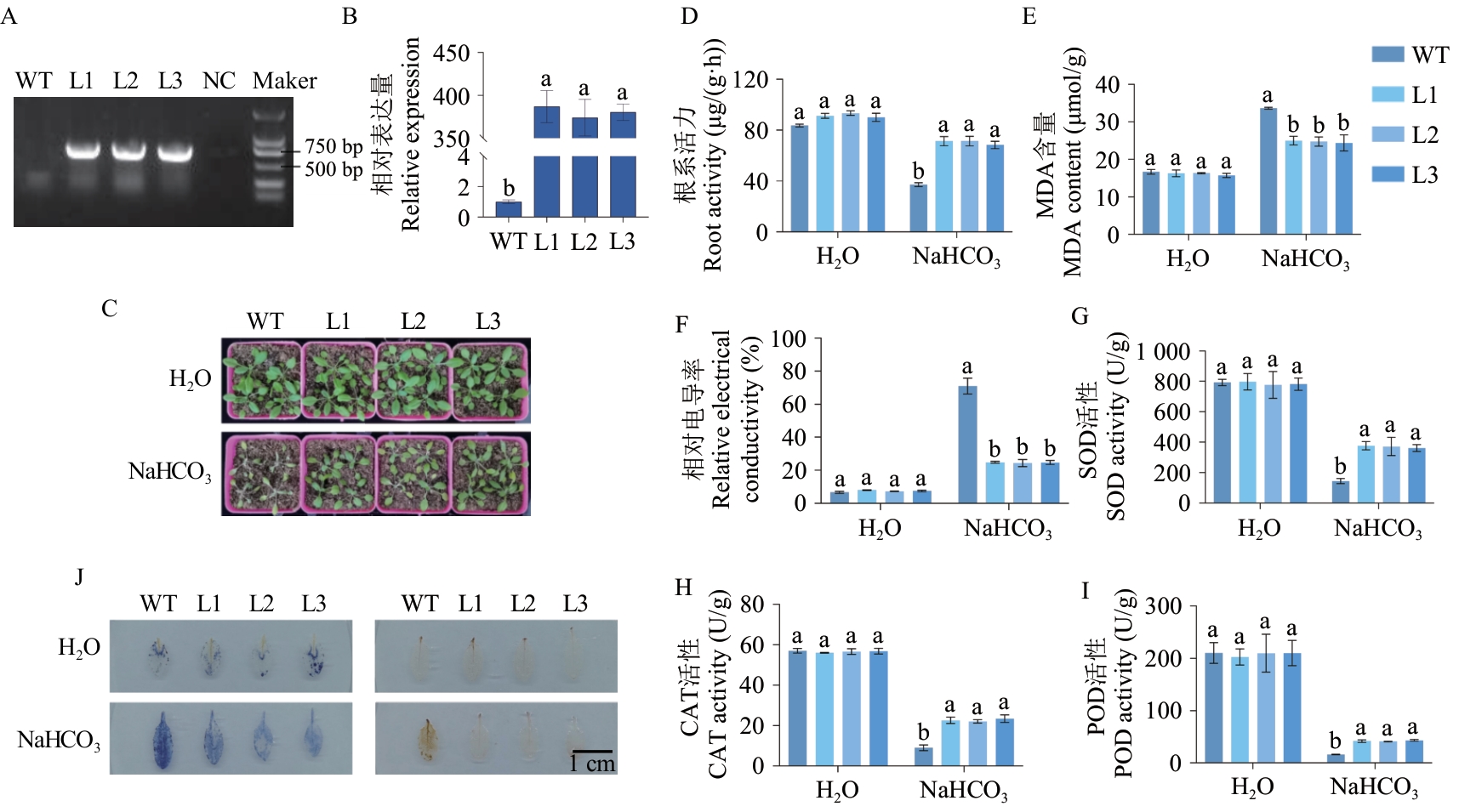
图4 VvHSP18.2转基因拟南芥的鉴定和过表达VvHSP18.2增强拟南芥NaHCO3抗性A:VvHSP18.2转基因拟南芥鉴定;B:转基因拟南芥中VvHSP18.2的表达水平;C:表型;D:根系活力;E:MDA含量;F:相对电导率;G:SOD活性;H:CAT活性;I:POD活性;J:NBT染色(左)和DAB染色(右);WT:野生型拟南芥;L1、L2、L3:3个VvHSP18.2过表达拟南芥株系
Fig. 4 Identification and overexpression of VvHSP18.2 transgenic Arabidopsis thaliana to enhance Arabidopsis NaHCO3 toleranceA: Identification of VvHSP18.2 transgenic Arabidopsis. B: Expressions of VvHSP18.2 in transgenic Arabidopsis phenotype chart. C: Phenotype chart. D: Root activity content. E: MDA content. F: Relative electrical conductivity. G: SOD activity. H: CAT activity. I: POD activity. J: NBT staining (left) and DAB staining (right). WT: Wild-type Arabidopsisthaliana background. L1, L2, and L3: Three transgenic lines harboring the VvHSP18.2 overexpression construct
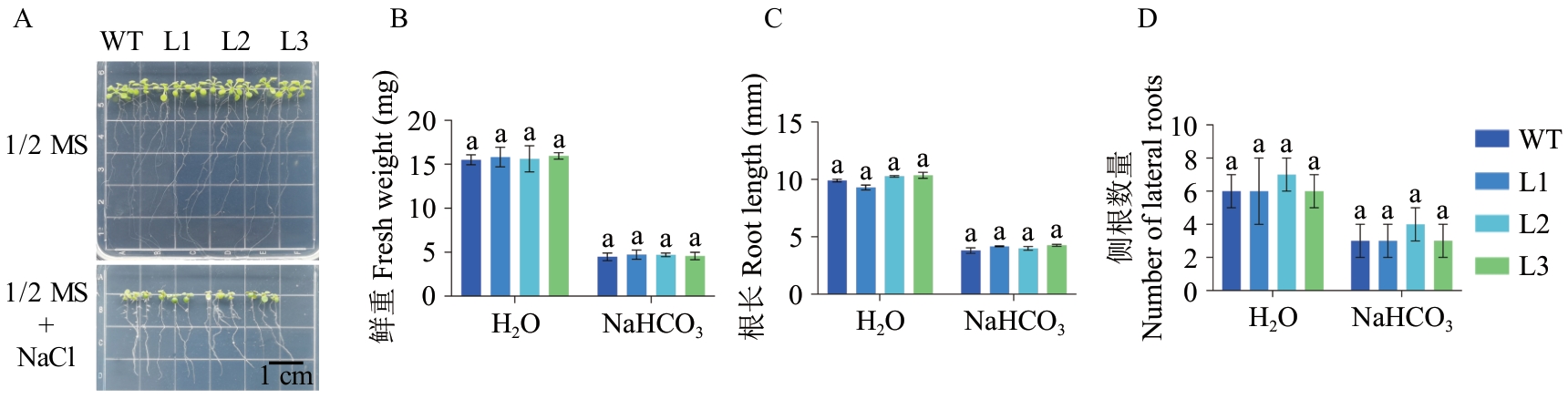
图5 过表达VvHSP18.2对拟南芥NaCl抗性的影响A:表型;B:鲜重;C:根长;D:侧根数目
Fig. 5 Effect of overexpression of VvHSP18.2 on NaCl resistance in Arabidopsis thalianaA: Phenotypes. B: Fresh weight. C: Root length. D: Number of lateral roots
| [1] | 徐莉, 谢祥恩, 陈毅群. 葡萄抗逆相关转录因子的研究进展[J]. 生物过程, 2024, 14(4): 235-241. |
| Xu L, Xie XE, Chen YQ. Research advances of transcription factors in grape stress resistance [J]. Bioprocess, 2024, 14(4): 235-241. | |
| [2] | 赵秀梅. 葡萄组培苗耐盐筛选技术研究[D]. 杨凌: 西北农林科技大学, 2004. |
| Zhao XM. Study on salt tolerance screening technology of grape tissue culture seedlings [D]. Yangling: Northwest A & F University, 2004. | |
| [3] | 朱自果, 韩燕, 李颖芳, 等. 葡萄耐盐碱性研究进展[J]. 落叶果树, 2024, 56(2): 66-69. |
| Zhu ZG, Han Y, Li YF, et al. Research progress on saline-alkalisalt-alkaline tolerance of grape [J]. Deciduous Fruits, 2024, 56(2): 66-69. | |
| [4] | Wang WX, Vinocur B, Shoseyov O, et al. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response [J]. Trends Plant Sci, 2004, 9(5): 244-252. |
