








生物技术通报 ›› 2025, Vol. 41 ›› Issue (1): 14-24.doi: 10.13560/j.cnki.biotech.bull.1985.2024-0470
武志健1,2( ), 刘广洋2,3, 林志豪2, 盛彬2,4, 陈鸽2, 许晓敏2, 王军伟1(
), 刘广洋2,3, 林志豪2, 盛彬2,4, 陈鸽2, 许晓敏2, 王军伟1( ), 徐东辉2,3(
), 徐东辉2,3( )
)
收稿日期:2024-05-20
出版日期:2025-01-26
发布日期:2025-01-22
通讯作者:
王军伟,男,博士,副教授,研究方向:蔬菜学;E-mail: JunweiWang87@126.com;作者简介:武志健,男,硕士研究生,研究方向:蔬菜学;E-mail: 3463625130@qq.com
基金资助:
WU Zhi-jian1,2( ), LIU Guang-yang2,3, LIN Zhi-hao2, SHENG Bin2,4, CHEN Ge2, XU Xiao-min2, WANG Jun-wei1(
), LIU Guang-yang2,3, LIN Zhi-hao2, SHENG Bin2,4, CHEN Ge2, XU Xiao-min2, WANG Jun-wei1( ), XU Dong-hui2,3(
), XU Dong-hui2,3( )
)
Received:2024-05-20
Published:2025-01-26
Online:2025-01-22
摘要:
提高种子在非生物胁迫下的萌发率可降低环境恶化对蔬菜安全构成的风险,为全球蔬菜产量提供保障。由于纳米材料具有尺寸小和独特的物理化学性质,在蔬菜生产中可应用于种子引发。纳米引发在改善非生物胁迫下蔬菜种子的萌发方面显示了突出作用。论文将用于调控蔬菜种子萌发的纳米材料分为碳基、硅基、金属颗粒和金属氧化物四类,并列举了部分纳米材料促进蔬菜种子萌发的适宜浓度。描述了不同种类纳米材料常用的合成方法及其影响因素,并比较了传统合成与绿色合成的利弊。重点综述了纳米引发对蔬菜种子和幼苗生理生化指标的影响并归纳为两种调控途径。纳米材料调控种子对水分和养分的吸收以及赤霉素合成等与萌发相关过程,从而促进萌发称为直接调控。间接调控是纳米材料产生活性氧,通过信号传导激活抗氧化系统,提高种子对非生物胁迫的抵抗力从而促进萌发。最后,阐述了纳米材料在种子中的应用,并对纳米引发未来的研究方向进行展望:(1)着重考虑纳米材料在长期条件下对环境的潜在风险,避免经过食物链对人类健康造成不利影响;(2)评估纳米材料在多种非生物胁迫下调节活性氧、保障种子萌发的性能;(3)探索由纳米材料引发产生的活性氧通过信号传导激活防御途径的整体通路;(4)补充纳米材料进入种子的准确机制。
武志健, 刘广洋, 林志豪, 盛彬, 陈鸽, 许晓敏, 王军伟, 徐东辉. 蔬菜种子萌发的纳米调控及其机制研究进展[J]. 生物技术通报, 2025, 41(1): 14-24.
WU Zhi-jian, LIU Guang-yang, LIN Zhi-hao, SHENG Bin, CHEN Ge, XU Xiao-min, WANG Jun-wei, XU Dong-hui. Research Progress of Nano-regulation of Vegetable Seed Germination and Its Mechanism[J]. Biotechnology Bulletin, 2025, 41(1): 14-24.
