








• 综述与专论 • 下一篇
王明藤1, 有小娟1, 孔军珂1, 李永伟1,2( ), 张莎莎3, 王庆丰3(
), 张莎莎3, 王庆丰3( )
)
收稿日期:2025-09-08
出版日期:2026-04-17
发布日期:2026-04-17
通讯作者:
李永伟lyw@hactcm.edu.cn作者简介:第一联系人:同等贡献
基金资助:
WANG Ming-teng1, YOU Xiao-juan1, KONG Jun-ke1, LI Yong-wei1,2( ), ZHANG Sha-sha3, WANG Qing-feng3(
), ZHANG Sha-sha3, WANG Qing-feng3( )
)
Received:2025-09-08
Published:2026-04-17
Online:2026-04-17
摘要:
全球范围内,多重耐药细菌的持续传播对公共卫生构成严重威胁,传统抗生素疗效持续下降,亟需探索新的抗感染策略。噬菌体可通过特异性识别并裂解宿主细菌发挥精准抗菌作用,但其应用受宿主谱有限、体内稳定性不足及免疫清除等因素制约;纳米材料通过膜破坏、产生活性氧等机制发挥广谱抗菌作用,但其在靶向性和生物相容性方面仍存在不足;二者协同构建的抗菌系统能显著增强对耐药菌感染的抑制效果。围绕噬菌体与纳米材料的协同抗菌作用,现有研究形成了若干具有代表性的组合策略,其差异主要体现在细菌特异性识别、生物膜穿透能力提升及杀菌动力学改善等方面。本文在系统梳理相关研究进展的基础上,对不同协同模式的作用特点、应用场景及研究局限进行了归纳比较。目前该策略临床转化仍面临噬菌体免疫原性、纳米材料生物相容性、规模化制备一致性及质量控制等因素的影响。未来研究应聚焦于噬菌体定向进化与修饰、可降解纳米载体开发以及标准化生产流程建立,以促进其向临床应用转化,为控制耐药菌感染提供新路径。
王明藤, 有小娟, 孔军珂, 李永伟, 张莎莎, 王庆丰. 噬菌体结合纳米材料在多重耐药细菌中的应用进展[J]. 生物技术通报, doi: 10.13560/j.cnki.biotech.bull.1985.2025-0955.
WANG Ming-teng, YOU Xiao-juan, KONG Jun-ke, LI Yong-wei, ZHANG Sha-sha, WANG Qing-feng. Advances in Bacteriophage-nanomaterial Synergistic Systems for Antimicrobial Applications against Multidrug-resistant Bacteria[J]. Biotechnology Bulletin, doi: 10.13560/j.cnki.biotech.bull.1985.2025-0955.
协同策略 Collaborative strategy | 模型/病原体 Model/Pathogen | 关键效能指标 Key performance indicators | 核心优势 Core advantages | 主要局限 Main limitations |
|---|---|---|---|---|
| 物理结合与空间共定位 | 小鼠角膜炎 | 眼部滞留时间最长12 h;角膜透明度和细胞修复显著改善[ | 增强稳定性+靶向黏附 | 规模化制备难度高 |
| 噬菌体导向靶向递送 | 金黄色葡萄球菌生物膜 | 生物膜强度减少95%;联合处理后存活率降至14%[ | 精准定位感染灶 | RBP宿主特异性限制 |
| 光热/酶响应级联激活 | 铜绿假单胞菌感染伤口 | 10 min内目标细菌完全失活;非目标细菌保留[ | 时空可控治疗 | 近红外设备依赖性强 |
| 基因编辑联合清除 | MRSA肺炎模型 | 细菌负荷降至<100 CFU/mL;炎症指标接近正常[ | 逆转耐药性+协同杀菌 | CRISPR脱靶风险 |
| 仿生纳米涂层免疫屏蔽 | 沙门氏菌肠炎模型 | 细胞内噬菌体裂解效率更高,清除生物膜并改善炎症[ | 突破胃肠屏障+免疫逃逸 | 涂层材料降解可控性待优化 |
| 微生态重塑 | DSS结肠炎小鼠 | 细胞存活率由40%升至85%;炎症因子下调,益生因子上调[ | 避免菌群失调+生态修复 | 菌群互作网络调控复杂 |
表1 不同噬菌体-纳米材料协同抗菌策略的代表性抗菌效能与优势对比
Table 1 Representative antibacterial performance and comparative features of different phage-nanomaterial synergistic strategies
协同策略 Collaborative strategy | 模型/病原体 Model/Pathogen | 关键效能指标 Key performance indicators | 核心优势 Core advantages | 主要局限 Main limitations |
|---|---|---|---|---|
| 物理结合与空间共定位 | 小鼠角膜炎 | 眼部滞留时间最长12 h;角膜透明度和细胞修复显著改善[ | 增强稳定性+靶向黏附 | 规模化制备难度高 |
| 噬菌体导向靶向递送 | 金黄色葡萄球菌生物膜 | 生物膜强度减少95%;联合处理后存活率降至14%[ | 精准定位感染灶 | RBP宿主特异性限制 |
| 光热/酶响应级联激活 | 铜绿假单胞菌感染伤口 | 10 min内目标细菌完全失活;非目标细菌保留[ | 时空可控治疗 | 近红外设备依赖性强 |