| [5] | Vierling E. The roles of heat shock proteins in plants [J]. Annu Rev Plant Physiol Plant Mol Biol, 1991, 42: 579-620. |
| [6] | Waters ER, Lee GJ, Vierling E. Evolution, structure and function of the small heat shock proteins in plants [J]. J Exp Bot, 1996, 47(3): 325-338. |
| [7] | 张琼, 王锦霞, 孟诗琪, 等. 甜菜热激蛋白基因BvHSP18.2的克隆和生物信息学分析 [J]. 中国农学通报, 2022, 38(27): 111-118. |
| Zhang Q, Wang JX, Meng SQ, et al. Sugar beet heat-shock protein gene BvHSP18.2: cloning and bioinformatics analysis [J]. Chin Agric Sci Bull, 2022, 38(27): 111-118. | |
| [8] | Kim DH, Xu ZY, Hwang I. AtHSP17.8 overexpression in transgenic lettuce gives rise to dehydration and salt stress resistance phenotypes through modulation of ABA-mediated signaling [J]. Plant Cell Rep, 2013, 32(12): 1953-1963. |
| [9] | Wang X, Zhang H, Shao LY, et al. Expression and function analysis of a rice OsHSP40 gene under salt stress [J]. Genes Genomics, 2019, 41(2): 175-182. |
| [10] | Sato Y, Yokoya S. Enhanced tolerance to drought stress in transgenic rice plants overexpressing a small heat-shock protein, sHSP17.7 [J]. Plant Cell Rep, 2008, 27(2): 329-334. |
| [11] | Wang J, Gao X, Dong J, et al. Over-expression of the heat-responsive wheat gene TaHSP23.9 in transgenic Arabidopsis conferred tolerance to heat and salt stress [J]. Front Plant Sci, 2020, 11: 243. |
| [12] | Yang T, Zhang L, Hao HY, et al. Nuclear-localized AtHSPR links abscisic acid-dependent salt tolerance and antioxidant defense in Arabidopsis [J]. Plant J, 2015, 84(6): 1274-1294. |
| [13] | Ma W, Li J, Liu FJ, et al. GhHSP24.7 mediates mitochondrial protein acetylation to regulate stomatal conductance in response to abiotic stress in cotton [J]. Crop J, 2023, 11(4): 1128-1139. |
| [14] | 易小渠. 狗牙根干旱高温胁迫相关基因的克隆和功能解析 [D]. 武汉: 华中农业大学, 2020. |
| Yi XQ. Cloning and functional elucidation of genes related to drought and high temperature stress in canine root [D]. Wuhan: Huazhong Agricultural University, 2020. | |