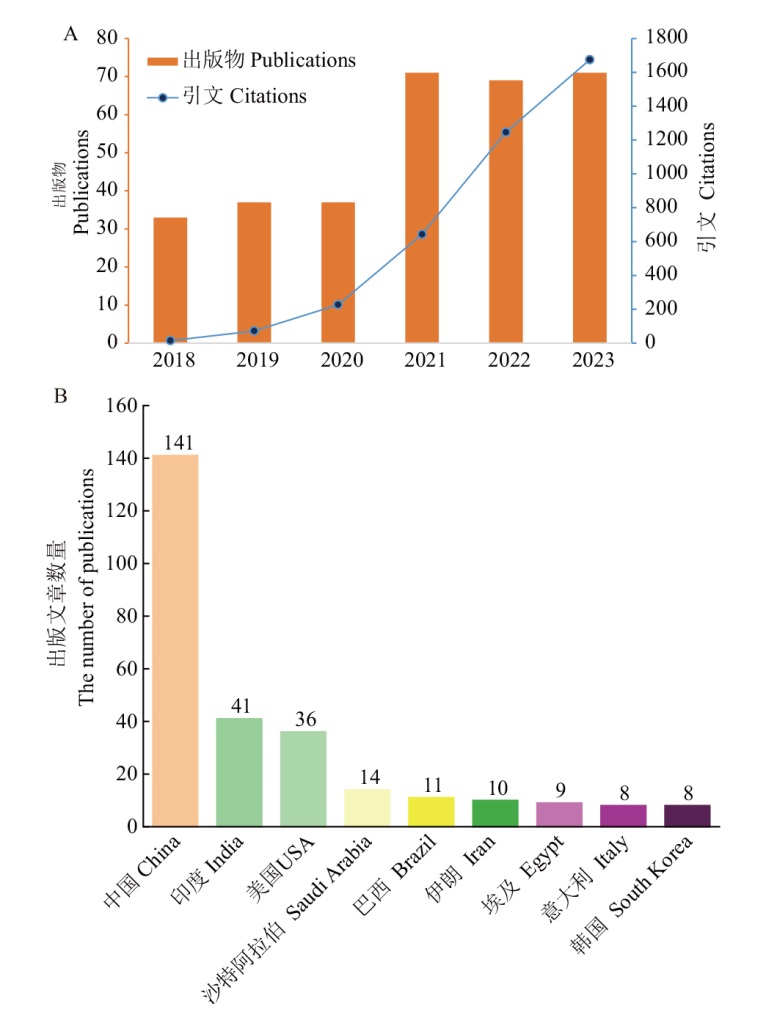
图1 近6年纳米材料在蔬菜研究上的出版物数量趋势、引文数量趋势和各国发表文章数量分布 A:出版物数量趋势和引文数量趋势;B:各国发表文章数量分布;图中数据来源于Web of Science
Fig. 1 Trend in the number of publications, frequency of citations and distribution of the number of publications in various countries for nanomaterials on vegetables over the last 6 years A: The trend in the number of publications and citations. B: The distribution of the number of publications in various countries. The data in the figure is from Web of Science
| 类别 Type | 纳米材料 Nanomaterial | 蔬菜 Vegetable | 浓度 Concentration/(mg·L-1) | 参考文献 Reference |
|---|---|---|---|---|
| 碳基Carbon-based | 碳纳米管 Carbon nanotube | 番茄、洋葱 Solanum lycopersicum, Allium cepa | 10 | [ |
| 石墨烯 Graphene | 萝卜 Raphanus sativus | 40 | [ | |
| 硅基 Silicon-based | 二氧化硅SiO2 | 番茄 S. lycopersicum | 8 000 | [ |
| 大豆 Glycine max | 500 | [ | ||
| 金属颗粒 Metallic particles | 金Au | 洋葱 A. cepa | 5.4 | [ |
| 银Ag | 苋菜 Amaranthus caudatus | 150 | [ | |
| 金属氧化物 Metal oxides | 氧化锌ZnO | 洋葱 A. cepa | 800 | [ |
| 辣椒 Capsicum annuum | 10 | [ | ||
| 二氧化钛TiO2 | 番茄 S. lycopersicum | 100 | [ | |
| 豌豆 Lathyrus sativus | 20 | [ | ||
| 氧化铁FeO | 菠菜 Spinacia oleracea | 5 | [ | |
| 生菜 Lactuca sativa | 1 | [ |
表1 促进蔬菜种子萌发的纳米材料
Table 1 Nanomaterials for promoting the germination of vegetable seeds
| 类别 Type | 纳米材料 Nanomaterial | 蔬菜 Vegetable | 浓度 Concentration/(mg·L-1) | 参考文献 Reference |
|---|---|---|---|---|
| 碳基Carbon-based | 碳纳米管 Carbon nanotube | 番茄、洋葱 Solanum lycopersicum, Allium cepa | 10 | [ |
| 石墨烯 Graphene | 萝卜 Raphanus sativus | 40 | [ | |
| 硅基 Silicon-based | 二氧化硅SiO2 | 番茄 S. lycopersicum | 8 000 | [ |
| 大豆 Glycine max | 500 | [ | ||
| 金属颗粒 Metallic particles | 金Au | 洋葱 A. cepa | 5.4 | [ |
| 银Ag | 苋菜 Amaranthus caudatus | 150 | [ | |
| 金属氧化物 Metal oxides | 氧化锌ZnO | 洋葱 A. cepa | 800 | [ |
| 辣椒 Capsicum annuum | 10 | [ | ||
| 二氧化钛TiO2 | 番茄 S. lycopersicum | 100 | [ | |
| 豌豆 Lathyrus sativus | 20 | [ | ||
| 氧化铁FeO | 菠菜 Spinacia oleracea | 5 | [ | |
| 生菜 Lactuca sativa | 1 | [ |
| 合成方法 Synthesis methods | 纳米材料 Nanomaterials | 优点 Advantages | 缺点 Disadvantages | 影响因素 Affecting parameter |
|---|---|---|---|---|
| 化学沉淀合成法Chemical precipitation synthesis method | 氧化铁FeO[ 氧化锌ZnO[ | 成本低、规模大、低温、节能 Low cost, large scale, low temperature, and energy saving | 聚集、产物粒径控制难、耗时长 Aggregation, product size not easy to control, and time-consuming | 温度、pH、溶剂类型、反应物与溶剂的混合比例 Temperature, pH, solvent type, and mixing rate of solvent and reagent |
