








生物技术通报 ›› 2026, Vol. 42 ›› Issue (2): 121-135.doi: 10.13560/j.cnki.biotech.bull.1985.2025-0826
姜逸1( ), 高明燕1, 张笛1, 付立霞2, 程旭1, 陈祥3, 龚建森1, 张莹1, 杨亲1, 俞燕1(
), 高明燕1, 张笛1, 付立霞2, 程旭1, 陈祥3, 龚建森1, 张莹1, 杨亲1, 俞燕1( )
)
收稿日期:2025-07-31
出版日期:2026-02-26
发布日期:2026-03-17
通讯作者:
俞燕,女,博士,研究员,研究方向 :病毒病原学、病毒感染与致病机制、疫苗研制;E-mail: yuyan_1020@126.com作者简介:姜逸,女,博士,副研究员,研究方向 :冠状病毒病原学、致病机制;E-mail: yijiang620@126.com
基金资助:
JIANG Yi1( ), GAO Ming-yan1, ZHANG Di1, FU Li-xia2, CHENG Xu1, CHEN Xiang3, GONG Jian-sen1, ZHANG Ying1, YANG Qin1, YU Yan1(
), GAO Ming-yan1, ZHANG Di1, FU Li-xia2, CHENG Xu1, CHEN Xiang3, GONG Jian-sen1, ZHANG Ying1, YANG Qin1, YU Yan1( )
)
Received:2025-07-31
Published:2026-02-26
Online:2026-03-17
摘要:
冠状病毒作为一类RNA病毒,其宿主范围广泛,能够有效感染哺乳动物和禽类。该类病毒对宿主细胞的入侵过程与棘突蛋白(spike, S)密切相关。冠状病毒的S蛋白是位于病毒表面的主要糖蛋白,由S1与S2亚基组成,其中S1亚基识别并结合宿主细胞受体,S2亚基在宿主蛋白酶裂解后发生构象重排,从而驱动病毒与细胞膜融合实现入侵。不同冠状病毒S蛋白上存在多个宿主蛋白酶识别的基序,能够被跨膜丝氨酸蛋白酶(transmembrane serine protease 2, TMPRSS2)、弗林蛋白酶(Furin)、组织蛋白酶L/B(cathepsin L/B, CTSL/B)以及胰蛋白酶等识别并发生裂解作用。这些蛋白酶裂解位点的分子特征与激活机制存在差异,并决定了病毒对宿主细胞的入侵途径与感染效率。而不同组织或细胞中的宿主蛋白酶表达水平与活性差异,影响了病毒的组织嗜性与传播途径。针对这些宿主蛋白酶开发的一些蛋白酶抑制剂,能够有效阻断病毒的入侵与传播作用,是目前抗冠状病毒药物研发的重要方向。本文系统总结了相关冠状病毒S蛋白关键裂解位点的分子特征,并探讨了不同宿主蛋白酶的激活机制,阐述了针对这些蛋白酶抑制剂的开发策略及其在抗病毒治疗中的应用前景。本综述拟通过对这一关键机制的探讨与解析,为深入理解冠状病毒致病机制提供理论支持,并为新型抗病毒药物的设计与研发提供科学依据。
姜逸, 高明燕, 张笛, 付立霞, 程旭, 陈祥, 龚建森, 张莹, 杨亲, 俞燕. 宿主蛋白酶与冠状病毒入侵:从S蛋白裂解机制到蛋白酶抑制剂的开发应用[J]. 生物技术通报, 2026, 42(2): 121-135.
JIANG Yi, GAO Ming-yan, ZHANG Di, FU Li-xia, CHENG Xu, CHEN Xiang, GONG Jian-sen, ZHANG Ying, YANG Qin, YU Yan. Host Proteases and Coronavirus Invasion: From S Protein Cleavage Mechanism to Development and Applications of Protease Inhibitor[J]. Biotechnology Bulletin, 2026, 42(2): 121-135.
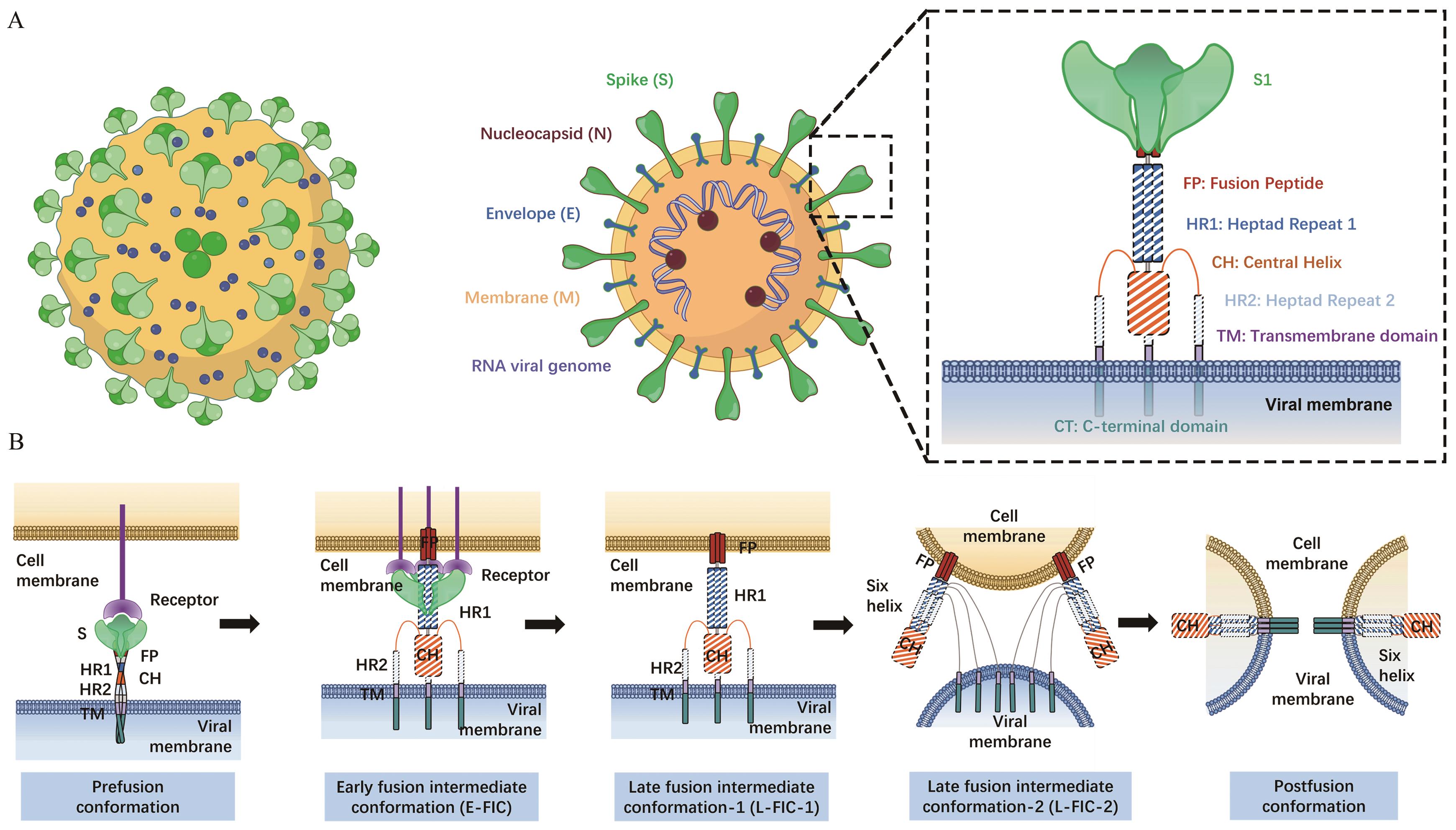
图1 冠状病毒S蛋白结构示意图A:冠状病毒及S蛋白结构;B:膜融合
Fig. 1 Schematic diagram of S protein structure of coronavirusA: Coronavirus and S protein structure. B: Membrane fusion
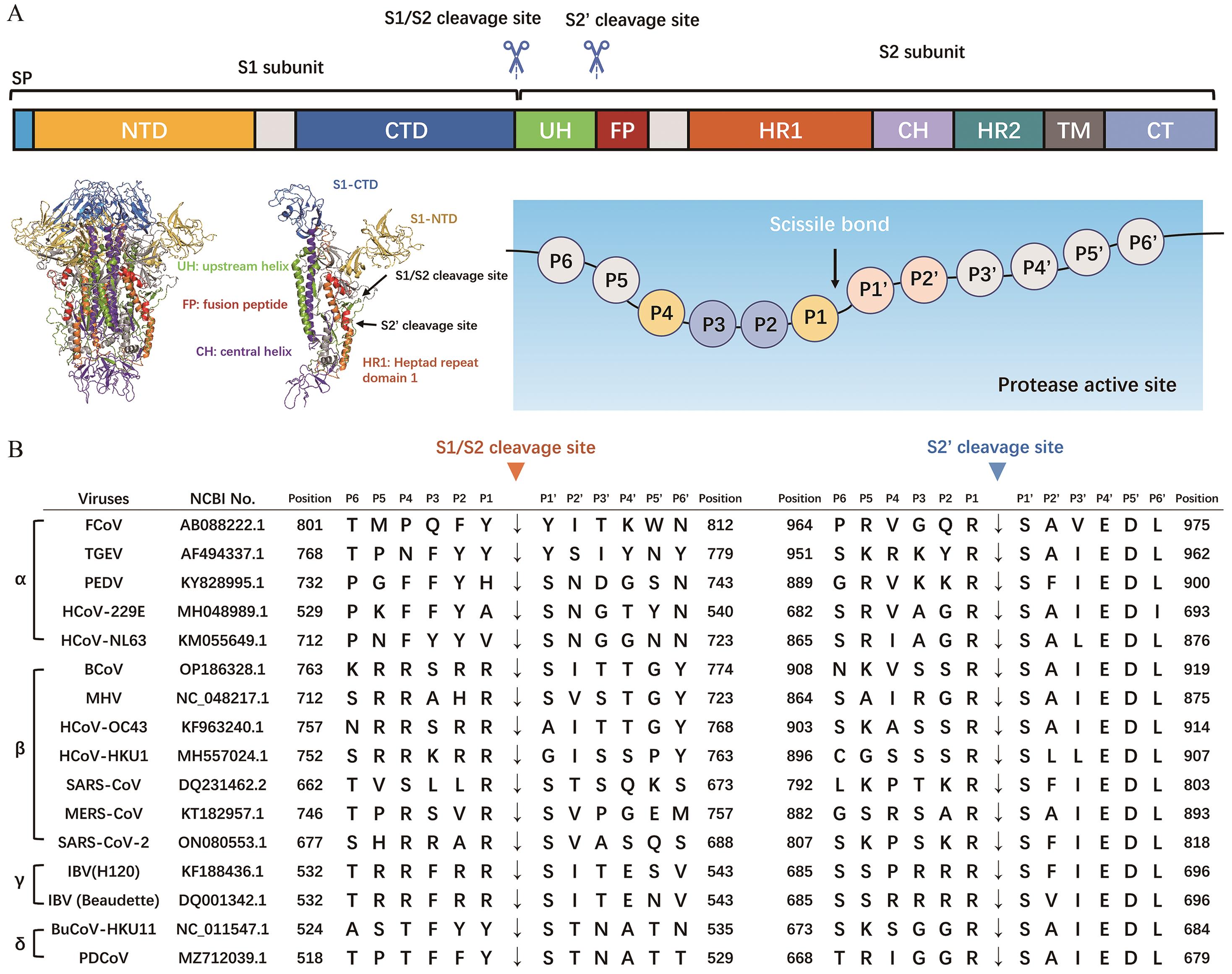
图2 不同冠状病毒蛋白酶裂解位点基序A:蛋白酶活化位点示意图;B:不同冠状病毒S1/S2或S2′裂解位点基序
Fig. 2 Cleavage site motifs of proteases among different coronavirusesA: Schematic of protease activation site. B: S1/S2 or S2′ cleavage site motifs of different coronaviruses
蛋白酶 Protease | 细胞定位 Cellular localization | 作用途径 Pathway | 识别基序 Recognized motif P2-P1↓P1′-P2′ | 是否依赖酸性环境 Acid dependence | 参考文献 References |
|---|---|---|---|---|---|
| Furin | TGN | 生物合成 | RXK/RR↓S | 不依赖 | [ |
| TMPRSS2 | 跨膜 | 质膜途径 | K/R↓S | 不依赖 | [ |
| CTSL/B | 内质体/溶酶体 | 内体途径 | R/K/G/A/S/T/Q/Y↓ | 依赖,pH 3.0-6.5 | [ |
| HAT | 跨膜 | 质膜途径 | K/R↓ | 不依赖 | [ |
| 胰蛋白酶 | 细胞外 | 质膜途径 | 选择性不高,主要K/R↓S | 中性pH值,最适pH 8.0 | [ |
| 弹性蛋白酶 | 细胞外 | 质膜途径 | A/S/G/V/W/Y/F/L/C/M↓ | 不依赖 | [ |
| 纤溶酶 | 细胞外 | 质膜途径 | K/R↓ | 不依赖 | [ |
表1 不同宿主蛋白酶分布与裂解特性
Table 1 Distribution and cleavage properties of proteases in different hosts
蛋白酶 Protease | 细胞定位 Cellular localization | 作用途径 Pathway | 识别基序 Recognized motif P2-P1↓P1′-P2′ | 是否依赖酸性环境 Acid dependence | 参考文献 References |
|---|---|---|---|---|---|
| Furin | TGN | 生物合成 | RXK/RR↓S | 不依赖 | [ |
| TMPRSS2 | 跨膜 | 质膜途径 | K/R↓S | 不依赖 | [ |
| CTSL/B | 内质体/溶酶体 | 内体途径 | R/K/G/A/S/T/Q/Y↓ | 依赖,pH 3.0-6.5 | [ |
| HAT | 跨膜 | 质膜途径 | K/R↓ | 不依赖 | [ |
| 胰蛋白酶 | 细胞外 | 质膜途径 | 选择性不高,主要K/R↓S | 中性pH值,最适pH 8.0 | [ |
| 弹性蛋白酶 | 细胞外 | 质膜途径 | A/S/G/V/W/Y/F/L/C/M↓ | 不依赖 | [ |
| 纤溶酶 | 细胞外 | 质膜途径 | K/R↓ | 不依赖 | [ |
| [72] | Regan AD, Shraybman R, Cohen RD, et al. Differential role for low pH and cathepsin-mediated cleavage of the viral spike protein during entry of serotype II feline coronaviruses [J]. Vet Microbiol, 2008, 132(3/4): 235-248. |
