








• 研究报告 • 下一篇
收稿日期:2025-10-10
出版日期:2026-03-09
通讯作者:
韩丽珍,女,博士,教授,研究方向 :微生物学;E-mail: lzhan1@gzu.edu.cn作者简介:孔兵兵,男,硕士研究生,研究方向 :农业微生物学;E-mail: 1148689145@qq.com
基金资助:
KONG Bing-bing1( ), WANG Qing1, JIANG Biao1, HAN Li-zhen1,2(
), WANG Qing1, JIANG Biao1, HAN Li-zhen1,2( )
)
Received:2025-10-10
Published:2026-03-09
摘要:
目的 探讨根际促生菌(plant-growth promoting rhizobacteria, PGPR)共发酵菌液制备的生物育苗基质对穴盘育苗条件下辣椒幼苗生长的影响及作用机制,为生物育苗基质的研发奠定基础。 方法 通过4株PGPR菌株的兼容性分析、不同菌株组合共发酵液的吲哚乙酸(indoleacetic acid, IAA)含量测定及对辣椒的浸种实验以筛选共发酵菌株;通过穴盘育苗分析共发酵菌液对辣椒种子萌发及幼苗长势的影响以确定生物育苗的菌株组合;通过不同比例共发酵液添加对苗盘期幼苗的生长影响以确定其最适添加量;通过分析共发酵菌液对育苗基质营养物质含量及微生物多样性的影响以解析其促进幼苗生长的机制。 结果 吡咯伯克霍尔德氏菌P10和弯曲芽胞杆菌HGD12菌株的共发酵液含有60.84 mg/L IAA,显著高于2个菌株的单菌培养液,且明显促进了浸种和穴盘育苗条件下辣椒种子的萌发和幼苗的长势。15%共发酵液添加到育苗基质中,对苗盘期幼苗的促生长作用最优,辣椒幼苗的株高、茎粗、鲜重、干重、叶片数、叶面积及叶绿素SPAD值分别显著提高29.17%、17.67%、64.68%、94.44%、22.30%、49.45%及17.83%,基质的有机质、碱解氮及可溶磷含量分别增加了11.31%、20.39%及14.14%。而且,共发酵菌液显著提高了基质的细菌物种丰度并降低了真菌的群落多样性,与基质营养物质含量显著正相关的拟杆菌门、芽单胞菌门、绿弯菌门、嗜盐菌门、罗兹菌门及担子菌门的丰度提高,而与其负相关的变形菌门、子囊菌门、球囊菌门等的丰度降低。 结论 P10和HGD12菌株的共发酵液添加到育苗基质中,可以提高基质的营养物质含量及改变基质的微生物群落结构,显著促进辣椒种子的萌发及苗盘期幼苗的生长。
孔兵兵, 王清, 蒋标, 韩丽珍. 根际促生菌共发酵菌液育苗基质对辣椒育苗的影响[J]. 生物技术通报, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1077.
KONG Bing-bing, WANG Qing, JIANG Biao, HAN Li-zhen. Effects of PGPR Co-fermented Broth Nursery Substrate on the Pepper Seedling[J]. Biotechnology Bulletin, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1077.