| 溶胶-凝胶合成法 Sol-gel synthesis method | 二氧化硅SiO2[ 银-二氧化钛Ag-TiO2[ | 粒径分布窄、精确控制尺寸和形貌、纯度高 Narrow size distribution, precise size and morphology control, and high purity | 成本高、耗时长、规模小、溶剂可能有害 High cost, time-consuming, small scale, and solvents may be harmful | pH、搅拌时间、合成温度/时间、溶剂量 pH, stirring time, synthesis temperature/time, and solvent amount |
| 水热合成法Hydrothermal synthesis method | 碳点CD[ 氧化铜CuO[ | 结晶度高、精确控制尺寸和形貌 High crystallinity, precise control of size and shape | 成本高、无法观察反应过程、可靠性低 High cost, impossible to observe the reaction process, and low reliability | 反应温度/时间、有机添加剂Reaction temperature/time, and organic additive |
| 乳液合成法Emulsion synthesis method | 二氧化硅 SiO2[ 氧化锆 ZrO2[ | 粒径分布窄、易制备、团聚最少 Narrow size distribution, easy preparation, and minimal agglomeration | 成本高、耗时长、需要大量的表面活性剂 High cost, time-consuming, requires a large amount of surfactant | 搅拌速度/持续时间、表面活性剂的类型 Stirring speed/duration, and type of surfactant |
表2 调控萌发的纳米材料的合成方法及其特点
Table 2 Features of synthesis methods of nanomaterials for regulating germination
| 合成方法 Synthesis methods | 纳米材料 Nanomaterials | 优点 Advantages | 缺点 Disadvantages | 影响因素 Affecting parameter |
|---|---|---|---|---|
| 化学沉淀合成法Chemical precipitation synthesis method | 氧化铁FeO[ 氧化锌ZnO[ | 成本低、规模大、低温、节能 Low cost, large scale, low temperature, and energy saving | 聚集、产物粒径控制难、耗时长 Aggregation, product size not easy to control, and time-consuming | 温度、pH、溶剂类型、反应物与溶剂的混合比例 Temperature, pH, solvent type, and mixing rate of solvent and reagent |
| 溶胶-凝胶合成法 Sol-gel synthesis method | 二氧化硅SiO2[ 银-二氧化钛Ag-TiO2[ | 粒径分布窄、精确控制尺寸和形貌、纯度高 Narrow size distribution, precise size and morphology control, and high purity | 成本高、耗时长、规模小、溶剂可能有害 High cost, time-consuming, small scale, and solvents may be harmful | pH、搅拌时间、合成温度/时间、溶剂量 pH, stirring time, synthesis temperature/time, and solvent amount |
| 水热合成法Hydrothermal synthesis method | 碳点CD[ 氧化铜CuO[ | 结晶度高、精确控制尺寸和形貌 High crystallinity, precise control of size and shape | 成本高、无法观察反应过程、可靠性低 High cost, impossible to observe the reaction process, and low reliability | 反应温度/时间、有机添加剂Reaction temperature/time, and organic additive |
| 乳液合成法Emulsion synthesis method | 二氧化硅 SiO2[ 氧化锆 ZrO2[ | 粒径分布窄、易制备、团聚最少 Narrow size distribution, easy preparation, and minimal agglomeration | 成本高、耗时长、需要大量的表面活性剂 High cost, time-consuming, requires a large amount of surfactant | 搅拌速度/持续时间、表面活性剂的类型 Stirring speed/duration, and type of surfactant |
| [1] | Yao JQ, Chen YN, Zhao Y, et al. Climatic and associated atmospheric water cycle changes over the Xinjiang, China[J]. J Hydrol, 2020, 585: 124823. |
| [2] |
Sharma JK, Sihmar M, Santal AR, et al. Impact assessment of major abiotic stresses on the proteome profiling of some important crop plants: a current update[J]. Biotechnol Genet Eng Rev, 2019, 35(2): 126-160.