| [73] | Xiao WW, Xiong YX, Wang YC, et al. Cathepsin L and transmembrane serine protease 11E mediate trypsin-independent entry of porcine deltacoronavirus into Huh7 cells [J]. J Virol, 2025, 99(9): e0105525. |
| [74] | Bertram S, Glowacka I, Müller MA, et al. Cleavage and activation of the severe acute respiratory syndrome coronavirus spike protein by human airway trypsin-like protease [J]. J Virol, 2011, 85(24): 13363-13372. |
| [75] | Ren WY, Hong WQ, Yang JY, et al. SARS-CoV-2 Delta and Omicron variants resist spike cleavage by human airway trypsin-like protease [J]. J Clin Investig, 2024, 134(18): e174304. |
| [76] | Kishimoto M, Uemura K, Sanaki T, et al. TMPRSS11D and TMPRSS13 activate the SARS-CoV-2 spike protein [J]. Viruses, 2021, 13(3): 384. |
| [77] | Wicht O, Li WT, Willems L, et al. Proteolytic activation of the porcine epidemic diarrhea coronavirus spike fusion protein by trypsin in cell culture [J]. J Virol, 2014, 88(14): 7952-7961. |
| [78] | Costello DA, Millet JK, Hsia CY, et al. Single particle assay of coronavirus membrane fusion with proteinaceous receptor-embedded supported bilayers [J]. Biomaterials, 2013, 34(32): 7895-7904. |
| [79] | Matsuyama S, Ujike M, Morikawa S, et al. Protease-mediated enhancement of severe acute respiratory syndrome coronavirus infection [J]. Proc Natl Acad Sci U S A, 2005, 102(35): 12543-12547. |
| [80] | Jaimes JA, Millet JK, Whittaker GR. Proteolytic cleavage of the SARS-CoV-2 spike protein and the role of the novel S1/S2 site [J]. iScience, 2020, 23(6): 101212. |
| [81] | Kim Y, Jang G, Lee DR, et al. Trypsin enhances SARS-CoV-2 infection by facilitating viral entry [J]. Arch Virol, 2022, 167(2): 441-458. |
| [82] | Hofmann M, Wyler R. Propagation of the virus of porcine epidemic diarrhea in cell culture [J]. J Clin Microbiol, 1988, 26(11): 2235-2239. |
| [83] | Shirato K, Matsuyama S, Ujike M, et al. Role of proteases in the release of porcine epidemic diarrhea virus from infected cells [J]. J Virol, 2011, 85(15): 7872-7880. |
| [84] | Xiao WW, Li Z, Chen CQ, et al. Revisiting the roles of trypsin in the productive infection of porcine deltacoronavirus in porcine-derived cells [J]. Virology, 2025, 604: 110453. |
| [85] | Korkmaz B, Gauthier F. Chapter 587 - Elastase-2/Leukocyte Elastase [M]//Rawlings ND, Salvesen G. Handbook of Proteolytic Enzymes (Third Edition). Academic Press. 2013: 2653-2661. |
| [86] | Heald-Sargent T, Gallagher T. Ready, set, fuse! the coronavirus spike protein and acquisition of fusion competence [J]. Viruses, 2012, 4(4): 557-580. |
| [87] | Kawabata K, Hagio T, Matsuoka S. The role of neutrophil elastase in acute lung injury [J]. Eur J Pharmacol, 2002, 451(1): 1-10. |
| [88] | Szturmowicz M, Demkow U. Neutrophil extracellular traps (NETs) in severe SARS-CoV-2 lung disease [J]. Int J Mol Sci, 2021, 22(16): 8854. |
| [89] | Kim Y, Oh C, Shivanna V, et al. Trypsin-independent porcine epidemic diarrhea virus US strain with altered virus entry mechanism [J]. BMC Vet Res, 2017, 13(1): 356. |
| [90] | Ji HL, Zhao RZ, Matalon S, et al. Elevated plasmin(ogen) as a common risk factor for COVID-19 susceptibility [J]. Physiol Rev, 2020, 100(3): 1065-1075. |
| [91] | Kam YW, Okumura Y, Kido H, et al. Cleavage of the SARS coronavirus spike glycoprotein by airway proteases enhances virus entry into human bronchial epithelial cells in vitro [J]. PLoS One, 2009, 4(11): e7870. |