| [15] | Zhang FJ, Li ZY, Zhang DE, et al. Identification of Hsp20 gene family in Malus domestica and functional characterization of Hsp20 class I gene MdHsp18.2b [J]. Physiol Plant, 2024, 176(2): e14288. |
| [16] | 曹晓蕾. 葡萄MYC2提高碱性盐耐性的分子机制[D]. 泰安: 山东农业大学, 2022. |
| Cao XL. Molecular mechanism of improving alkaline salt tolerance in grape MYC2 [D]. Tai’an: Shandong Agricultural University, 2022. | |
| [17] | Jin ZX, Lv XN, Sun YS, et al. Comprehensive discovery of salt-responsive alternative splicing events based on Iso-Seq and RNA-seq in grapevine roots [J]. Environ Exp Bot, 2021, 192: 104645. |
| [18] | 贾海红. GhMKK9介导的MAPK级联信号途径调控棉花抗枯萎病的分子机理研究 [D]. 泰安: 山东农业大学, 2018. |
| Jia HH. Molecular mechanism of GhMKK9 mediated MAPK cascade signaling pathway in regulating resistance to cotton Fusarium wilt [D]. Tai’an: Shandong Agricultural University, 2018. | |
| [19] | 相广庆. VvERF1B促进草酸分泌提高葡萄根系碱性盐耐性的分子机制 [D]. 泰安: 山东农业大学, 2024. |
| Xiang GQ. Molecular mechanisms underlying VvERF1B promoting oxalate acid secretion and improving alkaline salt tolerance in grapevine roots [D]. Tai’an: Shandong Agricultural University, 2024. | |
| [20] | Velikova V, Yordanov I, Edreva A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants protective role of exogenous polyamines [J]. Plant Sci, 2000, 151(1): 59-66. |
| [21] | 逯明辉, 宋慧, 李晓明, 等. 冷害过程中黄瓜叶片SOD、CAT和POD活性的变化 [J]. 西北植物学报, 2005, 25(8): 1570-1573. |
| Lu MH, Song H, Li XM, et al. Changes of SOD, CAT and POD activities in cucumber leaves during cold damage [J]. Acta Bot Boreali Occidentalia Sin, 2005, 25(8): 1570-1573. | |
| [22] | Zhang M, Shen ZW, Meng GQ, et al. Genome-wide analysis of the Brachypodium distachyon (L.) P. Beauv. Hsp90 gene family reveals molecular evolution and expression profiling under drought and salt stresses [J]. PLoS One, 2017, 12(12): e0189187. |
| [23] | 于洪涛. 斧翅沙芥叶片响应干旱胁迫的转录组学和蛋白质组学分析 [D]. 呼和浩特: 内蒙古农业大学, 2024. |