doi: 10.1080/02648725.2019.1657682 pmid: 31478455 |
| [3] | Ahire SA, Bachhav AA, Pawar TB, et al. The Augmentation of nanotechnology era: a concise review on fundamental concepts of nanotechnology and applications in material science and technology[J]. Results Chem, 2022, 4: 100633. |
| [4] | Singh PM, Tiwari A, Maity D, et al. Recent progress of nanomaterials in sustainable agricultural applications[J]. J Mater Sci, 2022, 57(24): 10836-10862. |
| [5] | Acharya P, Jayaprakasha GK, Crosby KM, et al. Green-synthesized nanoparticles enhanced seedling growth, yield, and quality of onion(Allium cepa L.)[J]. ACS Sustainable Chem Eng, 2019, 7(17): 14580-14590. |
| [6] |
Siddiqui MH, Al-Whaibi MH. Role of nano-SiO2 in germination of tomato(Lycopersicum esculentum seeds Mill.)[J]. Saudi J Biol Sci, 2014, 21(1): 13-17.
doi: 10.1016/j.sjbs.2013.04.005 pmid: 24596495 |
| [7] | Maynard AD. Don't define nanomaterials[J]. Nature, 2011, 475(7354): 31. |
| [8] |
Dendisová M, Jeništová A, Parchaňská-Kokaislová A, et al. The use of infrared spectroscopic techniques to characterize nanomaterials and nanostructures: a review[J]. Anal Chim Acta, 2018, 1031: 1-14.
doi: S0003-2670(18)30664-0 pmid: 30119727 |
| [9] | Husen A, Siddiqi KS. Carbon and fullerene nanomaterials in plant system[J]. J Nanobiotechnol, 2014, 12(1): 16. |
| [10] | Vanitha C, Kathiravan M, Umarani R, et al. Seed priming with nano silica alleviates drought stress through regulating antioxidant defense system and osmotic adjustment in soybean (Glycine max L.)[J]. Silicon, 2024, 16(5): 2157-2170.. |
| [11] |
Rizwan M, Ali S, Qayyum MF, et al. Effect of metal and metal oxide nanoparticles on growth and physiology of globally important food crops: a critical review[J]. J Hazard Mater, 2017, 322(Pt A): 2-16.
doi: S0304-3894(16)30499-X pmid: 27267650 |
| [12] | Haghighi M, Teixeira da Silva JA. The effect of carbon nanotubes on the seed germination and seedling growth of four vegetable species[J]. J Crop Sci Biotechnol, 2014, 17(4): 201-208. |
| [13] | Li ZW, Qiao J, Zhi CY, et al. Effects of graphene soaking and treatment on radish growth and quality[J]. Trans Chin Soc Agric Eng, 2022, 38(19): 87-93. |
| [14] | Acharya P, Jayaprakasha GK, Crosby K, et al. Gold nanoparticles enhances seed germination, growth and yield of onion(Allium cepa L.)[J]. Hortscience, 2019. 54(9): S151-S152. |
| [15] | Lateef A, Adelere IA, Gueguim-Kana EB, et al. Evaluation of feather hydrolysate-mediated silver nanoparticles as biofertilizers for the enhancement of vegetative growth and nutraceutical properties of vegetables[J]. Nanotechnol Environ Eng, 2024, 9(1): 47-65. |
| [16] | Tymoszuk A, Wojnarowicz J. Zinc oxide and zinc oxide nanoparticles impact on in vitro germination and seedling growth in Allium cepa L[J]. Materials, 2020, 13(12): 2784. |
| [17] | Singh N, Singh MK, Yadav RK, et al. Green synthesis and characterization of nano zinc oxide and comparative study of its impact on germination and metabolic activities of Solanum lycopersicum L. and Capsicum annuum L.[J]. Vegetos, 2024. DOI: https://doi.org/10.1007/s42535-024-00838-y. |
| [18] | Sonawane H, Arya S, Math S, et al. Myco-synthesized silver and titanium oxide nanoparticles as seed priming agents to promote seed germination and seedling growth of Solanum lycopersicum: a comparative study[J]. Int Nano Lett, 2021, 11(4): 371-379. |
| [19] | Hojjat SS. Effects of TiO2 nanoparticles on germination and growth characteristics of grass pea(Lathyrus sativus L.)seed under drought stress[J]. Nanotechnol Russia, 2020, 15(2): 204-211. |
