








生物技术通报 ›› 2025, Vol. 41 ›› Issue (1): 95-102.doi: 10.13560/j.cnki.biotech.bull.1985.2024-0608
张静安1( ), 胡孝龙2, 曹蓓蓓2, 廖敏2, 束长龙3, 张杰3, 王奎2(
), 胡孝龙2, 曹蓓蓓2, 廖敏2, 束长龙3, 张杰3, 王奎2( ), 操海群2(
), 操海群2( )
)
收稿日期:2024-06-27
出版日期:2025-01-26
发布日期:2025-01-22
通讯作者:
操海群,男,教授,研究方向:绿色防控与农产品安全;E-mail: caohaiqun@ahau.edu.cn;作者简介:张静安,男,硕士研究生,研究方向:生物防治;E-mail: 15855350220@163.com
基金资助:
ZHANG Jing-an1( ), HU Xiao-long2, CAO Bei-bei2, LIAO Min2, SHU Chang-long3, ZHANG Jie3, WANG Kui2(
), HU Xiao-long2, CAO Bei-bei2, LIAO Min2, SHU Chang-long3, ZHANG Jie3, WANG Kui2( ), CAO Hai-qun2(
), CAO Hai-qun2( )
)
Received:2024-06-27
Published:2025-01-26
Online:2025-01-22
摘要:
【目的】构建可以快速高效表达苏云金芽胞杆菌(Bacillus thuringiensis,Bt)杀虫基因的表达载体,提高Bt杀虫基因发掘及功能研究效率。【方法】以pUC18载体为基础,构建一个以cry1Ac基因启动子p1Ac指导、融合绿色荧光蛋白GFP的可视化快速表达载体p1Ac-GFP,并从生物活性、碱溶性、抗胰蛋白酶稳定性、培养条件等方面对其进行分析。【结果】p1Ac-GFP在大肠杆菌中指导表达的Cry1Ac蛋白与Bt来源的蛋白在杀虫活性方面无明显差异。同时,p1Ac-GFP表达的Cry1Ac蛋白能够溶解于50 mmol/L Na2CO3溶液中,可溶性组分可被胰蛋白酶消化为60 kD大小的核心活性片段。此外,p1Ac-GFP大肠杆菌宿主菌株可以发出绿色荧光,培养菌液荧光强度与所表达的Bt蛋白浓度呈较好的线性关系。培养条件优化结果显示,37℃、培养液与装瓶体积比为5/5、48 h的培养时间最适合p1Ac-GFP表达Bt Cry1Ac蛋白。【结论】p1Ac-GFP表达Bt蛋白时可以直接使用菌液荧光强度作为生物活性测定的浓度指示依据,有助于Bt基因杀虫活性功能的快速批量筛选。
张静安, 胡孝龙, 曹蓓蓓, 廖敏, 束长龙, 张杰, 王奎, 操海群. 苏云金芽胞杆菌可视化快速表达载体的构建与特性分析[J]. 生物技术通报, 2025, 41(1): 95-102.
ZHANG Jing-an, HU Xiao-long, CAO Bei-bei, LIAO Min, SHU Chang-long, ZHANG Jie, WANG Kui, CAO Hai-qun. Construction and Characterization of Rapid Visual Expression Vector for Bacillus thuringiensis[J]. Biotechnology Bulletin, 2025, 41(1): 95-102.