| 基因编辑联合清除 | MRSA肺炎模型 | 细菌负荷降至<100 CFU/mL;炎症指标接近正常[ | 逆转耐药性+协同杀菌 | CRISPR脱靶风险 |
| 仿生纳米涂层免疫屏蔽 | 沙门氏菌肠炎模型 | 细胞内噬菌体裂解效率更高,清除生物膜并改善炎症[ | 突破胃肠屏障+免疫逃逸 | 涂层材料降解可控性待优化 |
| 微生态重塑 | DSS结肠炎小鼠 | 细胞存活率由40%升至85%;炎症因子下调,益生因子上调[ | 避免菌群失调+生态修复 | 菌群互作网络调控复杂 |
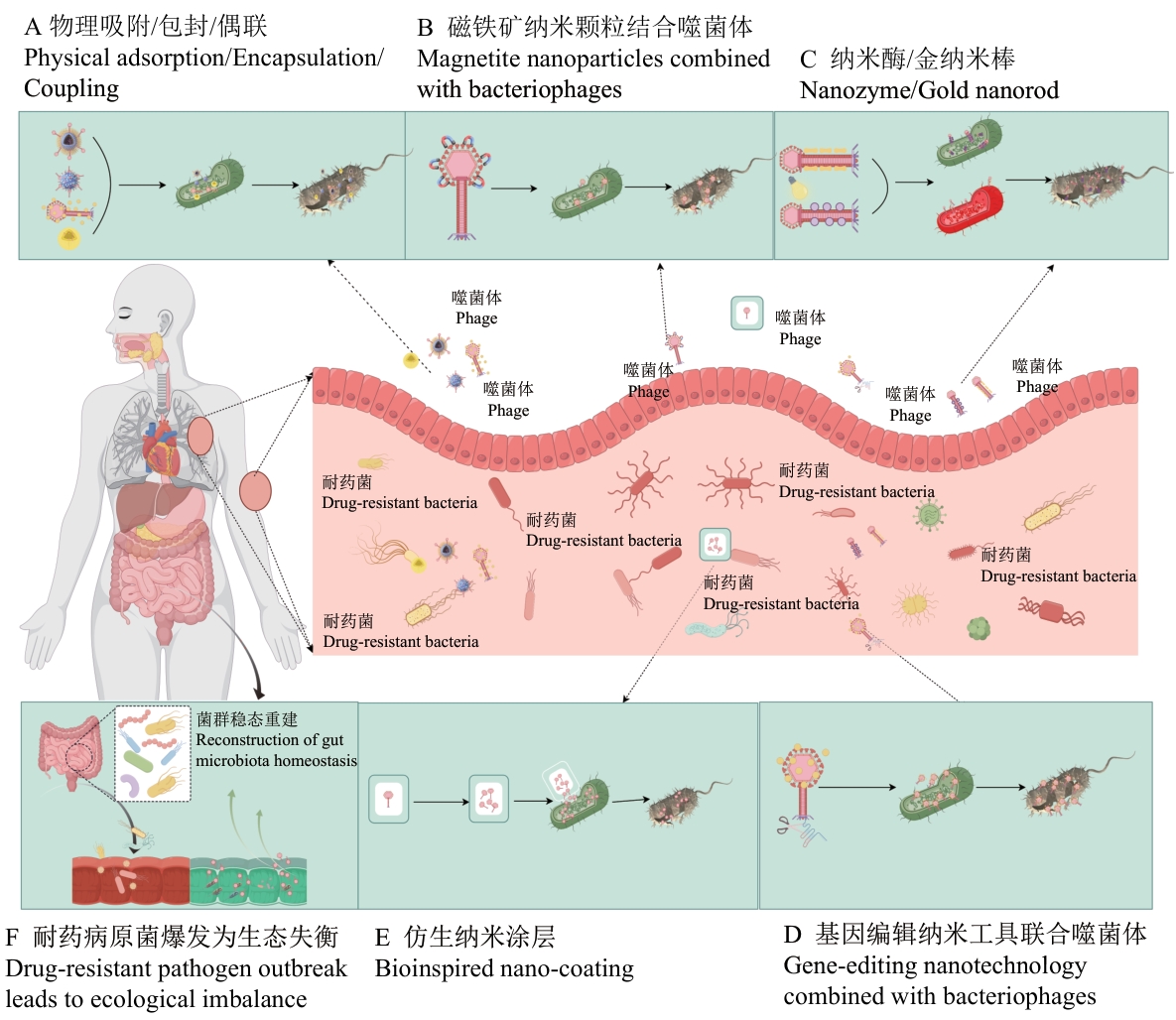
图2 以肺部感染与皮肤创伤口为例,总结噬菌体-纳米材料协同抗耐药菌的整体框架示意图
Fig. 2 Taking pulmonary infection and skin wounds as examples, summarize the overall framework diagram of the bacteriophage-nanomaterial synergistic anti-drug-resistant bacteria
协同策略 Combination strategy | 生物安全性风险 Biosafety risk | 生产可扩展性 Manufacturing scalability | 成本可控性 Cost controllability | 监管复杂度 Regulatory complexity | 近期临床可行性 Short-term clinical feasibility |
|---|---|---|---|---|---|
| 物理结合与空间共定位 | 低-中 | 中-高 | 中 | 低-中 | 较高 |
| 噬菌体导向靶向递送 | 中 | 中 | 中 | 中 | 中-较高 |
| 光热/酶响应级联激活 | 中 | 中 | 中-低 | 中-高 | 中 |
| 基因编辑联合清除 | 高 | 低 | 低 | 高 | 较低 |
| 仿生纳米涂层免疫屏蔽 | 低-中 | 中 | 中 | 中 | 较高 |
| 微生态重塑 | 中 | 中-低 | 中 | 中-高 | 中 |
表2 不同噬菌体-纳米材料协同策略的临床转化可行性对比
Table 2 Comparative translational feasibility of different phage-nanomaterial synergistic strategies
协同策略 Combination strategy | 生物安全性风险 Biosafety risk | 生产可扩展性 Manufacturing scalability | 成本可控性 Cost controllability | 监管复杂度 Regulatory complexity | 近期临床可行性 Short-term clinical feasibility |
|---|---|---|---|---|---|
| 物理结合与空间共定位 | 低-中 | 中-高 | 中 | 低-中 | 较高 |
| 噬菌体导向靶向递送 | 中 | 中 | 中 | 中 | 中-较高 |
| 光热/酶响应级联激活 | 中 | 中 | 中-低 | 中-高 | 中 |
| 基因编辑联合清除 | 高 | 低 | 低 | 高 | 较低 |
| 仿生纳米涂层免疫屏蔽 | 低-中 | 中 | 中 | 中 | 较高 |
| 微生态重塑 | 中 | 中-低 | 中 | 中-高 | 中 |
| [1] | Mendelson M, Laxminarayan R, Limmathurotsakul D, et al. Antimicrobial resistance and the great divide: inequity in priorities and agendas between the Global North and the Global South threatens global mitigation of antimicrobial resistance [J]. Lancet Glob Health, 2024, 12(3): e516-e521. |
| [2] | Salmond GPC, Fineran PC. A century of the phage: past, present and future [J]. Nat Rev Microbiol, 2015, 13(12): 777-786. |
| [3] | Naghavi M, Vollset SE, Ikuta KS, et al. Global burden of bacterial antimicrobial resistance 1990-2021: a systematic analysis with forecasts to 2050 [J]. Lancet, 2024, 404(10459): 1199-1226. |
| [4] | Hatfull GF, Dedrick RM, Schooley RT. Phage therapy for antibiotic-resistant bacterial infections [J]. Annu Rev Med, 2022, 73: 197-211. |
| [5] | Hamdi MF, Alsaedi AA, Hayder AQ, et al. Invasive bacteriophages between a bell and a hammer: a comprehensive review of pharmacokinetics and bacterial defense systems [J]. Discov Life, 2025, 55: 10. |