| [20] | Ndaba B, Roopnarain A, Vatsha B, et al. Synthesis, characterization, and evaluation of Artemisia afra-mediated iron nanoparticles as a potential nano-priming agent for seed germination[J]. ACS Agric Sci Technol, 2022, 2(6): 1218-1229. |
| [21] | Malek RNEHRA, Osman MS, Hafiz NA. Characterization and germination performance of lettuce sativa by iron oxide nanoparticles seed priming[J]. IOP Conf Ser: Earth Environ Sci, 2023, 1216(1): 012033. |
| [22] |
Sadat-Shojai M, Khorasani MT, Dinpanah-Khoshdargi E, et al. Synthesis methods for nanosized hydroxyapatite with diverse structures[J]. Acta Biomater, 2013, 9(8): 7591-7621.
doi: 10.1016/j.actbio.2013.04.012 pmid: 23583646 |
| [23] | Moulton MC, Braydich-Stolle LK, Nadagouda MN, et al. Synthesis, characterization and biocompatibility of “green” synthesized silver nanoparticles using tea polyphenols[J]. Nanoscale, 2010, 2(5): 763. |
| [24] | Chahardoli A, Karimi N, Fattahi A. Nigella arvensis leaf extract mediated green synthesis of silver nanoparticles: their characteristic properties and biological efficacy[J]. Adv Powder Technol, 2018, 29(1): 202-210. |
| [25] | de Francisco M, Mira S, Durães L, et al. Zn oxide nanoparticles and fine particles: synthesis, characterization and evaluation of the toxic effect on germination and vigour of Solanum licopersicum L[J]. Agronomy, 2024, 14(5): 980. |
| [26] | Ferrusquía-Jiménez NI, González-Arias B, Rosales A, et al. Elicitation of Bacillus cereus-amazcala(B.c-A)with SiO2 nanoparticles improves its role as a plant growth-promoting bacteria(PGPB)in chili pepper plants[J]. Plants, 2022, 11(24): 3445. |
| [27] | Gordillo-Delgado F, Zuluaga-Acosta J, Restrepo-Guerrero G. Effect of the suspension of Ag-incorporated TiO2 nanoparticles(Ag-TiO2 NPs)on certain growth, physiology and phytotoxicity parameters in spinach seedlings[J]. PLoS One, 2020, 15(12): e0244511. |
| [28] | Wang HB, Zhang ML, Song YX, et al. Carbon dots promote the growth and photosynthesis of mung bean sprouts[J]. Carbon, 2018, 136: 94-102. |
| [29] | Liu ZF, Guo SB, Fang X, et al. Antibacterial and plant growth-promoting properties of novel Fe3O4/Cu/CuO magnetic nanoparticles[J]. RSC Adv, 2022, 12(31): 19856-19867. |
| [30] | Park JY, Hwangbo SA, Bramhe SN, et al. Synthesis of surfactant free SiO2 nanoparticles via emulsion method[C]// 2015 IEEE 15th International Conference on Nanotechnology(IEEE-NANO). Rome, Italy. Piscataway, NJ: IEEE, 2015: 717-720. |
| [31] | 常鹰, 董仕节, 杜宽河, 等. 热喷涂用纳米氧化锆球形团聚颗粒的一步合成法: CN102502818-A[P]. 2012. |
| Chang Y, Dong SJ, Du KH, et al. One-step synthesis of nano zirconia spherical agglomeration particle for thermal spraying by adding yttrium salt and zirconium salt aqueous solution to oil phase liquid emulsion, standing, layering, sintering and forming the particle: CN102502818-A[P]. 2012. | |
| [32] | Lu L, Huang M, Huang YX, et al. Mn3O4 nanozymes boost endogenous antioxidant metabolites in cucumber(Cucumis sativus)plant and enhance resistance to salinity stress[J]. Environ Sci: Nano, 2020, 7(6): 1692-1703. |
| [33] | Aragão VPM, Navarro BV, Passamani LZ, et al. Free amino acids, polyamines, soluble sugars and proteins during seed germination and early seedling growth of Cedrela fissilis Vellozo(Meliaceae), an endangered hardwood species from the Atlantic Forest in Brazil[J]. Theor Exp Plant Physiol, 2015, 27(2): 157-169. |
| [34] |
Khan MN, Li YH, Khan Z, et al. Nanoceria seed priming enhanced salt tolerance in rapeseed through modulating ROS homeostasis and α-amylase activities[J]. J Nanobiotechnol, 2021, 19(1): 276.