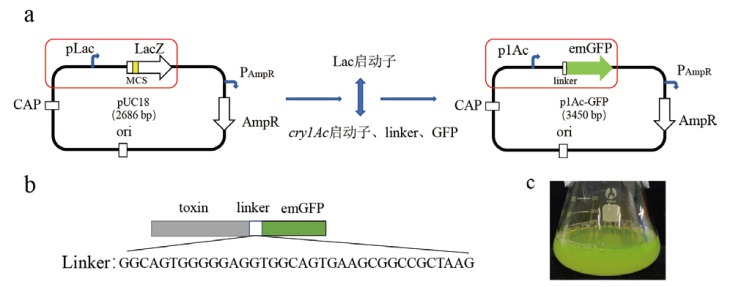
图1 p1Ac-GFP载体的构建图 a:载体构建图;b:linker序列;c:转化TOP10菌液示意图
Fig. 1 Construction diagram of p1Ac-GFP vector a: The vector construction diagram; b: the linker sequence; c: the schematic diagram of transforming TOP10 bacterial solution
| 装液量/锥形瓶体积(V/V) Liquid volume/Volume of the conical flask | 20℃培养时间 Culture time for 20℃/h | 装液量/锥形瓶体积(V/V) Liquid volume/Volume of the conical flask | 30℃培养时间 Culture time for 30℃/h | 装液量/锥形瓶体积(V/V) Liquid volume/Volume of the conical flask | 37℃培养时间Culture time for 37℃/h |
|---|---|---|---|---|---|
| 1/5 | 12 | 1/5 | 12 | 1/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 2/5 | 12 | 2/5 | 12 | 2/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 3/5 | 12 | 3/5 | 12 | 3/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 4/5 | 12 | 4/5 | 12 | 4/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 5/5 | 12 | 5/5 | 12 | 5/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 |
表1 p1Ac-GFP表达条件优化设计
Table 1 Optimized design of p1Ac-GFP expression conditions
| 装液量/锥形瓶体积(V/V) Liquid volume/Volume of the conical flask | 20℃培养时间 Culture time for 20℃/h | 装液量/锥形瓶体积(V/V) Liquid volume/Volume of the conical flask | 30℃培养时间 Culture time for 30℃/h | 装液量/锥形瓶体积(V/V) Liquid volume/Volume of the conical flask | 37℃培养时间Culture time for 37℃/h |
|---|---|---|---|---|---|
| 1/5 | 12 | 1/5 | 12 | 1/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 2/5 | 12 | 2/5 | 12 | 2/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 3/5 | 12 | 3/5 | 12 | 3/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 4/5 | 12 | 4/5 | 12 | 4/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 | |||
| 5/5 | 12 | 5/5 | 12 | 5/5 | 12 |
| 24 | 24 | 24 | |||
| 36 | 36 | 36 | |||
| 48 | 48 | 48 | |||
| 60 | 60 | 60 | |||
| 72 | 72 | 72 |
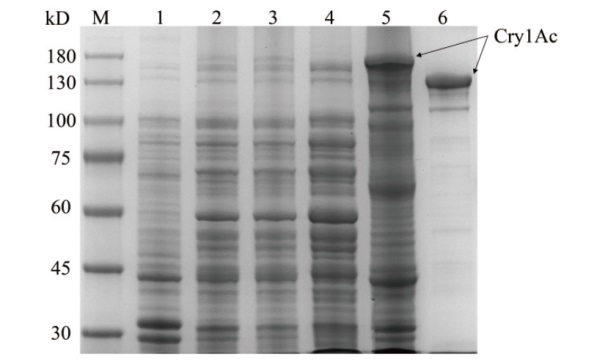