| [6] | Zhang S, Ahn J. Phage therapy as a novel alternative to antibiotics through adaptive evolution and fitness trade-offs [J]. Antibiotics, 2025, 14(10): 1040. |
| [7] | Qi ML, Tay A. Phage-nanomaterial platforms for precision antimicrobial therapy: from design to therapeutic application [J]. Nanoscale, 2025, 17(37): 21423-21457. |
| [8] | Hameed S, Sharif S, Ovais M, et al. Emerging trends and future challenges of advanced 2D nanomaterials for combating bacterial resistance [J]. Bioact Mater, 2024, 38: 225-257. |
| [9] | Xie MM, Gao M, Yun Y, et al. Antibacterial nanomaterials: mechanisms, impacts on antimicrobial resistance and design principles [J]. Angew Chem Int Ed, 2023, 62(17): e202217345. |
| [10] | Pangprasit N, Kongkaew A, Saipinta D, et al. Evaluation of antibacterial properties of zinc oxide nanoparticles against bacteria isolated from animal wounds [J]. Pharmaceutics, 2025, 17(2): 209. |
| [11] | Praharsha A, Gupta A, Bhardwaj K, et al. Silver nanoparticle-coated Bacteriophages: a novel therapeutic approach for combating Enterococcus faecalis in endodontic infections [J]. J Oral Biol Craniofacial Res, 2025, 15(5): 1051-1056. |
| [12] | Wan HP, Zhong XY, Yang SN, et al. Enhancing the therapeutic potential of peptide antibiotics using bacteriophage mimicry strategies [J]. Adv Sci, 2025, 12(3): 2411753. |
| [13] | Roach DR, Donovan DM. Antimicrobial bacteriophage-derived proteins and therapeutic applications [J]. Bacteriophage, 2015, 5(3): e1062590. |
| [14] | Shi ZX, Hong X, Li ZX, et al. Characterization of the novel broad-spectrum lytic phage Phage_Pae01 and its antibiofilm efficacy against Pseudomonas aeruginosa [J]. Front Microbiol, 2024, 15: 1386830. |
| [15] | Köhler T, Luscher A, Falconnet L, et al. Personalized aerosolised bacteriophage treatment of a chronic lung infection due to multidrug-resistant Pseudomonas aeruginosa [J]. Nat Commun, 2023, 14: 3629. |
| [16] | Cadamuro RD, Elois MA, Von Tönnemann Pilati G, et al. Role of lysogenic phages in the dissemination of antibiotic resistance genes applied in the food chain [J]. Foods, 2025, 14(7): 1082. |
| [17] | Le S, Wei LL, Wang J, et al. Bacteriophage protein Dap1 regulates evasion of antiphage immunity and Pseudomonas aeruginosa virulence impacting phage therapy in mice [J]. Nat Microbiol, 2024, 9(7): 1828-1841. |