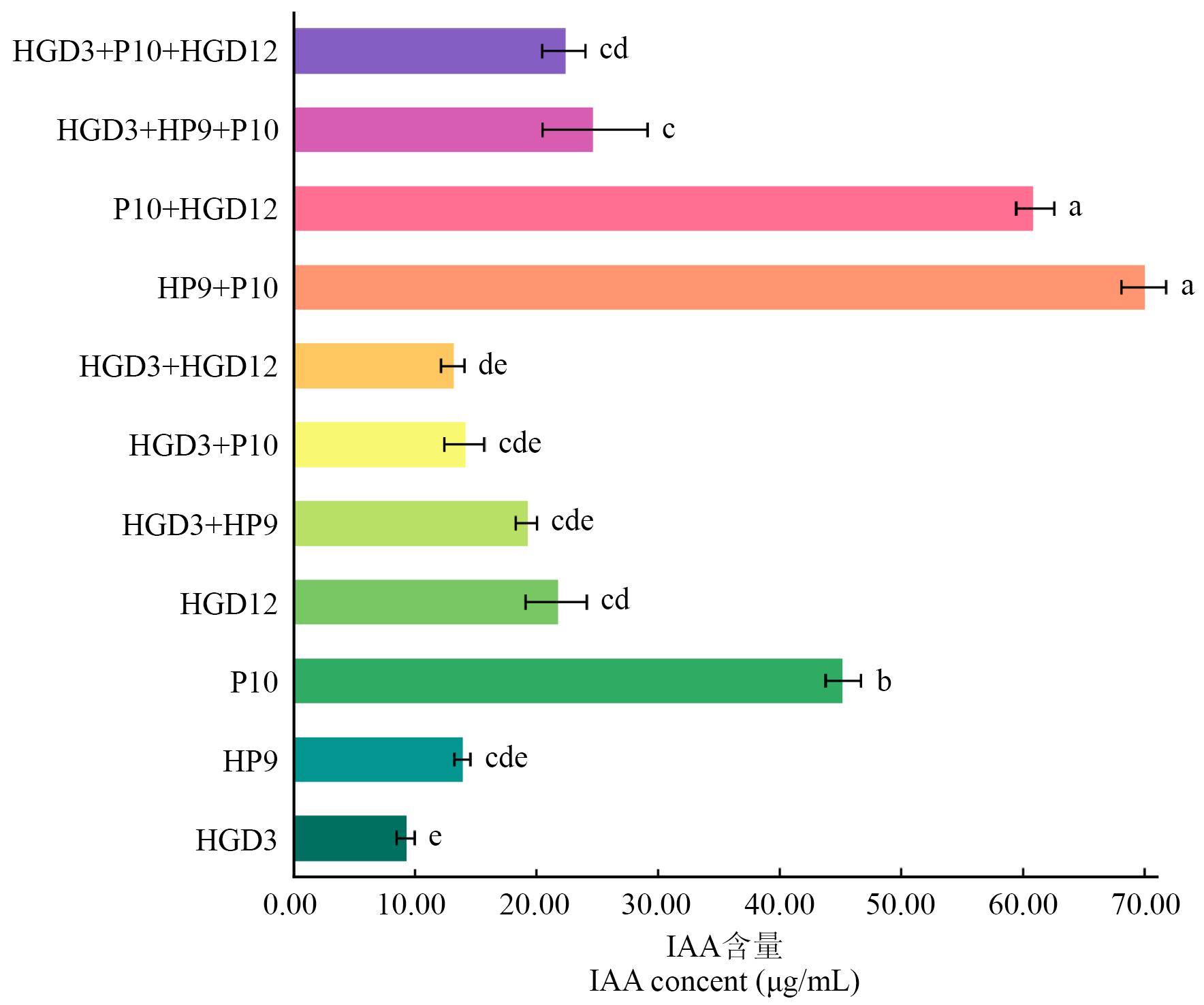
图1 不同PGPR菌株组合共发酵液中的IAA含量小写字母代表处理组间差异显著(P<0.05)
Fig. 1 IAA content in the co-fermented broth of different PGPR strainsLowercase letters indicate significant differences between treatment groups (P<0.05)
指标 Index | 萌发率 Germination rate (%) | 发芽势 Germination force (%) | 发芽指数 Germination index (%) | 活力指数 Vitality index | 根长 Root length (mm) | 茎粗 Stem thick (mm) | 鲜重 Fresh weight (mg) | 子叶展开率 Leaf expansion rate (%) |
|---|---|---|---|---|---|---|---|---|
| CK | 78.89±4.78b | 27.78±4.81c | 56.74±3.11c | 104.89±10.35c | 13.53±0.25bc | 8.60±1.05c | 184.67±11.50b | 18.13±2.25b |
| HP9+P10 | 92.72±2.11a | 36.16±11.41b | 77.48±4.71a | 131.99±4.03ab | 12.70±2.19c | 9.30±0.98b | 170.67±9.02c | 19.82±0.77b |
| P10+HGD12 | 91.69±1.81a | 39.39±2.53a | 71.20±2.25b | 141.22±4.89a | 16.60±1.35a | 10.07±0.57a | 198.33±2.52a | 26.22±1.83a |
表1 不同菌株组合共发酵液浸种对辣椒种子萌发的影响
Table 1 Effects on the germination of pepper seed soaked with co-fermented brothof different PGPR strains
指标 Index | 萌发率 Germination rate (%) | 发芽势 Germination force (%) | 发芽指数 Germination index (%) | 活力指数 Vitality index | 根长 Root length (mm) | 茎粗 Stem thick (mm) | 鲜重 Fresh weight (mg) | 子叶展开率 Leaf expansion rate (%) |
|---|---|---|---|---|---|---|---|---|
| CK | 78.89±4.78b | 27.78±4.81c | 56.74±3.11c | 104.89±10.35c | 13.53±0.25bc | 8.60±1.05c | 184.67±11.50b | 18.13±2.25b |
| HP9+P10 | 92.72±2.11a | 36.16±11.41b | 77.48±4.71a | 131.99±4.03ab | 12.70±2.19c | 9.30±0.98b | 170.67±9.02c | 19.82±0.77b |
| P10+HGD12 | 91.69±1.81a | 39.39±2.53a | 71.20±2.25b | 141.22±4.89a | 16.60±1.35a | 10.07±0.57a | 198.33±2.52a | 26.22±1.83a |