图2 Cry1Ac蛋白的SDS-PAGE分析 M:Marker;1:p1Ac-GFP空载体蛋白(阴性对照);2-5:p1Ac-GFP表达的Cry1Ac蛋白(分别为菌液,超声破碎菌液、上清、沉淀);6:HD73菌株表达的Cry1Ac蛋白(阳性对照)
Fig. 2 SDS-PAGE analysis of Cry1Ac protein M: Marker; 1: p1Ac-GFP empty vector protein(negative control); 2-5: Cry1Ac protein expressed by p1Ac-GFP(bacterial solution, ultrasonic crushing bacterial solution, ultrasonic crushing supernatant, ultrasonic crushing precipitation, respectively); 6: Cry1Ac protein expressed by strain HD73(positive control)
| 蛋白Protein | 校正死亡率±SD Corrected mortality /% |
|---|---|
| p1Ac-GFP空载体蛋白 | 4.60 ± 3.98 b |
| p1Ac-GFP Cry1Ac蛋白 | 100.00 ± 0.00 a |
| HD73 Cry1Ac蛋白 | 96.55 ± 3.45 a |
表2 Cry1Ac蛋白对小菜蛾生物活性测定结果
Table 2 Bioassay of Cry1Ac proteins against P. xylostella
| 蛋白Protein | 校正死亡率±SD Corrected mortality /% |
|---|---|
| p1Ac-GFP空载体蛋白 | 4.60 ± 3.98 b |
| p1Ac-GFP Cry1Ac蛋白 | 100.00 ± 0.00 a |
| HD73 Cry1Ac蛋白 | 96.55 ± 3.45 a |
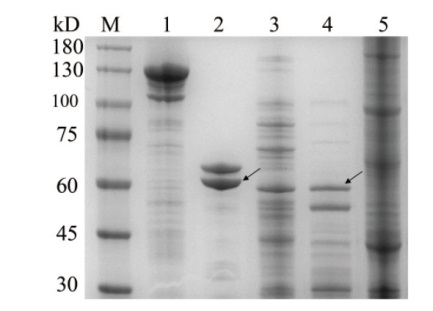
图3 Cry1Ac蛋白的溶解性与酶解性 M:Marker;1:HD73表达的Cry1Ac蛋白(碱溶后可溶组分);2:HD73表达的Cry1Ac蛋白(胰蛋白酶活化);3:p1Ac-GFP表达的Cry1Ac蛋白(碱溶后可溶组分);4:p1Ac-GFP表达的Cry1Ac蛋白(胰蛋白活化);5:p1Ac-GFP表达的Cry1Ac蛋白(碱溶后不可溶组分)
Fig. 3 Solubility and enzymatic hydrolysis of Cry1Ac protein M: Marker; 1: Cry1Ac protein expressed by HD73(soluble components after dissolution); 2: Cry1Ac protein expressed by HD73(trypsin activation); 3: Cry1Ac protein expressed by p1Ac-GFP(soluble components after dissolution); 4: Cry1Ac protein expressed by p1Ac-GFP(trypsin activation); 5: Cry1Ac protein expressed by p1Ac-GFP(insoluble components after dissolution)
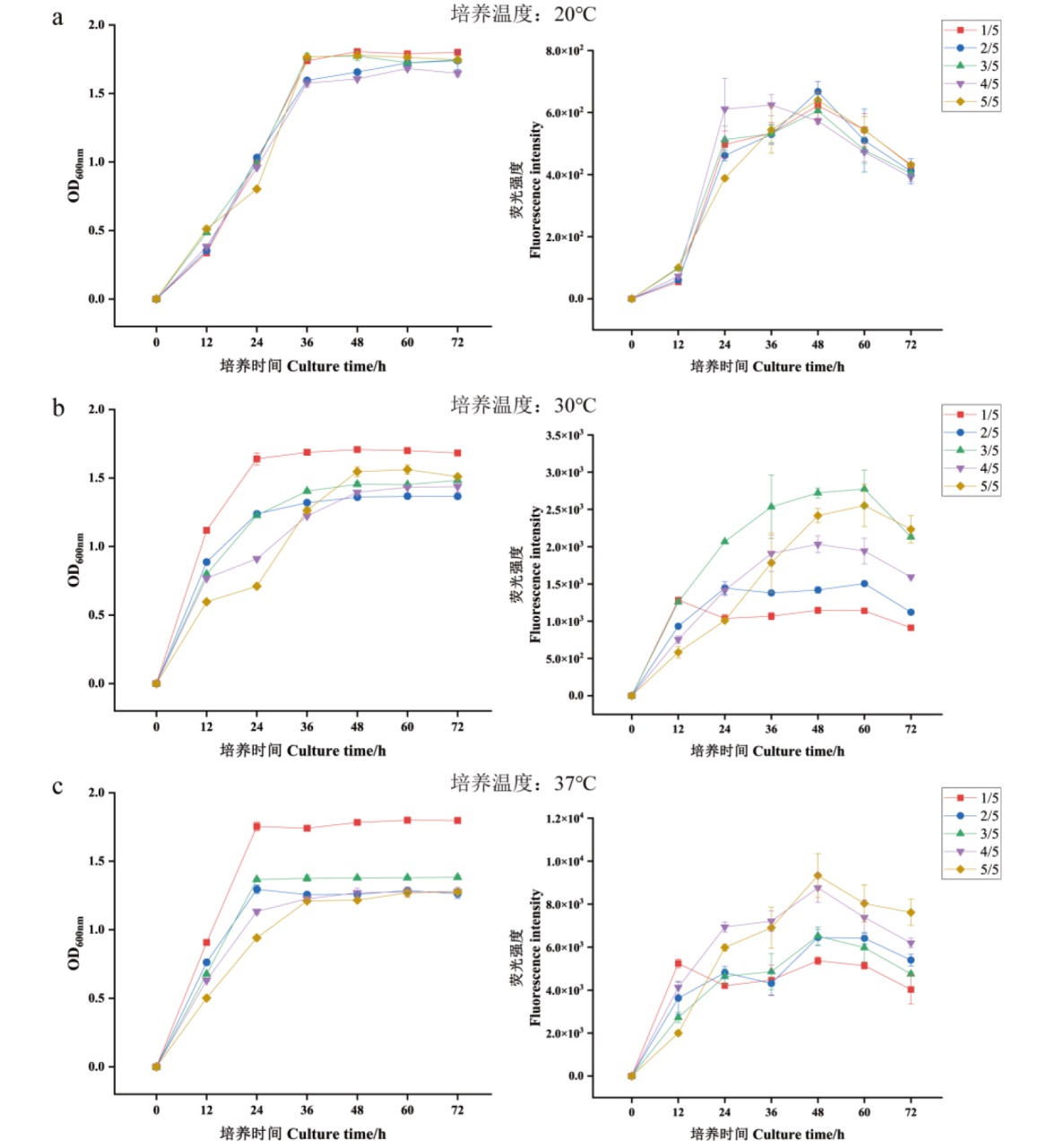
图5 p1Ac-GFP表达Cry1Ac蛋白的条件优化 a:20℃下不同培养时间与装液量对菌液荧光强度与OD600nm的影响;b:30℃下不同培养时间与装液量对菌液荧光强度与OD600nm的影响;c:37℃下不同培养时间与装液量对菌液荧光强度与OD600nm的影响
Fig. 5 Optimization of condition when Cry1Ac expressed by p1Ac-GFP a: The effects of different culture time and bottling amount on fluorescence intensity and OD600nm of bacterial solution at 20℃; b: the effects of different culture time and bottling amount on fluorescence intensity and OD600nm of bacterial solution at 30℃; c: the effects of different culture time and bottling amount on fluorescence intensity and OD600nm of bacterial solution at 37℃