| [92] | Kaur U, Chakrabarti SS, Ojha B, et al. Targeting host cell proteases to prevent SARS-CoV-2 invasion [J]. Curr Drug Targets, 2021, 22(2): 192-201. |
| [93] | Glowacka I, Bertram S, Müller MA, et al. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response [J]. J Virol, 2011, 85(9): 4122-4134. |
| [94] | Han YT, Ma YL, Wang ZQ, et al. TMPRSS13 promotes the cell entry of swine acute diarrhea syndrome coronavirus [J]. J Med Virol, 2024, 96(6): e29712. |
| [95] | McKee DL, Sternberg A, Stange U, et al. Candidate drugs against SARS-CoV-2 and COVID-19 [J]. Pharmacol Res, 2020, 157: 104859. |
| [96] | Hernández-Mitre MP, Morpeth SC, Venkatesh B, et al. TMPRSS2 inhibitors for the treatment of COVID-19 in adults: a systematic review and meta-analysis of randomized clinical trials of nafamostat and camostat mesylate [J]. Clin Microbiol Infect, 2024, 30(6): 743-754. |
| [97] | Hoffmann M, Schroeder S, Kleine-Weber H, et al. Nafamostat mesylate blocks activation of SARS-CoV-2: new treatment option for COVID-19 [J]. Antimicrob Agents Chemother, 2020, 64(6): e00754-20. |
| [98] | Li K, Meyerholz DK, Bartlett JA, et al. The TMPRSS2 inhibitor nafamostat reduces SARS-CoV-2 pulmonary infection in mouse models of COVID-19 [J]. mBio, 2021, 12(4): e00970-21. |
| [99] | Lucas JM, Heinlein C, Kim T, et al. The androgen-regulated protease TMPRSS2 activates a proteolytic cascade involving components of the tumor microenvironment and promotes prostate cancer metastasis [J]. Cancer Discov, 2014, 4(11): 1310-1325. |
| [100] | Wettstein L, Weil T, Conzelmann C, et al. Alpha-1 antitrypsin inhibits TMPRSS2 protease activity and SARS-CoV-2 infection [J]. Nat Commun, 2021, 12: 1726. |
| [101] | Walker B, Lynas JF. Strategies for the inhibition of serine proteases [J]. Cell Mol Life Sci CMLS, 2001, 58(4): 596-624. |
| [102] | Singh S, O’Reilly S, Gewaid H, et al. Reactive centre loop mutagenesis of SerpinB3 to target TMPRSS2 and furin: inhibition of SARS-CoV-2 cell entry and replication [J]. Int J Mol Sci, 2022, 23(20): 12522. |
| [103] | Keller C, Böttcher-Friebertshäuser E, Lohoff M. TMPRSS2, a novel host-directed drug target against SARS-CoV-2 [J]. Signal Transduct Target Ther, 2022, 7: 251. |
| [104] | Mahoney M, Damalanka VC, Tartell MA, et al. A novel class of TMPRSS2 inhibitors potently block SARS-CoV-2 and MERS-CoV viral entry and protect human epithelial lung cells [J]. Proc Natl Acad Sci U S A, 2021, 118(43): e2108728118. |
| [105] | Boon ACM, Bricker TL, Fritch EJ, et al. Efficacy of host cell serine protease inhibitor MM3122 against SARS-CoV-2 for treatment and prevention of COVID-19 [J]. J Virol, 2024, 98(5): e01903-23. |
| [106] | Cheng YW, Chao TL, Li CL, et al. Furin inhibitors block SARS-CoV-2 spike protein cleavage to suppress virus production and cytopathic effects [J]. Cell Rep, 2020, 33(2): 108254. |
| [107] | Tanikawa T, Hayashi T, Suzuki R, et al. Inhibitory effect of honokiol on furin-like activity and SARS-CoV-2 infection [J]. J Tradit Complementary Med, 2022, 12(1): 69-72. |
| [108] | Jean F, Stella K, Thomas L, et al. α1-Antitrypsin Portland, a bioengineered serpin highly selective for furin: Application as an antipathogenic agent [J]. Proc Natl Acad Sci U S A, 1998, 95(13): 7293-7298. |
| [1] | Woo PCY, Lau SKP, Huang Y, et al. Coronavirus diversity, phylogeny and interspecies jumping [J]. Exp Biol Med (Maywood), 2009, 234(10): 1117-1127. |