指标 Index | 萌发率 Germination rate (%) | 成活率 Survival rate (%) | 叶片数 Leaf number | 叶面积 Leaf area (cm2) | 株高 Plant height (cm) | 茎粗 Stem diameter (cm) | 根长 Root length (cm) | 鲜重 Fresh weight (g) | 干重 Dry weight (g) | 壮苗指数 Strong seedling index | 叶绿素(SPAD值) Chlorophyll (SPAD) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CK | 65.28±4.81c | 62.50±8.33c | 4.50±0.55b | 6.89±1.92b | 3.42±0.80b | 1.33±0.17b | 10.13±1.18ab | 0.45±0.12b | 0.04±0.02b | 0.02±0.01b | 16.64±1.55c |
| HP9+P10 | 72.22±12.03b | 75.00±14.43b | 4.83±0.41b | 6.31±1.67b | 3.27±0.20b | 1.28±0.07b | 8.80±1.93c | 0.40±0.06b | 0.04±0.01b | 0.01±0.00b | 19.28±1.09b |
| P10+HGD12 | 93.06±4.81a | 83.33±4.17a | 7.33±0.52a | 16.99±2.90a | 6.55±0.49a | 2.20±0.13a | 11.10±2.55a | 1.37±0.11a | 0.15±0.02a | 0.05±0.01a | 32.51±1.25a |
表2 不同生物育苗基质对辣椒穴盘育苗的影响
Table 2 Effects of different bio-nursery substrates on pepper seedling in hole tray
指标 Index | 萌发率 Germination rate (%) | 成活率 Survival rate (%) | 叶片数 Leaf number | 叶面积 Leaf area (cm2) | 株高 Plant height (cm) | 茎粗 Stem diameter (cm) | 根长 Root length (cm) | 鲜重 Fresh weight (g) | 干重 Dry weight (g) | 壮苗指数 Strong seedling index | 叶绿素(SPAD值) Chlorophyll (SPAD) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CK | 65.28±4.81c | 62.50±8.33c | 4.50±0.55b | 6.89±1.92b | 3.42±0.80b | 1.33±0.17b | 10.13±1.18ab | 0.45±0.12b | 0.04±0.02b | 0.02±0.01b | 16.64±1.55c |
| HP9+P10 | 72.22±12.03b | 75.00±14.43b | 4.83±0.41b | 6.31±1.67b | 3.27±0.20b | 1.28±0.07b | 8.80±1.93c | 0.40±0.06b | 0.04±0.01b | 0.01±0.00b | 19.28±1.09b |
| P10+HGD12 | 93.06±4.81a | 83.33±4.17a | 7.33±0.52a | 16.99±2.90a | 6.55±0.49a | 2.20±0.13a | 11.10±2.55a | 1.37±0.11a | 0.15±0.02a | 0.05±0.01a | 32.51±1.25a |