| [1] |
彭琦, 周子珊, 张杰. 苏云金芽胞杆菌杀虫晶体蛋白研究进展[J]. 中国生物防治学报, 2015, 31(5): 712-722.
doi: 10.16409/j.cnki.2095-039x.2015.05.011 |
| Peng Q, Zhou ZS, Zhang J. Research prospects in insecticidal crystal proteins of Bacillus thuringiensis[J]. Chin J Biol Contr, 2015, 31(5): 712-722. | |
| [2] | Caballero J, Jiménez-Moreno N, Orera I, et al. Unraveling the composition of insecticidal crystal proteins in Bacillus thuringiensis: a proteomics approach[J]. Appl Environ Microbiol, 2020, 86(12): e00476-20. |
| [3] | Bravo A, Gill SS, Soberón M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control[J]. Toxicon, 2007, 49(4): 423-435. |
| [4] | Crickmore N, Berry C, Panneerselvam S, et al. A structure-based nomenclature for Bacillus thuringiensis and other bacteria-derived pesticidal proteins[J]. J Invertebr Pathol, 2021, 186: 107438. |
| [5] | Ramalakshmi A, Udayasuriyan V, Balasubramani V. Molecular cloning of a new cry2A-type gene from Bacillus thuringiensis strain Nn10 and its expression studies[J]. Microb Pathog, 2022, 164: 105415. |
| [6] | Liang Z, Ali Q, Wang YJ, et al. Toxicity of Bacillus thuringiensis strains derived from the novel crystal protein Cry31Aa with high nematicidal activity against rice parasitic nematode Aphelenchoides besseyi[J]. Int J Mol Sci, 2022, 23(15): 8189. |
| [7] | Torres-Quintero MC, Arenas-Sosa I, Zuñiga-Navarrete F, et al. Characterization of insecticidal Cry1Cb2 protein from Bacillus thuringiensis toxic to Myzus persicae(Sulzer)[J]. J Invertebr Pathol, 2022, 189: 107731. |
| [8] |
Peng Q, Yu QY, Song FP. Expression of cry genes in Bacillus thuringiensis biotechnology[J]. Appl Microbiol Biotechnol, 2019, 103(4): 1617-1626.