| [2] | Drosten C, Günther S, Preiser W, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome [J]. N Engl J Med, 2003, 348(20): 1967-1976. |
| [3] | van Boheemen S, de Graaf M, Lauber C, et al. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans [J]. mBio, 2012, 3(6): e00473-12. |
| [4] | Enjuanes L, Almazán F, Sola I, et al. Biochemical aspects of coronavirus replication and virus-host interaction [J]. Annu Rev Microbiol, 2006, 60: 211-230. |
| [5] | Guillén J, Pérez-Berná AJ, Moreno MR, et al. Identification of the membrane-active regions of the severe acute respiratory syndrome coronavirus spike membrane glycoprotein using a 16/18-mer peptide scan: implications for the viral fusion mechanism [J]. J Virol, 2005, 79(3): 1743-1752. |
| [6] | Xing LX, Liu ZM, Wang XL, et al. Early fusion intermediate of ACE2-using coronavirus spike acting as an antiviral target [J]. Cell, 2025, 188(5): 1297-1314.e24. |
| [7] | Belouzard S, Chu VC, Whittaker GR. Activation of the SARS coronavirus spike protein via sequential proteolytic cleavage at two distinct sites [J]. Proc Natl Acad Sci U S A, 2009, 106(14): 5871-5876. |
| [8] | Millet JK, Whittaker GR. Host cell proteases: Critical determinants of coronavirus tropism and pathogenesis [J]. Virus Res, 2015, 202: 120-134. |
| [9] | de Haan CAM, Stadler K, Godeke GJ, et al. Cleavage inhibition of the murine coronavirus spike protein by a furin-like enzyme affects cell-cell but not virus-cell fusion [J]. J Virol, 2004, 78(11): 6048-6054. |
| [10] | Park JE, Li K, Barlan A, et al. Proteolytic processing of Middle East respiratory syndrome coronavirus spikes expands virus tropism [J]. Proc Natl Acad Sci U S A, 2016, 113(43): 12262-12267. |
| [11] | Millet JK, Whittaker GR. Host cell entry of Middle East respiratory syndrome coronavirus after two-step, furin-mediated activation of the spike protein [J]. Proc Natl Acad Sci U S A, 2014, 111(42): 15214-15219. |
| [12] | Yamada Y, Liu DX. Proteolytic activation of the spike protein at a novel RRRR/S motif is implicated in furin-dependent entry, syncytium formation, and infectivity of coronavirus infectious bronchitis virus in cultured cells [J]. J Virol, 2009, 83(17): 8744-8758. |
| [13] | Hansen GH, Delmas B, Besnardeau L, et al. The coronavirus transmissible gastroenteritis virus causes infection after receptor-mediated endocytosis and acid-dependent fusion with an intracellular compartment [J]. J Virol, 1998, 72(1): 527-534. |
| [14] | Shen LW, Mao HJ, Wu YL, et al. TMPRSS2: a potential target for treatment of influenza virus and coronavirus infections [J]. Biochimie, 2017, 142: 1-10. |
| [15] | Seidah NG, Prat A. The biology and therapeutic targeting of the proprotein convertases [J]. Nat Rev Drug Discov, 2012, 11(5): 367-383. |
| [16] | de Haan CAM, Haijema BJ, Schellen P, et al. Cleavage of group 1 coronavirus spike proteins: how furin cleavage is traded off against heparan sulfate binding upon cell culture adaptation [J]. J Virol, 2008, 82(12): 6078-6083. |
| [17] | de Haan CAM, Rottier PJM. Molecular interactions in the assembly of coronaviruses [J]. Adv Virus Res, 2005, 64: 165-230. |
| [18] | Choe Y, Leonetti F, Greenbaum DC, et al. Substrate profiling of cysteine proteases using a combinatorial peptide library identifies functionally unique specificities [J]. J Biol Chem, 2006, 281(18): 12824-12832. |