不同处理 Different treatment | 添加比例 Addition ratio (%) | 株高 Plant height (cm) | 茎粗 Stem diameter (mm) | 鲜重 Plant weight (g) | 干重 Dry weight (g) | 叶片数 Leaf number | 叶面积 Leaf area (cm2) | 叶绿素SPADC hlorophyll (SPAD) |
|---|---|---|---|---|---|---|---|---|
| CK | 12.48±0.85fg | 2.49±0.12gh | 2.01±0.25ghi | 0.18±0.02h | 7.67±0.98fg | 23.48±3.59f | 31.12±1.33k | |
| LB | 5 | 12.44±1.30fg | 2.64±0.27defh | 2.25±0.13fg | 0.20±0.03gh | 7.75±1.22defg | 28.35±2.45de | 32.74±1.70j |
| 10 | 12.22±1.11fg | 2.35±0.19hi | 1.91±0.35ghij | 0.18±0.04h | 7.25±0.87gh | 22.54±3.17f | 33.19±1.71ij | |
| 15 | 12.36±0.65fg | 2.32±0.17i | 1.75±0.15ij | 0.17±0.03h | 6.88±0.60h | 22.07±2.88f | 34.99±2.20fgh | |
| 20 | 10.79±0.78h | 2.56±0.24g | 1.89±0.34hij | 0.16±0.03h | 7.71±0.84efg | 21.20±1.75f | 35.43±1.32defgh | |
| 25 | 8.22±1.42i | 2.34±0.15hi | 1.62±0.33j | 0.16±0.04h | 7.38±0.69gh | 21.95±3.94f | 34.49±2.03ghi | |
| P10 | 5 | 16.42±0.28b | 2.73±0.05cdef | 2.78±0.08cd | 0.26±0.01def | 8.58±0.34bcd | 28.88±0.68cde | 34.48±0.33ghi |
| 10 | 14.76±0.22de | 2.62±0.02efg | 2.60±0.07de | 0.25±0.01def | 8.42±0.19cdef | 33.35±1.06ab | 34.05±0.36hi | |
| 15 | 14.89±0.32de | 2.80±0.05bcd | 2.88±0.13cd | 0.29±0.01cde | 8.92±0.23abc | 33.26±1.16ab | 35.11±0.3efgh | |
| 20 | 14.88±0.71de | 2.85±0.08bc | 2.74±0.14cde | 0.30±0.02bcd | 8.58±0.26bcd | 35.48±1.59a | 35.74±0.32cdefg | |
| 25 | 14.56±0.32e | 2.60±0.04fg | 2.80±0.16cd | 0.27±0.02def | 8.50±0.26cde | 34.14±1.04ab | 35.86±0.36cdefg | |
| HGD12 | 5 | 14.68±0.47de | 2.77±0.06bcdef | 2.79±0.11cd | 0.27±0.01def | 8.92±0.23abc | 32.52±1.27abc | 34.58±0.26gh |
| 10 | 15.5±0.33bcde | 2.76±0.04bcdef | 2.95±0.15cd | 0.28±0.02def | 8.75±0.28abc | 32.33±1.43abcd | 36.89±0.73abc | |