| [18] | Martins WMBS, Li M, Sands K, et al. Effective phage cocktail to combat the rising incidence of extensively drug-resistant Klebsiella pneumoniae sequence type 16 [J]. Emerg Microbes Infect, 2022, 11(1): 1015-1023. |
| [19] | Mu YQ, Song YQ, Tian XR, et al. Leveraging collateral sensitivity to counteract the evolution of bacteriophage resistance in bacteria [J]. mLife, 2025, 4(2): 143-154. |
| [20] | Xu YJ, Yang T, Miao Y, et al. Injectable phage-loaded microparticles effectively release phages to kill methicillin-resistant Staphylococcus aureus [J]. ACS Appl Mater Interfaces, 2024, 16(14): 17232-17241. |
| [21] | Zhang HL, Zhu R, Wang ZF, et al. Programming virulent bacteriophages by developing a multiplex genome engineering method [J]. mBio, 2025, 16(6): e03582-e03524. |
| [22] | Vikal A, Maurya R, Patel P, et al. Nano revolution: harnessing nanoparticles to combat antibiotic-resistant bacterial infections [J]. Curr Pharm Des, 2025, 31(7): 498-506. |
| [23] | Tu YS, Lv M, Xiu P, et al. Destructive extraction of phospholipids from Escherichia coli membranes by graphene nanosheets [J]. Nature Nanotech, 2013, 8(8): 594-601. |
| [24] | Ali A, Ali SR, Hussain R, et al. Comparative study of silica and silica-decorated ZnO and ag nanocomposites for antimicrobial and photocatalytic applications [J]. Sci Rep, 2025, 15: 5010. |
| [25] | Pokhrel LR, Jacobs ZL, Dikin D, et al. Five nanometer size highly positive silver nanoparticles are bactericidal targeting cell wall and adherent fimbriae expression [J]. Sci Rep, 2022, 12: 6729. |
| [26] | Zhang LP, Yang YT, Wang JX, et al. A portable, sprayable, highly malleable, elastic, and hydrophobic antibacterial fibrous wound dressing for infected wound healing [J]. Adv Fiber Mater, 2025, 7(2): 528-540. |
| [27] | de Carvalho Lima EN, Octaviano ALM, Piqueira JRC, et al. Coronavirus and carbon nanotubes: seeking immunological relationships to discover immunotherapeutic possibilities [J]. Int J Nanomed, 2022, 17: 751-781. |
| [28] | Liu ZW, Zhao XY, Yu BR, et al. Rough carbon-iron oxide nanohybrids for near-infrared-II light-responsive synergistic antibacterial therapy [J]. ACS Nano, 2021, 15(4): 7482-7490. |
| [29] | Meng L, Yang FM, Cao ZP, et al. Wearing bacteriophages individually with an adhesive drug-loadable nanohelmet for treating ocular infections [J]. Sci Adv, 2025, 11(28): eadx4183. |