| [19] | Kawase M, Shirato K, van der Hoek L, et al. Simultaneous treatment of human bronchial epithelial cells with serine and cysteine protease inhibitors prevents severe acute respiratory syndrome coronavirus entry [J]. J Virol, 2012, 86(12): 6537-6545. |
| [20] | Henrich S, Cameron A, Bourenkov GP, et al. The crystal structure of the proprotein processing proteinase furin explains its stringent specificity [J]. Nat Struct Mol Biol, 2003, 10(7): 520-526. |
| [21] | Nakayama K. Furin: a mammalian subtilisin/Kex2p-like endoprotease involved in processing of a wide variety of precursor proteins [J]. Biochem J, 1997, 327 ( Pt 3)(Pt 3): 625-635. |
| [22] | Khatri R, Lohiya B, Kaur G, et al. Understanding the role of conserved proline and serine residues in the SARS-CoV-2 spike cleavage sites in the virus entry, fusion, and infectivity [J]. 3 Biotech, 2023, 13(10): 323. |
| [23] | Kirschke H. Chapter 410 - Cathepsin L [M]//Rawlings ND, Salvesen G. Handbook of Proteolytic Enzymes (Third Edition). Academic Press: Elsevier, 2013: 1808-1817. |
| [24] | Zhao MM, Zhu Y, Zhang L, et al. Novel cleavage sites identified in SARS-CoV-2 spike protein reveal mechanism for cathepsin L-facilitated viral infection and treatment strategies [J]. Cell Discov, 2022, 8: 53. |
| [25] | Evnin Luke B. Substrate specificity of trypsin investigated by using a genetic selection [J]. Proc Natl Acad Sci U S A, 1990, 87(17): 6659-6663. |
| [26] | Baird JTT, Craik CS. Chapter 575 - Trypsin [M]//Rawlings ND, Salvesen G. Handbook of Proteolytic Enzymes (Third Edition). Academic Press: Elsevier, 2013: 2594-2600. |
| [27] | Belouzard S, Madu I, Whittaker GR. Elastase-mediated activation of the severe acute respiratory syndrome coronavirus spike protein at discrete sites within the S2 domain [J]. J Biol Chem, 2010, 285(30): 22758-22763. |
| [28] | Bieth, 2004. Leukocyte elastase. In: Barrett, Rawlings, Woessner (Eds.), Handbook of Proteolytic Enzymes [M]. vol. 2, second ed, pp. 1517-1523. |
| [29] | Backes BJ, Harris JL, Leonetti F, et al. Synthesis of positional-scanning libraries of fluorogenic peptide substrates to define the extended substrate specificity of plasmin and thrombin [J]. Nat Biotechnol, 2000, 18(2): 187-193. |
| [30] | Kleine-Weber H, Elzayat MT, Hoffmann M, et al. Functional analysis of potential cleavage sites in the MERS-coronavirus spike protein [J]. Sci Rep, 2018, 8: 16597. |
| [31] | Reinke LM, Spiegel M, Plegge T, et al. Different residues in the SARS-CoV spike protein determine cleavage and activation by the host cell protease TMPRSS2 [J]. PLoS One, 2017, 12(6): e0179177. |
| [32] | Jackson CB, Farzan M, Chen B, et al. Mechanisms of SARS-CoV-2 entry into cells [J]. Nat Rev Mol Cell Biol, 2022, 23(1): 3-20. |
| [33] | Rajah MM, Hubert M, Bishop E, et al. SARS-CoV-2 Alpha, Beta, and Delta variants display enhanced Spike-mediated syncytia formation [J]. EMBO J, 2021, 40(24): e108944. |
| [34] | Escalera A, Gonzalez-Reiche AS, Aslam S, et al. Mutations in SARS-CoV-2 variants of concern link to increased spike cleavage and virus transmission [J]. Cell Host Microbe, 2022, 30(3): 373-387.e7. |
| [35] | Liu Y, Liu JY, Johnson BA, et al. Delta spike P681R mutation enhances SARS-CoV-2 fitness over Alpha variant [J]. Cell Rep, 2022, 39(7): 110829. |
| [36] | Saito A, Irie T, Suzuki R, et al. Enhanced fusogenicity and pathogenicity of SARS-CoV-2 Delta P681R mutation [J]. Nature, 2022, 602(7896): 300-306. |