| 15 | 15.23±0.38cde | 2.78±0.04bcde | 2.87±0.17cd | 0.26±0.01def | 8.25±0.25cdef | 31.76±1.21abcd | 35.89±0.62cdefg | |
| 20 | 13.21±0.30f | 2.77±0.06bcdef | 2.40±0.10ef | 0.24±0.01fg | 8.42±0.29cdef | 27.84±1.26e | 36.39±0.37cdef | |
| 25 | 11.75±0.36gh | 2.61±0.05efg | 2.17±0.09fgh | 0.24±0.01ef | 8.50±0.29cde | 27.40±0.88e | 36.48±0.37bcde | |
| P10+HGD12 | 5 | 17.88±2.35a | 3.02±0.20a | 3.11±0.62bc | 0.34±0.09ab | 9.50±1.24a | 32.08±6.49abcd | 36.55±1.20bcd |
| 10 | 17.79±1.46a | 3.03±0.28a | 3.52±0.38a | 0.36±0.04a | 9.46±0.66a | 35.32±4.27a | 36.35±1.53cdef | |
| 15 | 16.12±1.22bc | 3.05±0.21ab | 3.31±0.45ab | 0.35±0.04a | 9.38±0.51ab | 35.09±5.49a | 36.67±1.59bcd | |
| 20 | 15.78±1.10bcd | 2.88±0.16abc | 2.82±0.12cd | 0.32±0.07abc | 8.58±0.90bcd | 30.98±5.26bcde | 37.82±1.01ab | |
| 25 | 12.64±0.79fg | 2.81±0.13bcd | 2.65±0.39de | 0.27±0.04def | 8.75±0.75abc | 30.68±6.12bcde | 38.01±1.14a |
表3 不同施加比例的P10+HGD12共发酵液育苗基质对辣椒穴盘育苗的影响
Table 3 Effects of different proportion of P10+HGD12 co-fermented broth on pepper seedling cultivation in hole tray
不同处理 Different treatment | 添加比例 Addition ratio (%) | 株高 Plant height (cm) | 茎粗 Stem diameter (mm) | 鲜重 Plant weight (g) | 干重 Dry weight (g) | 叶片数 Leaf number | 叶面积 Leaf area (cm2) | 叶绿素SPADC hlorophyll (SPAD) |
|---|---|---|---|---|---|---|---|---|
| CK | 12.48±0.85fg | 2.49±0.12gh | 2.01±0.25ghi | 0.18±0.02h | 7.67±0.98fg | 23.48±3.59f | 31.12±1.33k | |
| LB | 5 | 12.44±1.30fg | 2.64±0.27defh | 2.25±0.13fg | 0.20±0.03gh | 7.75±1.22defg | 28.35±2.45de | 32.74±1.70j |
| 10 | 12.22±1.11fg | 2.35±0.19hi | 1.91±0.35ghij | 0.18±0.04h | 7.25±0.87gh | 22.54±3.17f | 33.19±1.71ij | |