doi: 10.1186/s12951-021-01026-9 pmid: 34530815 |
| [35] | El-Badri AM, Batool M, Mohamed IAA, et al. Mitigation of the salinity stress in rapeseed(Brassica napus L.)productivity by exogenous applications of bio-selenium nanoparticles during the early seedling stage[J]. Environ Pollut, 2022, 310: 119815. |
| [36] |
Mahakham W, Sarmah AK, Maensiri S, et al. Nanopriming technology for enhancing germination and starch metabolism of aged rice seeds using phytosynthesized silver nanoparticles[J]. Sci Rep, 2017, 7: 8263.
doi: 10.1038/s41598-017-08669-5 pmid: 28811584 |
| [37] | Ernest V, Shiny PJ, Mukherjee A, et al. Silver nanoparticles: a potential nanocatalyst for the rapid degradation of starch hydrolysis by α-amylase[J]. Carbohydr Res, 2012, 352: 60-64. |
| [38] | Yepes-Molina L, Bárzana G, Carvajal M. Controversial regulation of gene expression and protein transduction of aquaporins under drought and salinity stress[J]. Plants, 2020, 9(12): 1662. |
| [39] | Khodakovskaya MV, de Silva K, Nedosekin DA, et al. Complex genetic, photothermal, and photoacoustic analysis of nanoparticle-plant interactions[J]. Proc Natl Acad Sci U S A, 2011, 108(3): 1028-1033. |
| [40] | Rosental L, Nonogaki H, Fait A. Activation and regulation of primary metabolism during seed germination[J]. Seed Sci Res, 2014, 24(1): 1-15. |
| [41] | Fait A, Nesi AN, Angelovici R, et al. Targeted enhancement of glutamate-to-γ-aminobutyrate conversion in Arabidopsis seeds affects carbon-nitrogen balance and storage reserves in a development-dependent manner[J]. Plant Physiol, 2011, 157(3): 1026-1042. |
| [42] | Acharya P, Jayaprakasha GK, Semper J, et al. 1H nuclear magnetic resonance and liquid chromatography coupled with mass spectrometry-based metabolomics reveal enhancement of growth-promoting metabolites in onion seedlings treated with green-synthesized nanomaterials[J]. J Agric Food Chem, 2020, 68(46): 13206-13220. |
| [43] | Wang XP, Han HY, Liu XQ, et al. Multi-walled carbon nanotubes can enhance root elongation of wheat(Triticum aestivum)plants[J]. J Nanopart Res, 2012, 14(6): 841. |
| [44] | Guha T, Das H, Mukherjee A, et al. Elucidating ROS signaling networks and physiological changes involved in nanoscale zero valent iron primed rice seed germination sensu stricto[J]. Free Radic Biol Med, 2021, 171: 11-25. |
| [45] | Setty J, Samant SB, Yadav MK, et al. Beneficial effects of bio-fabricated selenium nanoparticles as seed nanopriming agent on seed germination in rice(Oryza sativa L.)[J]. Sci Rep, 2023, 13(1): 22349. |
| [46] |
Rodríguez-Gacio MDELC, Matilla-Vázquez MA, Matilla AJ. Seed dormancy and ABA signaling: the breakthrough goes on[J]. Plant Signal Behav, 2009, 4(11): 1035-49.
pmid: 19875942 |
| [47] | Shu K, Zhang HW, Wang SF, et al. ABI4 regulates primary seed dormancy by regulating the biogenesis of abscisic acid and gibberellins in Arabidopsis[J]. PLoS Genet, 2013, 9(6): e1003577. |
| [48] | Mittler R, Zandalinas SI, Fichman Y, et al. Reactive oxygen species signalling in plant stress responses[J]. Nat Rev Mol Cell Biol, 2022, 23: 663-679. |
| [49] |
Mittler R. ROS are good[J]. Trends Plant Sci, 2017, 22(1): 11-19.