| [30] | Park J, Hassan MA, Nabawy A, et al. Engineered bacteriophage-polymer nanoassemblies for treatment of wound biofilm infections [J]. ACS Nano, 2024, 18(39): 26928-26936. |
| [31] | Olszewska-Widdrat A, Bennet M, Mickoleit F, et al. Bacteriophage-templated assembly of magnetic nanoparticles and their actuation potential [J]. ChemNanoMat, 2021, 7(8): 942-949. |
| [32] | Manoharadas S, Altaf M, Alrefaei AF, et al. Concerted dispersion of Staphylococcus aureus biofilm by bacteriophage and ‘green synthesized’ silver nanoparticles [J]. RSC Adv, 2021, 11(3): 1420-1429. |
| [33] | Jin LL, Cao FF, Gao Y, et al. Microenvironment-activated nanozyme-armed bacteriophages efficiently combat bacterial infection [J]. Adv Mater, 2023, 35(30): 2301349. |
| [34] | Peng H, Borg RE, Dow LP, et al. Controlled phage therapy by photothermal ablation of specific bacterial species using gold nanorods targeted by chimeric phages [J]. Proc Natl Acad Sci U S A, 2020, 117(4): 1951-1961. |
| [35] | Peng H, Rossetto D, Mansy SS, et al. Treatment of wound infections in a mouse model using Zn2+-releasing phage bound to gold nanorods [J]. ACS Nano, 2022, 16(3): 4756-4774. |
| [36] | Park JY, Moon BY, Park JW, et al. Genetic engineering of a temperate phage-based delivery system for CRISPR/Cas9 antimicrobials against Staphylococcus aureus [J]. Sci Rep, 2017, 7: 44929. |
| [37] | Li XS, Gui SM, Gui R, et al. Multifunctional clustered regularly interspaced short palindromic repeats (CRISPR)-Cas9-based nanobomb against carbapenem-resistant Acinetobacter baumannii infection through cascade reaction and amplification synergistic effect [J]. ACS Nano, 2023, 17(24): 24632-24653. |
| [38] | Lin SS, Xie GC, He J, et al. Enhancing phage therapy by coating single bacteriophage-infected bacteria with polymer to preserve phage vitality [J]. Nat Biomed Eng, 2025, 9(7): 1155-1171. |
| [39] | Hsu BB, Gibson TE, Yeliseyev V, et al. Dynamic modulation of the gut microbiota and metabolome by bacteriophages in a mouse model [J]. Cell Host Microbe, 2019, 25(6): 803-814.e5. |
| [40] | Liang XL, Yang S, Radosevich M, et al. Bacteriophage-driven microbial phenotypic heterogeneity: ecological and biogeochemical importance [J]. NPJ Biofilms Microbiomes, 2025, 11: 82. |
| [41] | Yadegar A, Pakpour S, Ibrahim FF, et al. Beneficial effects of fecal microbiota transplantation in recurrent Clostridioides difficile infection [J]. Cell Host Microbe, 2023, 31(5): 695-711. |