| 15 | 12.36±0.65fg | 2.32±0.17i | 1.75±0.15ij | 0.17±0.03h | 6.88±0.60h | 22.07±2.88f | 34.99±2.20fgh | |
| 20 | 10.79±0.78h | 2.56±0.24g | 1.89±0.34hij | 0.16±0.03h | 7.71±0.84efg | 21.20±1.75f | 35.43±1.32defgh | |
| 25 | 8.22±1.42i | 2.34±0.15hi | 1.62±0.33j | 0.16±0.04h | 7.38±0.69gh | 21.95±3.94f | 34.49±2.03ghi | |
| P10 | 5 | 16.42±0.28b | 2.73±0.05cdef | 2.78±0.08cd | 0.26±0.01def | 8.58±0.34bcd | 28.88±0.68cde | 34.48±0.33ghi |
| 10 | 14.76±0.22de | 2.62±0.02efg | 2.60±0.07de | 0.25±0.01def | 8.42±0.19cdef | 33.35±1.06ab | 34.05±0.36hi | |
| 15 | 14.89±0.32de | 2.80±0.05bcd | 2.88±0.13cd | 0.29±0.01cde | 8.92±0.23abc | 33.26±1.16ab | 35.11±0.3efgh | |
| 20 | 14.88±0.71de | 2.85±0.08bc | 2.74±0.14cde | 0.30±0.02bcd | 8.58±0.26bcd | 35.48±1.59a | 35.74±0.32cdefg | |
| 25 | 14.56±0.32e | 2.60±0.04fg | 2.80±0.16cd | 0.27±0.02def | 8.50±0.26cde | 34.14±1.04ab | 35.86±0.36cdefg | |
| HGD12 | 5 | 14.68±0.47de | 2.77±0.06bcdef | 2.79±0.11cd | 0.27±0.01def | 8.92±0.23abc | 32.52±1.27abc | 34.58±0.26gh |
| 10 | 15.5±0.33bcde | 2.76±0.04bcdef | 2.95±0.15cd | 0.28±0.02def | 8.75±0.28abc | 32.33±1.43abcd | 36.89±0.73abc | |
| 15 | 15.23±0.38cde | 2.78±0.04bcde | 2.87±0.17cd | 0.26±0.01def | 8.25±0.25cdef | 31.76±1.21abcd | 35.89±0.62cdefg | |
| 20 | 13.21±0.30f | 2.77±0.06bcdef | 2.40±0.10ef | 0.24±0.01fg | 8.42±0.29cdef | 27.84±1.26e | 36.39±0.37cdef | |
| 25 | 11.75±0.36gh | 2.61±0.05efg | 2.17±0.09fgh | 0.24±0.01ef | 8.50±0.29cde | 27.40±0.88e | 36.48±0.37bcde | |
| P10+HGD12 | 5 | 17.88±2.35a | 3.02±0.20a | 3.11±0.62bc | 0.34±0.09ab | 9.50±1.24a | 32.08±6.49abcd | 36.55±1.20bcd |