doi: S1360-1385(16)30112-1 pmid: 27666517 |
| [50] | Ma XM, Geiser-Lee J, Deng Y, et al. Interactions between engineered nanoparticles(ENPs)and plants: Phytotoxicity, uptake and accumulation[J]. Sci Total Environ, 2010, 408(16): 3053-3061. |
| [51] |
Chapman JM, Muhlemann JK, Gayomba SR, et al. RBOH-dependent ROS synthesis and ROS scavenging by plant specialized metabolites to modulate plant development and stress responses[J]. Chem Res Toxicol, 2019, 32(3): 370-396.
doi: 10.1021/acs.chemrestox.9b00028 pmid: 30781949 |
| [52] | Chen S, Liu HL, Yangzong ZX, et al. Seed priming with reactive oxygen species-generating nanoparticles enhanced maize tolerance to multiple abiotic stresses[J]. Environ Sci Technol, 2023, 57(48): 19932-19941. |
| [53] | Zhou XD, Jia XR, Zhang ZH, et al. AgNPs seed priming accelerated germination speed and altered nutritional profile of Chinese cabbage[J]. Sci Total Environ, 2022, 808: 151896. |
| [54] | Suzuki N, Miller G, Salazar C, et al. Temporal-spatial interaction between reactive oxygen species and abscisic acid regulates rapid systemic acclimation in plants[J]. Plant Cell, 2013, 25(9): 3553-3569. |
| [55] | Weidemüller P, Kholmatov M, Petsalaki E, et al. Transcription factors: bridge between cell signaling and gene regulation[J]. Proteomics, 2021, 21(23/24): e2000034. |
| [56] | Wang CT, Ru JN, Liu YW, et al. The maize WRKY transcription factor ZmWRKY40 confers drought resistance in transgenic Arabidopsis[J]. Int J Mol Sci, 2018, 19(9): 2580. |
| [57] | Zhang GF, Li GD, Xiang Y, et al. The transcription factor ZmMYB-CC10 improves drought tolerance by activating ZmAPX4 expression in maize[J]. Biochem Biophys Res Commun, 2022, 604: 1-7. |
| [58] | Abdel Hamed Abdel Latef A, Abu Alhmad MF, Abdelfattah KE. The possible roles of priming with ZnO nanoparticles in mitigation of salinity stress in lupine(Lupinus termis)plants[J]. J Plant Growth Regul, 2017, 36(1): 60-70. |
| [59] |
Jhansi K, Jayarambabu N, Reddy KP, et al. Biosynthesis of MgO nanoparticles using mushroom extract: effect on peanut(Arachis hypogaea L.)seed germination[J]. 3 Biotech, 2017, 7(4): 263.
doi: 10.1007/s13205-017-0894-3 pmid: 28791210 |
| [60] | Keller AA, Wang HT, Zhou DX, et al. Stability and aggregation of metal oxide nanoparticles in natural aqueous matrices[J]. Environ Sci Technol, 2010, 44(6): 1962-1967. |
| [61] | Singh J, Kumar S, Alok A, et al. The potential of green synthesized zinc oxide nanoparticles as nutrient source for plant growth[J]. J Clean Prod, 2019, 214: 1061-1070. |
| [62] | Lahiani MH, Dervishi E, Ivanov I, et al. Comparative study of plant responses to carbon-based nanomaterials with different morphologies[J]. Nanotechnology, 2016, 27(26): 265102. |
| [63] |
Villagarcia H, Dervishi E, de Silva K, et al. Surface chemistry of carbon nanotubes impacts the growth and expression of water channel protein in tomato plants[J]. Small, 2012, 8(15): 2328-2334.
doi: 10.1002/smll.201102661 pmid: 22514121 |
| [64] | do Espirito Santo Pereira A, Caixeta Oliveira H, Fernandes Fraceto L, et al. Nanotechnology potential in seed priming for sustainable agriculture[J]. Nanomaterials, 2021, 11(2): 267. |
| [65] | Lee JHJ, Kasote DM. Nano-priming for inducing salinity tolerance, disease resistance, yield attributes, and alleviating heavy metal toxicity in plants[J]. Plants, 2024, 13(3): 446. |
| [66] | Bhuvaneshwari S, Padmalochana K, Natarajan A, et al. Green synthesis and characterization of ZnO nanoparticles using seaweed extract of Halimeda opuntia and their application in seed germination of maize[J]. Biomass Convers Biorefin, 2024. DOI: https://doi.org/10.1007/s13399-024-05713-z. |
| [67] |
Mickelbart MV, Hasegawa PM, Bailey-Serres J. Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability[J]. Nat Rev Genet, 2015, 16: 237-251.