| [42] | Wdowiak M, Raza S, Grotek M, et al. Phage/nanoparticle cocktails for a biocompatible and environmentally friendly antibacterial therapy [J]. Appl Microbiol Biotechnol, 2025, 109: 129. |
| [43] | Hu QL, Li JY, Wang T, et al. Polyphenolic nanoparticle-modified probiotics for microenvironment remodeling and targeted therapy of inflammatory bowel disease [J]. ACS Nano, 2024, 18(20): 12917-12932. |
| [44] | Wang PW, Lee WT, Wu YN, et al. Opportunities for nanomedicine in Clostridioides difficile infection [J]. Antibiotics, 2021, 10(8): 948. |
| [45] | Berkson JD, Wate CE, Allen GB, et al. Phage-specific immunity impairs efficacy of bacteriophage targeting Vancomycin Resistant Enterococcus in a murine model [J]. Nat Commun, 2024, 15: 2993. |
| [46] | Aranaga C, Pantoja LD, Martínez EA, et al. Phage therapy in the era of multidrug resistance in bacteria: a systematic review [J]. Int J Mol Sci, 2022, 23(9): 4577. |
| [47] | Bao J, Wu NN, Zeng YG, et al. Non-active antibiotic and bacteriophage synergism to successfully treat recurrent urinary tract infection caused by extensively drug-resistant Klebsiella pneumoniae [J]. Emerg Microbes Infect, 2020, 9(1): 771-774. |
| [48] | Jault P, Leclerc T, Jennes S, et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): a randomised, controlled, double-blind phase 1/2 trial [J]. Lancet Infect Dis, 2019, 19(1): 35-45. |
| [49] | Qin JH, Wu NN, Bao J, et al. Heterogeneous Klebsiella pneumoniae co-infections complicate personalized bacteriophage therapy [J]. Front Cell Infect Microbiol, 2021, 10: 608402. |
| [50] | Schooley RT, Biswas B, Gill JJ, et al. Development and use of personalized bacteriophage-based therapeutic cocktails to treat a patient with a disseminated resistant Acinetobacter baumannii infection [J]. Antimicrob Agents Chemother, 2017, 61(10): e00954-e00917. |
| [51] | Tan X, Chen HS, Zhang M, et al. Clinical experience of personalized phage therapy against carbapenem-resistant Acinetobacter baumannii lung infection in a patient with chronic obstructive pulmonary disease [J]. Front Cell Infect Microbiol, 2021, 11: 631585. |
| [52] | Wu NN, Dai J, Guo MQ, et al. Pre-optimized phage therapy on secondary Acinetobacter baumannii infection in four critical COVID-19 patients [J]. Emerg Microbes Infect, 2021, 10(1): 612-618. |
| [53] | Dedrick RM, Freeman KG, Nguyen JA, et al. Potent antibody-mediated neutralization limits bacteriophage treatment of a pulmonary Mycobacterium abscessus infection [J]. Nat Med, 2021, 27(8): 1357-1361. |