| [109] | Varughese KI, Ahmed FR, Carey PR, et al. Crystal structure of a papain-E-64 complex [J]. Biochemistry, 1989, 28(3): 1330-1332. |
| [110] | Hu J, Gao QZ, He CL, et al. Development of cell-based pseudovirus entry assay to identify potential viral entry inhibitors and neutralizing antibodies against SARS-CoV-2 [J]. Genes Dis, 2020, 7(4): 551-557. |
| [111] | Mellott DM, Tseng CT, Drelich A, et al. A clinical-stage cysteine protease inhibitor blocks SARS-CoV-2 infection of human and monkey cells [J]. ACS Chem Biol, 2021, 16(4): 642-650. |
| [112] | Zhou YC, Vedantham P, Lu K, et al. Protease inhibitors targeting coronavirus and filovirus entry [J]. Antivir Res, 2015, 116: 76-84. |
| [113] | 周雯雯, 尤宝庆, 郑怡凡, 等. 组织蛋白酶L小分子抑制剂的抗SARS-CoV-2活性研究 [J]. 药学学报, 2024, 59(3): 600-607. |
| Zhou WW, You BQ, Zheng YF, et al. Anti-SARS-CoV-2 activity of small molecule inhibitors of cathepsin L [J]. Acta Pharm Sin, 2024, 59(3): 600-607. | |
| [114] | Zhou N, Pan T, Zhang JS, et al. Glycopeptide antibiotics potently inhibit cathepsin L in the late endosome/lysosome and block the entry of Ebola virus, middle east respiratory syndrome coronavirus (MERS-CoV), and severe acute respiratory syndrome coronavirus (SARS-CoV) [J]. J Biol Chem, 2016, 291(17): 9218-9232. |
| [115] | Mao BL, Le-Trilling VTK, Tang HH, et al. Diphyllin elicits a doubled-pronged attack on the entry of SARS-CoV-2 by inhibiting cathepsin L and furin [J]. Virus Res, 2024, 350: 199485. |
| [116] | Li YH, Wang K, Sun HM, et al. Omicsynin B4 potently blocks coronavirus infection by inhibiting host proteases cathepsin L and TMPRSS2 [J]. Antivir Res, 2023, 214: 105606. |
| [117] | Essalmani R, Jain J, Susan-Resiga D, et al. Distinctive roles of furin and TMPRSS2 in SARS-CoV-2 infectivity [J]. J Virol, 2022, 96(8): e00128-22. |
| [37] | Wicht O, Burkard C, de Haan CAM, et al. Identification and characterization of a proteolytically primed form of the murine coronavirus spike proteins after fusion with the target cell [J]. J Virol, 2014, 88(9): 4943-4952. |
| [38] | Choi A, Kots ED, Singleton DT, et al. Analysis of the molecular determinants for furin cleavage of the spike protein S1/S2 site in defined strains of the prototype coronavirus murine hepatitis virus (MHV) [J]. Virus Res, 2024, 340: 199283. |
| [39] | Hoffmann M, Kleine-Weber H, Pöhlmann S. A multibasic cleavage site in the spike protein of SARS-CoV-2 is essential for infection of human lung cells [J]. Mol Cell, 2020, 78(4): 779-784.e5. |
| [40] | Escalera A, Laporte M, Turner S, et al. The impact of S2 mutations on Omicron SARS-CoV-2 cell surface expression and fusogenicity [J]. Emerg Microbes Infect, 2024, 13: 2297553. |
| [41] | Park SB, Khan M, Chiliveri SC, et al. SARS-CoV-2 Omicron variants harbor spike protein mutations responsible for their attenuated fusogenic phenotype [J]. Commun Biol, 2023, 6: 556. |
| [42] | Johnson BA, Xie XP, Bailey AL, et al. Loss of furin cleavage site attenuates SARS-CoV-2 pathogenesis [J]. Nature, 2021, 591(7849): 293-299. |
| [43] | Hoffmann M, Kleine-Weber H, Graichen L, et al. Acquisition of a multibasic cleavage site does not increase MERS-CoV entry into Calu-3 human lung cells [J]. J Virol, 2024, 98(11): e01305-24. |
| [44] | Cheng JL, Zhao Y, Hu YX, et al. The furin-S2’ site in avian coronavirus plays a key role in central nervous system damage progression [J]. J Virol, 2021, 95(11): JVI.02447-JVI.02420. |