doi: 10.1007/s00253-018-9552-x pmid: 30617537 |
| [9] | 郑文, 叶伟星, 彭东海, 等. 基于cry1Ac表达调控元件的苏云金芽孢杆菌表达载体构建[J]. 湖北农业科学, 2012, 51(2): 400-405. |
| Zheng W, Ye WX, Peng DH, et al. Construction of Bacillus thuringiensis expression vector by using regulatory elements from cry1Ac gene[J]. Hubei Agric Sci, 2012, 51(2): 400-405. | |
| [10] | Wang GJ, Zhang J, Song FP, et al. Engineered Bacillus thuringiensis GO33A with broad insecticidal activity against lepidopteran and coleopteran pests[J]. Appl Microbiol Biotechnol, 2006, 72(5): 924-930. |
| [11] | Kant S, Kapoor R, Banerjee N. Identification of a catabolite-responsive element necessary for regulation of the cry4A gene of Bacillus thuringiensis subsp. israelensis[J]. J Bacteriol, 2009, 191(14): 4687-4692. |
| [12] | 李朝睿, 杜立新, 彭琦, 等. 苏云金芽胞杆菌高效表达载体的构建[J]. 微生物学通报, 40(2): 350-361. |
| Li CR, Du LX, Peng Q, et al. Construction of high-level expression vector for Bacillus thuringiensis[J]. Microbiology China, 40(2): 350-361. | |
| [13] | 杜立新. 苏云金芽胞杆菌cry8E基因转录调控机制研究[D]. 保定: 河北农业大学, 2011. |
| Du LX. Study on transcriptional regulation of cry8E gene of Bacillus thuringiensis[D]. Baoding: Hebei Agricultural University, 2011. | |
| [14] |
Schnepf HE, Wong HC, Whiteley HR. Expression of a cloned Bacillus thuringiensis crystal protein gene in Escherichia coli[J]. J Bacteriol, 1987, 169(9): 4110-4118.
doi: 10.1128/jb.169.9.4110-4118.1987 pmid: 3040677 |
| [15] | 张春鸽, 赵灿, 束长龙, 等. 不同启动子表达Cry1Ie蛋白的特性分析[J]. 生物技术通报, 2012, 28(11): 192-196. |
| Zhang CG, Zhao C, Shu CL, et al. Analysis of expressed Cry1Ie protein initiated by different promoters[J]. Biotechnol Bull, 2012, 28(11): 192-196. | |
| [16] | 马君兰, 束长龙, 刘东明, 等. 大肠杆菌中利用苏云金芽胞杆菌强启动子p1Ac指导Cry1Ac蛋白表达的特性分析[J]. 生物技术通报, 2011, 27(2): 80-84. |
| Ma JL, Shu CL, Liu DM, et al. The nature analysis of expressed Cry1Ac protein initiated by strong promoter p1Ac from Bacillus thuringensis in E. coli[J]. Biotechnol Bull, 2011, 27(2): 80-84. | |
| [17] | Zhou ZS, Yang SJ, Shu CL, et al. Comparison and optimization of the method for Cry1Ac protoxin preparation in HD73 strain[J]. J Integr Agric, 2015, 14(8): 1598-1603. |
| [18] |
Rueden CT, Schindelin J, Hiner MC, et al. ImageJ2: ImageJ for the next generation of scientific image data[J]. BMC Bioinformatics, 2017, 18(1): 529.
doi: 10.1186/s12859-017-1934-z pmid: 29187165 |
| [19] |
王建, 杨小雪, 王丹丹, 等. 对草地贪夜蛾高毒力的苏云金芽胞杆菌菌株筛选与杀虫活性研究[J]. 中国生物防治学报, 2021, 37(4): 660-670.
doi: 10.16409/j.cnki.2095-039x.2021.03.008 |
| Wang J, Yang XX, Wang DD, et al. Screening and insecticidal activity of Bacillus thuringiensis strains with high toxicity against Spodoptera frugiperda[J]. Chin J Biol Contr, 2021, 37(4): 660-670. | |
| [20] |
Agaisse H, Lereclus D. How does Bacillus thuringiensis produce so much insecticidal crystal protein?[J]. J Bacteriol, 1995, 177(21): 6027-6032.