| [54] | Jia HJ, Jia PP, Yin SP, et al. Engineering bacteriophages for enhanced host range and efficacy: insights from bacteriophage-bacteria interactions [J]. Front Microbiol, 2023, 14: 1172635. |
| [55] | Herai RH. Avoiding the off-target effects of CRISPR/Cas9 system is still a challenging accomplishment for genetic transformation [J]. Gene, 2019, 700: 176-178. |
| [56] | Zheng LG, Tan Y, Hu YC, et al. CRISPR/Cas-based genome editing for human gut commensal Bacteroides species [J]. ACS Synth Biol, 2022, 11(1): 464-472. |
| [57] | Javaid N, Choi S. CRISPR/Cas system and factors affecting its precision and efficiency [J]. Front Cell Dev Biol, 2021, 9: 761709. |
| [58] | Praphawatvet T, Peters JI, Williams RO. Inhaled nanoparticles-An updated review [J]. Int J Pharm, 2020, 587: 119671. |
| [59] | Elsayed MM, Elkenany RM, EL-Khateeb AY, et al. Isolation and encapsulation of bacteriophage with chitosan nanoparticles for biocontrol of multidrug-resistant methicillin-resistant Staphylococcus aureus isolated from broiler poultry farms [J]. Sci Rep, 2024, 14: 4702. |
| [60] | Kaur S, Kumari A, Kumari Negi A, et al. Nanotechnology based approaches in phage therapy: overcoming the pharmacological barriers [J]. Front Pharmacol, 2021, 12: 699054. |
| [61] | Tran TK, Nguyen MK, Lin C, et al. Review on fate, transport, toxicity and health risk of nanoparticles in natural ecosystems: Emerging challenges in the modern age and solutions toward a sustainable environment [J]. Sci Total Environ, 2024, 912: 169331. |
| [62] | Elrys AS, Wen YH, Feng D, et al. Cadmium inhibits carbon and nitrogen cycling through soil microbial biomass and reduces soil nitrogen availability [J]. J Hazard Mater, 2025, 489: 137524. |
| [63] | Wiebe KG, Cook BWM, Lightly TJ, et al. Investigation into scalable and efficient enterotoxigenic Escherichia coli bacteriophage production [J]. Sci Rep, 2024, 14: 3618. |
| [64] | García R, Latz S, Romero J, et al. Bacteriophage production models: an overview [J]. Front Microbiol, 2019, 10: 1187. |
| [65] | 杨蓉, 赵晨, 李端华, 等. 噬菌体药物开发及质控研究进展 [J]. 国外医药: 抗生素分册, 2024, 45(1): 28-34. |
| Yang R, Zhao C, Li DH, et al. Progress in development and quality control of bacteriophage drugs [J]. World Notes Antibiot, 2024, 45(1): 28-34. | |