| Yu HT. Transcriptomic and proteomic analysis of Pugionium dolabratum maxim. leaves responsed to drought stress [D]. Hohhot: Inner Mongolia Agricultural University, 2024. | |
| [24] | Zhao P, Wang D, Wang R, et al. Genome-wide analysis of the potato Hsp20 gene family: identification, genomic organization and expression profiles in response to heat stress [J]. BMC Genomics, 2018, 19(1): 61. |
| [25] | Zhang BS, Liu Z, Zhou RN, et al. Genome-wide analysis of soybean DnaJA-family genes and functional characterization of GmDnaJA6 responses to saline and alkaline stress [J]. Crop J, 2023, 11(4): 1230-1241. |
| [26] | 杨永青, 郭岩. 热激蛋白Hsp17.6CII在调控植物耐盐碱中的应用: CN201611224135.1 [P]. 2020-02-14. |
| Yang YQ, Guo Y. Use of heat shock protein Hsp17.6CII in regulating plant salt-alkali tolerance: CN201611224135.1 [P]. 2020-02-14. | |
| [27] | Zhai MZ, Sun YD, Jia CX, et al. Over-expression of JrsHSP17.3 gene from Juglans regia confer the tolerance to abnormal temperature and NaCl stresses [J]. J Plant Biol, 2016, 59(5): 549-558. |
| [28] | Ni ZY, Liu N, Yu YH, et al. The cotton 70-kDa heat shock protein GhHSP70-26 plays a positive role in the drought stress response [J]. Environ Exp Bot, 2021,191: 104628. |
| [29] | 仲维婷, 张琼, 兴旺, 刘大丽.异源表达甜菜BvHSP18.2基因增强拟南芥对镉胁迫的耐受性研究 [J].中国农学通报, 2024, 40(29): 14-20 |
| Zhong WT, Zhang Q, Xing W, Liu DL. Heterologous expression of sugar beet BvHSP18.2 gene enhances tolerance to cadmium stress in Arabidopsis thaliana [J]. Chinese Agricultural Science Bulletin, 2024, 40(29): 14-20. | |
| [30] | Chang HW, Wu TT, Shalmani A, et al. Heat shock protein HvHSP16.9 from wild barley enhances tolerance to salt stress [J]. Physiol Mol Biol Plants, 2024, 30(5): 687-704. |
| [31] | Lee KW, Cha JY, Kim KH, et al. Overexpression of alfalfa mitochondrial HSP23 in prokaryotic and eukaryotic model systems confers enhanced tolerance to salinity and arsenic stress [J]. Biotechnology Letters, 2012, 34: 167-174. |