| [45] | Li WT, Wicht O, van Kuppeveld FJM, et al. A single point mutation creating a furin cleavage site in the spike protein renders porcine epidemic diarrhea coronavirus trypsin independent for cell entry and fusion [J]. J Virol, 2015, 89(15): 8077-8081. |
| [46] | Bickerton E, Maier HJ, Stevenson-Leggett P, et al. The S2 subunit of infectious bronchitis virus beaudette is a determinant of cellular tropism [J]. J Virol, 2018, 92(19): e01044-18. |
| [47] | Jiang Y, Gao MY, Cheng X, et al. The V617I substitution in avian coronavirus IBV spike protein plays a crucial role in adaptation to primary chicken kidney cells [J]. Front Microbiol, 2020, 11: 604335. |
| [48] | 姜逸, 周生, 俞燕, 等. 表达不同致病型S基因重组鸡传染性支气管炎病毒的细胞嗜性研究 [J]. 病毒学报, 2017, 33(5): 738-744. |
| Jiang Y, Zhou S, Yu Y, et al. Cell tropism of the recombinant chicken infectious bronchitis virus expressing the S gene of different pathogenic strains [J]. Chin J Virol, 2017, 33(5): 738-744. | |
| [49] | Jiang Y, Xue M, Tang MJ, et al. Adaptation of the infectious bronchitis virus H120 vaccine strain to Vero cell lines [J]. Vet Microbiol, 2023, 280: 109709. |
| [50] | Jiang Y, Cheng X, Gao MY, et al. Two mutations on S2 subunit were critical for Vero cell tropism expansion of infectious bronchitis virus HV80 [J]. Vet Microbiol, 2024, 294: 110134. |
| [51] | Liu DX, Liang JQ, Fung TS. Human coronavirus-229E, -OC43, -NL63, and-HKU1 (Coronaviridae) [M]//Encyclopedia of Virology. Amsterdam: Elsevier, 2021: 428-440. |
| [52] | Belouzard S, Millet JK, Licitra BN, et al. Mechanisms of coronavirus cell entry mediated by the viral spike protein [J]. Viruses, 2012, 4(6): 1011-1033. |
| [53] | Shirato K, Kawase M, Matsuyama S. Middle east respiratory syndrome coronavirus infection mediated by the transmembrane serine protease TMPRSS2 [J]. J Virol, 2013, 87(23): 12552-12561. |
| [54] | Bestle D, Heindl MR, Limburg H, et al. TMPRSS2 and furin are both essential for proteolytic activation of SARS-CoV-2 in human airway cells [J]. Life Sci Alliance, 2020, 3(9): e202000786. |
| [55] | Shi W, Fan WL, Bai J, et al. TMPRSS2 and MSPL facilitate trypsin-independent porcine epidemic diarrhea virus replication in vero cells [J]. Viruses, 2017, 9(5): 114. |
| [56] | Lukassen S, Chua RL, Trefzer T, et al. SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells [J]. EMBO J, 2020: e105114. |
| [57] | Kakizaki M, Hashimoto R, Nagata N, et al. The respective roles of TMPRSS2 and cathepsins for SARS-CoV-2 infection in human respiratory organoids [J]. J Virol, 2025, 99: e01853-24. |
| [58] | Yang Y, Liu C, Du LY, et al. Two mutations were critical for bat-to-human transmission of middle east respiratory syndrome coronavirus [J]. J Virol, 2015, 89(17): 9119-9123. |
| [59] | Turk V, Stoka V, Vasiljeva O, et al. Cysteine cathepsins: From structure, function and regulation to new frontiers [J]. Biochim Biophys Acta Proteins Proteom, 2012, 1824(1): 68-88. |
| [60] | Biniossek ML, Nägler DK, Becker-Pauly C, et al. Proteomic identification of protease cleavage sites characterizes prime and non-prime specificity of cysteine cathepsins B, L, and S [J]. J Proteome Res, 2011, 10(12): 5363-5373. |
| [61] | Bosch BJ, Bartelink W, Rottier PJM. Cathepsin L functionally cleaves the severe acute respiratory syndrome coronavirus class I fusion protein upstream of rather than adjacent to the fusion peptide [J]. J Virol, 2008, 82(17): 8887-8890. |