doi: 10.1038/nrg3901 pmid: 25752530 |
| [1] | 殷缘, 程爽, 刘定豪, 邓晓霞, 李凯月, 王竞红, 蔺吉祥. 外源过氧化氢(H2O2)影响非生物胁迫下植物生长与生理代谢机制的研究进展[J]. 生物技术通报, 2025, 41(1): 1-13. |
| [2] | 李禹欣, 李苗, 杜晓芬, 韩康妮, 连世超, 王军. 谷子SiSAP基因家族的鉴定与表达分析[J]. 生物技术通报, 2025, 41(1): 143-156. |
| [3] | 袁柳娇, 黄文琳, 陈崇志, 梁敏, 黄梓淇, 陈雪雪, 陈日檬, 王锂韫. 盐胁迫对广藿香叶片生理特性、超微结构及药效成分的影响[J]. 生物技术通报, 2025, 41(1): 230-239. |
| [4] | 文静, 李倩倩, 张明达, 谭茗月, 金博阳, 沈秀丽, 杜志强. Duox 2调控克氏原螯虾肠组织抗细菌先天免疫的分子机制[J]. 生物技术通报, 2025, 41(1): 324-332. |
| [5] | 满全财, 孟姿诺, 李伟, 蔡心汝, 苏润东, 付长青, 高顺娟, 崔江慧. 马铃薯AQP基因家族鉴定及表达分析[J]. 生物技术通报, 2024, 40(9): 51-63. |
| [6] | 申鹏, 高雅彬, 丁红. 马铃薯SAT基因家族的鉴定和表达分析[J]. 生物技术通报, 2024, 40(9): 64-73. |
| [7] | 李勇慧, 鲍星星, 段一珂, 赵运霞, 于相丽, 陈尧, 张延召. 灵宝杜鹃bZIP家族全基因组鉴定及表达特征分析[J]. 生物技术通报, 2024, 40(8): 186-198. |
| [8] | 崔原瑗, 王昭懿, 白双宇, 任毓昭, 豆飞飞, 刘彩霞, 刘凤楼, 王掌军, 李清峰. 大麦非特异性磷脂酶C基因家族全基因组鉴定及苗期胁迫表达分析[J]. 生物技术通报, 2024, 40(8): 74-82. |
| [9] | 刘丹丹, 王雷刚, 孙明慧, 焦小雨, 吴琼, 王文杰. 茶树海藻糖-6-磷酸合成酶(TPS)基因家族鉴定与表达分析[J]. 生物技术通报, 2024, 40(8): 152-163. |
| [10] | 余纽, 柳帆, 杨锦昌. 油楠SgTPS7的克隆及其在萜类生物合成和非生物胁迫中的功能[J]. 生物技术通报, 2024, 40(8): 164-173. |
| [11] | 吴丁洁, 陈盈盈, 徐静, 刘源, 张航, 李瑞丽. 植物赤霉素氧化酶及其功能研究进展[J]. 生物技术通报, 2024, 40(7): 43-54. |
| [12] | 杜仲阳, 杨泽, 梁梦静, 刘义珍, 崔红利, 史达明, 薛金爱, 孙岩, 张春辉, 季春丽, 李润植. 纳米硒(SeNPs)缓解烟草幼苗铅胁迫和促生效应[J]. 生物技术通报, 2024, 40(7): 183-196. |
| [13] | 蔡志成, 王媛媛, 桑晓涵, 曾丽仙, 邓文韬, 王佳媚. 低温等离子体活化溶液在抑菌及清除生物被膜中的研究进展[J]. 生物技术通报, 2024, 40(6): 95-104. |
| [14] | 胡雅丹, 伍国强, 刘晨, 魏明. MYB转录因子在调控植物响应逆境胁迫中的作用[J]. 生物技术通报, 2024, 40(6): 5-22. |
| [15] | 常雪瑞, 王田田, 王静. 辣椒E2基因家族的鉴定及分析[J]. 生物技术通报, 2024, 40(6): 238-250. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||