| [32] | Wu J, Gao T, Hu J, et al. Research advances in function and regulation mechanisms of plant small heat shock proteins (sHSPs) under environmental stresses [J]. Sci Total Environ, 2022, 15: 825:154054. |
| [33] | Xu JY, Xue CC, Xue D, et al. Overexpression of GmHsp90s, a heat shock protein 90 (Hsp90) gene family cloning from soybean, decrease damage of abiotic stresses in Arabidopsis thaliana [J]. PLoS One, 2013, 8(7): e69810. |
| [34] | Ono K, Hibino T, Kohinata T, et al. Overexpression of DnaK from a halotolerantcyanobacterium aphanothece halophytica enhances the high-temperatue tolerance of tobacco during germination and early growth [J]. Plant Sci, 2001, 160(3): 455-461. |
| [35] | Sung DY, Guy CL. Physiological and molecular assessment of altered expression of Hsp70-1 in Arabidopsis. Evidence for pleiotropic consequences [J]. Plant Physiol, 2003, 132(2): 979-987. |
| [36] | Alvim FC, Carolino SM, Cascardo JC, et al. Enhanced accumulation of BiP in transgenic plants confers tolerance to water stress [J]. Plant Physiol, 2001, 126(3): 1042-1054. |
| [37] | Sugino M, Hibino T, Tanaka Y, et al. Overexpression of DnaK from a halotolerantcyanobacterium aphanothece halophytica acquires resistance to salt stress in transgenic tobacco plants [J]. Plant Sci, 1999, 146(2): 81-88. |
| [38] | Jiang C, Bi Y, Li M, et al. A small heat shock protein gene (RcHSP17.8) from Chinese rose confers resistance to various abiotic stresses in transgenic tobacco [J]. Plant Cell Tissue Organ Cult, 2020, 141 (2), 407-415. |
| [39] | Fu C, Liu X, Yang W, et al. Enhanced salt tolerance in tomato plants constitutively expressing heat-shock protein in the endoplasmic reticulum [J]. Genet Mol Res, 2016, 15(2): gmr.15028301. |
| [40] | Sun J, Cheng G, Huang L, et al. Modified expression of a heat shock protein gene, CaHSP22.0, results in high sensitivity to heat and salt stress in pepper (Capsicum annuum L.) [J]. Sci. Hortic, 2019, 249: 364-373. |