| [62] | Willett BJ, Grove J, MacLean OA, et al. SARS-CoV-2 Omicron is an immune escape variant with an altered cell entry pathway [J]. Nat Microbiol, 2022, 7(8): 1161-1179. |
| [63] | Simmons G, Gosalia DN, Rennekamp AJ, et al. Inhibitors of cathepsin L prevent severe acute respiratory syndrome coronavirus entry [J]. Proc Natl Acad Sci U S A, 2005, 102(33): 11876-11881. |
| [64] | Gierer S, Bertram S, Kaup F, et al. The spike protein of the emerging betacoronavirus EMC uses a novel coronavirus receptor for entry, can be activated by TMPRSS2, and is targeted by neutralizing antibodies [J]. J Virol, 2013, 87(10): 5502-5511. |
| [65] | Ou TL, Mou HH, Zhang LZ, et al. Hydroxychloroquine-mediated inhibition of SARS-CoV-2 entry is attenuated by TMPRSS2 [J]. PLoS Pathog, 2021, 17(1): e1009212. |
| [66] | Ou XY, Liu Y, Lei XB, et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV [J]. Nat Commun, 2020, 11: 1620. |
| [67] | Daniloski Z, Jordan TX, Wessels HH, et al. Identification of required host factors for SARS-CoV-2 infection in human cells [J]. Cell, 2021, 184(1): 92-105.e16. |
| [68] | Bolsinger MM, Drobny A, Wilfling S, et al. SARS-CoV-2 spike protein induces time-dependent CTSL upregulation in HeLa cells and alveolarspheres [J]. J Cell Biochem, 2024, 125(9): e30627. |
| [69] | Kawase M, Shirato K, Matsuyama S, et al. Protease-mediated entry via the endosome of human coronavirus 229E [J]. J Virol, 2009, 83(2): 712-721. |
| [70] | Qiu ZZ, Hingley ST, Simmons G, et al. Endosomal proteolysis by cathepsins is necessary for murine coronavirus mouse hepatitis virus type 2 spike-mediated entry [J]. J Virol, 2006, 80(12): 5768-5776. |
| [71] | Park JE, Cruz DJM, Shin HJ. Clathrin- and serine proteases-dependent uptake of porcine epidemic diarrhea virus into Vero cells [J]. Virus Res, 2014, 191: 21-29. |
| [1] | 刘余, 刘筱筱, 郑建豪, 李彬, 何翃闳, 毛立. 牛冠状病毒单克隆抗体的制备和鉴定[J]. 生物技术通报, 2026, 42(1): 67-75. |
| [2] | 谭玉荣, 陈东亮, 杨守臻, 赖振光, 唐向民, 孙祖东, 曾维英. 大豆抗豆卷叶螟GmKTI1-like的功能研究[J]. 生物技术通报, 2025, 41(6): 99-108. |
| [3] | 郑巧, 林华, 徐浩, 安微, 薛昌华, 张婧, 韩国全. SARS-CoV-2 Omicron 变异株多重微滴式数字 PCR 定量方法的建立及应用[J]. 生物技术通报, 2024, 40(2): 80-89. |
| [4] | 王祥锟, 宋学宏, 刘金龙, 郭培红, 庄晓峰, 韦良孟, 周凡, 张树宇, 高攀攀, 魏凯. 新型冠状病毒亚单位疫苗研制及其高效免疫增强剂的筛选[J]. 生物技术通报, 2023, 39(1): 305-314. |
| [5] | 刘晓玫, 王东鑫, 张春, 魏双施. AAV介导的RNAi对SARS-CoV-2 S基因表达的抑制作用[J]. 生物技术通报, 2022, 38(3): 188-193. |
| [6] | 胡秀文, 刘华, 王宇, 唐雪明, 王金斌, 曾海娟, 蒋玮, 李红. CRISPR-Cas系统在核酸检测中的应用研究[J]. 生物技术通报, 2021, 37(9): 266-273. |
| [7] | 瞿欢, 李成, 陈汭, 廖艺杰, 曹三杰, 文翼平, 颜其贵, 黄小波. 猪δ冠状病毒S1-CTD的截短表达及间接ELISA抗体方法的建立[J]. 生物技术通报, 2021, 37(5): 273-280. |
| [8] | 李家俊, 郑潇, 盛杰, 徐瑶. 新型冠状病毒及其临床检测方法研究进展[J]. 生物技术通报, 2021, 37(4): 282-292. |
| [9] | 陈汭, 付嘉钰, 刘浩宇, 李成, 赵玉佳, 胡靖飞, 瞿欢, 曹三杰, 文心田, 文翼平, 赵勤, 伍锐, 黄小波. 猪δ冠状病毒(PDCoV)N蛋白的原核表达及多克隆抗体制备[J]. 生物技术通报, 2020, 36(8): 104-110. |
| [10] | 吕继洲, 吴绍强, 张舟, 邓俊花, 袁向芬, 王彩霞, 冯春燕, 林祥梅. 新型冠状病毒实时荧光双重逆转录RPA的建立及其在食品检测中的应用[J]. 生物技术通报, 2020, 36(11): 238-244. |
| [11] | 韩斐, 江明锋, 冉嫆, 王刚. PAS蛋白融合修饰研究进展[J]. 生物技术通报, 2019, 35(12): 152-158. |
| [12] | 梁丽琴,阎婧,张鑫,郝泽婷,段江燕. CRISPR技术的发展及应用研究进展[J]. 生物技术通报, 2018, 34(5): 9-16. |
| [13] | 张倩倩, 刘康泰, 韩宗晟, 李荣贵. 重组中东呼吸综合征病毒刺突蛋白S1亚基及其编码DNA接种小鼠的抗血清水平研究[J]. 生物技术通报, 2018, 34(10): 150-156. |
| [14] | 王玉, 王建华, 刘允军. AM79 EPSPS蛋白的体外模拟胃肠液消化稳定性研究[J]. 生物技术通报, 2017, 33(6): 176-181. |
| [15] | 龙翔宇, 梁启福, 戚继艳, 方永军, 唐朝荣. 橡胶树半胱氨酸蛋白酶抑制剂HbCYS2的克隆与表达分析[J]. 生物技术通报, 2017, 33(3): 86-92. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||