| [66] | Serrano DR, Kara A, Yuste I, et al. 3D printing technologies in personalized medicine, nanomedicines, and biopharmaceuticals [J]. Pharmaceutics, 2023, 15(2): 313. |
| [67] | Zhang FM, Kamm MA, Wu X, et al. Preferred reporting items for microbiotherapy (PRIM) guidelines across medical disciplines: an international Delphi consensus [J]. J Gastro And Hepatol, 2025, 40(6): 1364-1373. |
| [68] | Cieślik M, Wójcicki M, Migdał P, et al. Fighting biofilm: bacteriophages eliminate biofilm formed by multidrug-resistant Enterobacter hormaechei on urological catheters [J]. Med Microbiol Immunol, 2025, 214: 33. |
| [69] | Omura SN, Alfonse LE, Ornstein A, et al. Structural basis for target DNA cleavage and guide RNA processing by CRISPR-Casλ2 [J]. Commun Biol, 2025, 8: 876. |
| [70] | Levrier A, Karpathakis I, Nash B, et al. PHEIGES: all-cell-free phage synthesis and selection from engineered genomes [J]. Nat Commun, 2024, 15: 2223. |
| [71] | Nethery MA, Hidalgo-Cantabrana C, Roberts A, et al. CRISPR-based engineering of phages for in situ bacterial base editing [J]. Proc Natl Acad Sci U S A, 2022, 119(46): e2206744119. |
| [1] | 武志健, 刘广洋, 林志豪, 盛彬, 陈鸽, 许晓敏, 王军伟, 徐东辉. 蔬菜种子萌发的纳米调控及其机制研究进展[J]. 生物技术通报, 2025, 41(1): 14-24. |
| [2] | 王梦雅, 刘家齐, 姜海霖, 李菁华, 赵春燕, 黄红兰. 肠侵袭性大肠埃希菌噬菌体DK-13的生物学特性及应用[J]. 生物技术通报, 2024, 40(3): 296-304. |
| [3] | 高聪, 萧楚健, 鲁帅, 王苏蓉, 袁卉华, 曹云英. 氧化石墨烯对拟南芥生长的促进作用[J]. 生物技术通报, 2022, 38(6): 120-128. |
| [4] | 张玮玮, 杨慧霞, 薛屏. 纳米载体固定化脂肪酶及其在生物柴油转化中的应用进展[J]. 生物技术通报, 2020, 36(1): 160-166. |
| [5] | 高梦迪, 盛茂银, 傅籍锋. 纳米材料对植物生长发育的影响[J]. 生物技术通报, 2019, 35(7): 172-180. |
| [6] | 耿慧君, 邹伟, 崔惠敬, 李晓宇, 王丽丽, 徐永平. 基于转录组学的金黄色葡萄球菌噬菌体安全性评估[J]. 生物技术通报, 2019, 35(12): 64-75. |
| [7] | 李宸葳, 杜再慧, 林少华, 罗云波, 许文涛. Pb2+功能核酸生物传感器的研究进展[J]. 生物技术通报, 2019, 35(1): 131-139. |
| [8] | 李舒婷, 周子琦, 田杰生, 许文涛. 磁小体介导的生物传感器研究进展[J]. 生物技术通报, 2018, 34(9): 70-78. |
| [9] | 郑力榕, 罗云波, 许文涛. 光及光热纳米材料在生物传感、药物靶向运输和生物成像中的应用[J]. 生物技术通报, 2018, 34(9): 79-89. |
| [10] | 李舒婷, 贺万崇, 黄昆仑, 许文涛. 介孔二氧化硅介导的功能核酸检测技术研究进展[J]. 生物技术通报, 2018, 34(9): 139-148. |
| [11] | 彭立, 张兴梅. 核酸适配子结合无机纳米材料在肿瘤研究中的应用[J]. 生物技术通报, 2017, 33(11): 48-53. |
| [12] | 高启禹, 徐光翠, 陈红丽, 周晨妍. 纳米材料固定化酶的研究进展[J]. 生物技术通报, 2013, 0(6): 20-24. |
| [13] | 高启禹, 李宏彬, 陈红丽, 孔雨, 周晨妍. 壳聚糖修饰的PLGA纳米颗粒固定化碱性磷酸单酯酶的技术研究[J]. 生物技术通报, 2013, 0(5): 199-204. |
| [14] | 顿文涛, 李勉, 李燕, 李聪, 赵仲麟,袁超. 纳米生物传感器在生物医药中的应用[J]. 生物技术通报, 2013, 0(4): 49-54. |
| [15] | 卢艳敏. 纳米基因载体研究进展[J]. 生物技术通报, 2013, 0(2): 61-66. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||