| [41] | Sun XB, Sun CY, Li ZG, et al. AsHSP17, a creeping bentgrass small heat shock protein modulates plant photosynthesis and ABA-dependent and independent signalling to attenuate plant response to abiotic stress [J]. Plant Cell Environ, 2016, 39(6): 1320-1337. |
| [1] | 李亚妮, 韩鸿宇, 耿梦爽, 米若兰, 王韦琪, 于文静, 孟宪文, 李传友. ChiC基因调控番茄灰霉病抗性的机制研究[J]. 生物技术通报, 2026, 42(3): 255-262. |
| [2] | 张高翔, 吴玉碧, 郭亚静, 纪薇, 杨忠义. 葡萄WD40基因家族鉴定及表达量分析[J]. 生物技术通报, 2026, 42(3): 324-337. |
| [3] | 彭楚, 孙娟利, 郑蓓蓓, 张若西, 韩月彭, 赵云. 果实花色苷酶促降解机制研究进展[J]. 生物技术通报, 2026, 42(3): 133-144. |
| [4] | 王芳, 邵会茹, 吕林龙, 赵点, 胡振, 吕建珍, 姜亮. 植物和细菌TurboID邻近蛋白标记方法的建立[J]. 生物技术通报, 2025, 41(9): 44-53. |
| [5] | 李珊, 马登辉, 马红义, 姚文孔, 尹晓. 葡萄SKP1基因家族鉴定与表达分析[J]. 生物技术通报, 2025, 41(9): 147-158. |
| [6] | 李雅琼, 格桑拉毛, 陈启迪, 杨宇环, 何花转, 赵耀飞. 异源过表达高粱SbSnRK2.1增强拟南芥对盐胁迫的抗性[J]. 生物技术通报, 2025, 41(8): 115-123. |
| [7] | 冯冰, 闫彩霞, 刘艺, 董凯悦, 赵楠, 赵瑞, 陈少良. 灰杨PcAHL17负调控拟南芥的镉耐受性[J]. 生物技术通报, 2025, 41(6): 269-283. |
| [8] | 张勇, 宋盛龙, 李永泰, 张新宇, 李艳军. 陆地棉GhSWEET9基因的克隆及抗黄萎病功能分析[J]. 生物技术通报, 2025, 41(6): 144-154. |
| [9] | 程慧娟, 王昕, 石小涛, 马东旭, 龚大春, 胡骏鹏, 谢智文. 转录因子CREA敲除对黑曲霉形态和分泌β-葡萄糖苷酶的影响[J]. 生物技术通报, 2025, 41(6): 344-354. |
| [10] | 昝舒雯, 谢欢欢, 张宇琴, 王文娟, 张鹏飞, 梁晋军, 温鹏飞. VvAGAMOUS通过VvCRABS CLAW调控葡萄心皮发育[J]. 生物技术通报, 2025, 41(5): 208-217. |
| [11] | 樊玥妮, 仙保山, 师艺萍, 任梦圆, 徐佳慧, 魏绍巍, 许晓敬, 罗晓峰, 舒凯. SPINDLY和SECRET AGENT介导的蛋白糖基化调控植物发育与逆境响应[J]. 生物技术通报, 2025, 41(4): 1-8. |
| [12] | 刘彤彤, 李肖慧, 杨骏龙, 陈旺, 玉猛, 王超凡, 王凤茹, 客绍英. ZmSTART1调控玉米维管束建成的功能研究[J]. 生物技术通报, 2025, 41(4): 115-122. |
| [13] | 刘丽, 王辉, 关天舒, 李柏宏, 于舒怡. 葡萄脱落酸受体VvPYL4互作蛋白的筛选及互作蛋白基因表达[J]. 生物技术通报, 2025, 41(4): 188-197. |
| [14] | 颜伟, 陈慧婷, 叶青, 刘广超, 刘新, 侯丽霞. 葡萄HCT基因家族鉴定及其对低温胁迫的响应[J]. 生物技术通报, 2025, 41(2): 175-186. |
| [15] | 薛晓斌, 宁琳, 周鱼, 刘虹君, 高照祖, 王振平, 李栋梅. 酿酒葡萄VvOMTs基因家族鉴定及启动子功能分析[J]. 生物技术通报, 2025, 41(12): 168-176. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||