doi: 10.1128/jb.177.21.6027-6032.1995 pmid: 7592363 |
| [21] | Palma L, Muñoz D, Berry C, et al. Bacillus thuringiensis toxins: an overview of their biocidal activity[J]. Toxins, 2014, 6(12): 3296-3325. |
| [22] | Wang K, Shu CL, Zhang J. Effective bacterial insecticidal proteins against coleopteran pests: a review[J]. Arch Insect Biochem Physiol, 2019, 102(3): e21558. |
| [23] |
Baranek J, Banaszak M, Kaznowski A, et al. A novel Bacillus thuringiensis Cry9Ea-like protein with high insecticidal activity towards Cydia pomonella larvae[J]. Pest Manag Sci, 2021, 77(3): 1401-1408.
doi: 10.1002/ps.6157 pmid: 33099864 |
| [24] | Cao BB, Shu CL, Geng LL, et al. Cry78Ba1, one novel crystal protein from Bacillus thuringiensis with high insecticidal activity against rice planthopper[J]. J Agric Food Chem, 2020, 68(8): 2539-2546. |
| [25] | Geng C, Liu YY, Li MM, et al. Dissimilar crystal proteins Cry5Ca1 and Cry5Da1 synergistically act against Meloidogyne incognita and delay Cry5Ba-based nematode resistance[J]. Appl Environ Microbiol, 2017, 83(18): e03505-16. |
| [26] |
Zhang Q, Hua G, Adang MJ. Effects and mechanisms of Bacillus thuringiensis crystal toxins for mosquito larvae[J]. Insect Sci, 2017, 24(5): 714-729.
doi: 10.1111/1744-7917.12401 pmid: 27628909 |
| [27] | Panwar BS, Kaur S. Structural characterization and heterologous expression of a new cyt gene cloned from Bacillus thuringiensis[J]. J Mol Model, 2019, 25(5): 136. |
| [28] |
Bukhari DAA, Shakoori AR. Cloning and expression of Bacillus thuringiensis cry11 crystal protein gene in Escherichia coli[J]. Mol Biol Rep, 2009, 36(7): 1661-1670.
doi: 10.1007/s11033-008-9366-5 pmid: 18821029 |
| [29] | Amadio AF, Navas LE, Sauka DH, et al. Identification, cloning and expression of an insecticide cry8 gene from Bacillus thuringiensis INTA Fr7-4[J]. J Mol Microbiol Biotechnol, 2013, 23(6): 401-409. |
| [30] | Sazhenskiy V, Zaritsky A, Itsko M. Expression in Escherichia coli of the native cyt1Aa from Bacillus thuringiensis subsp. israelensis[J]. Appl Environ Microbiol, 2010, 76(10): 3409-3411. |
| [31] |
Reyaz AL, Arulselvi PI. Cloning, characterization and expression of a novel haplotype cry2A-type gene from Bacillus thuringiensis strain SWK1 native to Himalayan valley Kashmir[J]. J Invertebr Pathol, 2016, 136: 1-6.
doi: 10.1016/j.jip.2016.02.005 pmid: 26906447 |
| [32] | 余宗兰, 贺利业, 龚莉, 等. 苏云金芽胞杆菌cry1Ie新基因的克隆、表达与生物活性测定[J]. 植物保护学报, 2016, 43(2): 201-206. |
| Yu ZL, He LY, Gong L, et al. Cloning, expression, and biological activity of a new cry1Ie gene from Bacillus thuringiensis strain[J]. J Plant Prot, 2016, 43(2): 201-206. | |
| [33] | 张月, 李海涛, 刘荣梅, 等. 苏云金芽胞杆菌cry2Ab34基因的克隆、表达和杀虫活性分析[J]. 生物技术通报, 2017, 33(4): 185-190. |
| Zhang Y, Li HT, Liu RM, et al. Cloning, expression, and insecticidal activities of gene cry2Ab34 from Bacillus thuringiensis[J]. Biotechnol Bull, 2017, 33(4): 185-190. | |
| [34] |
Pardo-López L, Soberón M, Bravo A. Bacillus thuringiensis insecticidal three-domain cry toxins: mode of action, insect resistance and consequences for crop protection[J]. FEMS Microbiol Rev, 2013, 37(1): 3-22.
doi: 10.1111/j.1574-6976.2012.00341.x pmid: 22540421 |
| [35] |
Paulmurugan R, Gambhir SS. Novel fusion protein approach for efficient high-throughput screening of small molecule-mediating protein-protein interactions in cells and living animals[J]. Cancer Res, 2005, 65(16): 7413-7420.
pmid: 16103094 |
| [1] | 饶峻, 赵晨, 李端华, 廖豪, 黄加雨, 王辂. 自诱导策略在麦角硫因生物合成中的应用[J]. 生物技术通报, 2025, 41(1): 333-346. |
| [2] | 王美玲, 耿丽丽, 房瑜, 束长龙, 张杰. 苏云金芽胞杆菌4BM1菌株对油菜菌核病的防治潜力[J]. 生物技术通报, 2024, 40(9): 260-269. |
| [3] | 王玉书, 赵琳琳, 赵爽, 胡琦, 白慧霞, 王欢, 曹业萍, 范震宇. 大白菜BrCYP83B1基因的克隆及表达分析[J]. 生物技术通报, 2024, 40(6): 152-160. |
| [4] | 王周, 余杰, 王金华, 王永泽, 赵筱. 厌氧表达乳酸脱氢酶以提高大肠杆菌产D-乳酸光学纯度[J]. 生物技术通报, 2024, 40(5): 290-299. |
| [5] | 张震, 李清, 徐菁, 陈凯园, 张春芝, 祝光涛. 马铃薯线粒体靶向表达载体的构建与应用[J]. 生物技术通报, 2024, 40(5): 66-73. |
| [6] | 庄棵, 梁至轩, 何英婷, 谢秋玲. 大肠杆菌DH5α通过外膜囊泡传递抗生素抗性基因AmpR[J]. 生物技术通报, 2024, 40(12): 275-281. |
| [7] | 杨红艳, 韩筱, 杨建军. pDNA质粒在一次性生物反应器中的放大生产研究[J]. 生物技术通报, 2024, 40(1): 168-175. |
| [8] | 陈彩萍, 任昊, 龙腾飞, 何冰, 鲁兆祥, 孙坚. 大肠杆菌Nissle 1917对炎症性肠病治疗作用的研究进展[J]. 生物技术通报, 2023, 39(6): 109-118. |
| [9] | 吴莉丹, 冉雪琴, 牛熙, 黄世会, 李升, 王嘉福. 猪源致病性大肠杆菌基因组比较与毒力因子分析[J]. 生物技术通报, 2023, 39(12): 287-299. |
| [10] | 侯炜辰, 叶柯, 李洁, 张洋子, 许文涛, 朱龙佼, 李相阳. 基于抗体-适配体夹心生物传感器检测大肠杆菌O157: H7[J]. 生物技术通报, 2023, 39(12): 81-89. |
| [11] | 李奕雅, 吴一凡, 丁能水, 范小萍, 陈凡. 荧光素酶辅助定量大肠杆菌破碎效果的方法[J]. 生物技术通报, 2023, 39(12): 90-98. |
| [12] | 唐瑞琪, 赵心清, 朱笃, 汪涯. 大肠杆菌对木质纤维素水解液抑制物的胁迫耐受性[J]. 生物技术通报, 2023, 39(11): 205-216. |
| [13] | 李仁瀚, 张乐乐, 刘春立, 刘秀霞, 白仲虎, 杨艳坤, 李业. 基于紫色杆菌素生物合成途径的L-色氨酸生物传感器的构建[J]. 生物技术通报, 2023, 39(10): 80-92. |
| [14] | 高伟欣, 黄火清, 赵晶, 张鑫, 杨宁, 杨浩萌. 应用于基因编辑的核糖核蛋白复合体的构建与活性验证[J]. 生物技术通报, 2022, 38(8): 60-68. |
| [15] | 孙曼銮, 葛赛, 卜佳, 朱壮彦. 大肠杆菌核糖核酸酶调控机制研究[J]. 生物技术通报, 2022, 38(3): 234-245. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||