| 10 | 17.79±1.46a | 3.03±0.28a | 3.52±0.38a | 0.36±0.04a | 9.46±0.66a | 35.32±4.27a | 36.35±1.53cdef | |
| 15 | 16.12±1.22bc | 3.05±0.21ab | 3.31±0.45ab | 0.35±0.04a | 9.38±0.51ab | 35.09±5.49a | 36.67±1.59bcd | |
| 20 | 15.78±1.10bcd | 2.88±0.16abc | 2.82±0.12cd | 0.32±0.07abc | 8.58±0.90bcd | 30.98±5.26bcde | 37.82±1.01ab | |
| 25 | 12.64±0.79fg | 2.81±0.13bcd | 2.65±0.39de | 0.27±0.04def | 8.75±0.75abc | 30.68±6.12bcde | 38.01±1.14a |

图2 生物育苗基质上穴盘育苗辣椒的生长状况A:不同浓度共发酵菌液育苗基质上的幼苗状况;B:不同菌株发酵液处理基质上15%添加比例的幼苗状况。其中5%、10%、15%、20%和25%分别代表添加不同比例P10+HGD12共发酵液处理组,CK-H2O及CK-LB分别代表水对照处理组和LB对照处理组,P10+HGD12、P10及HGD12分别代表15%比例的共发酵液、P10菌株菌液及HGD12菌液处理组
Fig. 2 Growth of pepper seedlings on the bio-nursery substrate using hole trayA: Seedling growth on substrates with different concentration of co-fermented broth. B: Seedling on substrates treated with fermented broth from different strains at a 15% inoculation ratio. 5%, 10%, 15%, 20% and 25% refers to the treatment group with different concentrations of P10+HGD12. CK-H2O and CK-LB is the control with water and the control with 15% LB, respectively. P10+HGD12, P10 and HGD12 refers to the treatment group with 15% P10+HGD12 co-fermented broth, P10 broth and HGD12 broth, respectively
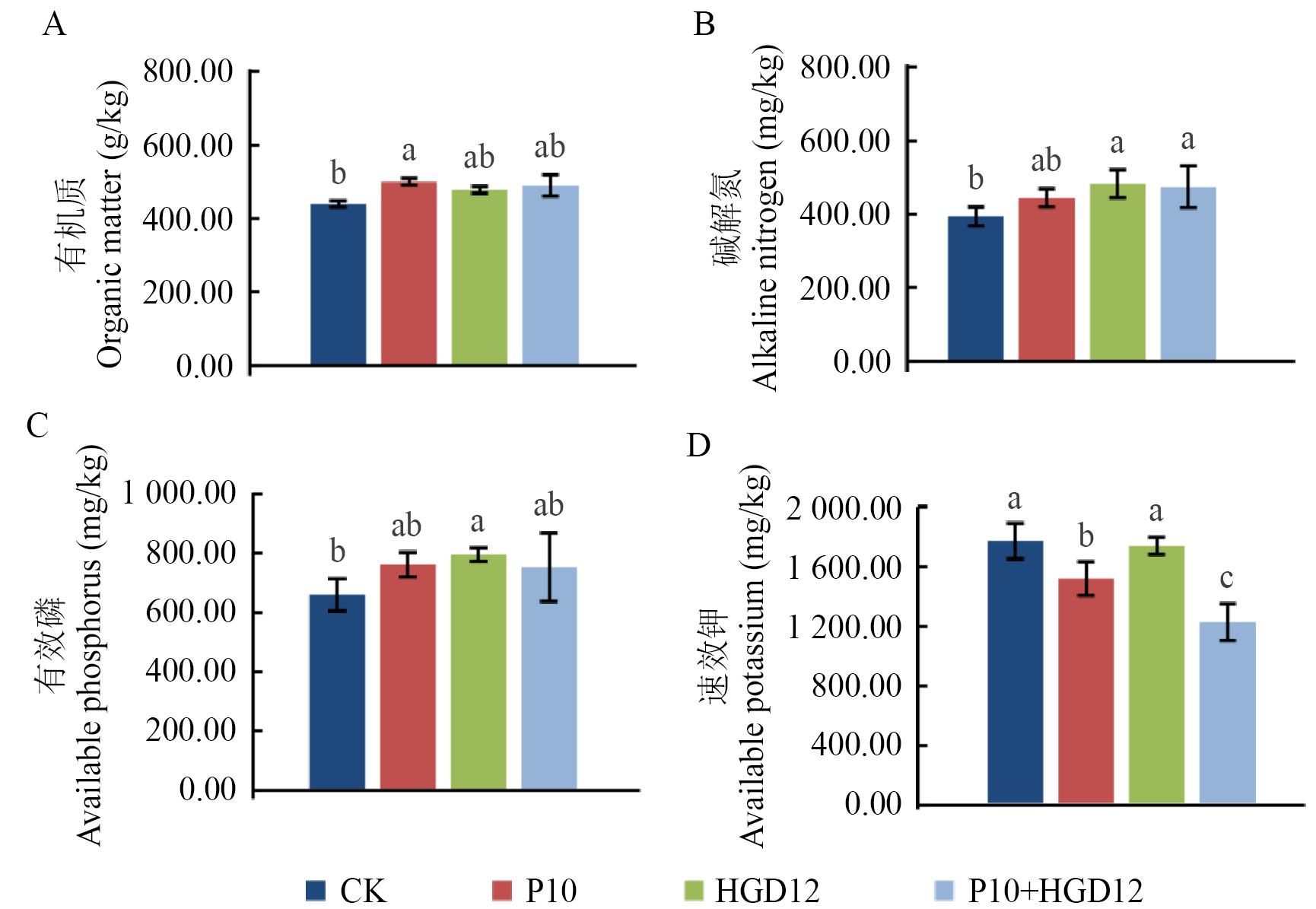
图3 不同处理育苗基质的养分含量P10、HGD12和P10+HGD12分别代表育苗基质中添加的15%比例的菌株发酵液;CK代表水为对照。图中小写字母分别代表4个处理在每一个测定指标上的差异显著性(P<0.05)
Fig. 3 Nutrient content of nursery substrate under different treatmentsP10, HGD12, and P10+HGD12 indicates the 15% proportion of fermented broth added to the nursery substrate, respectively; CK is the control with water. Lowercase letter indicates the significant differences of each index between these four treatments (P<0.05), respectively
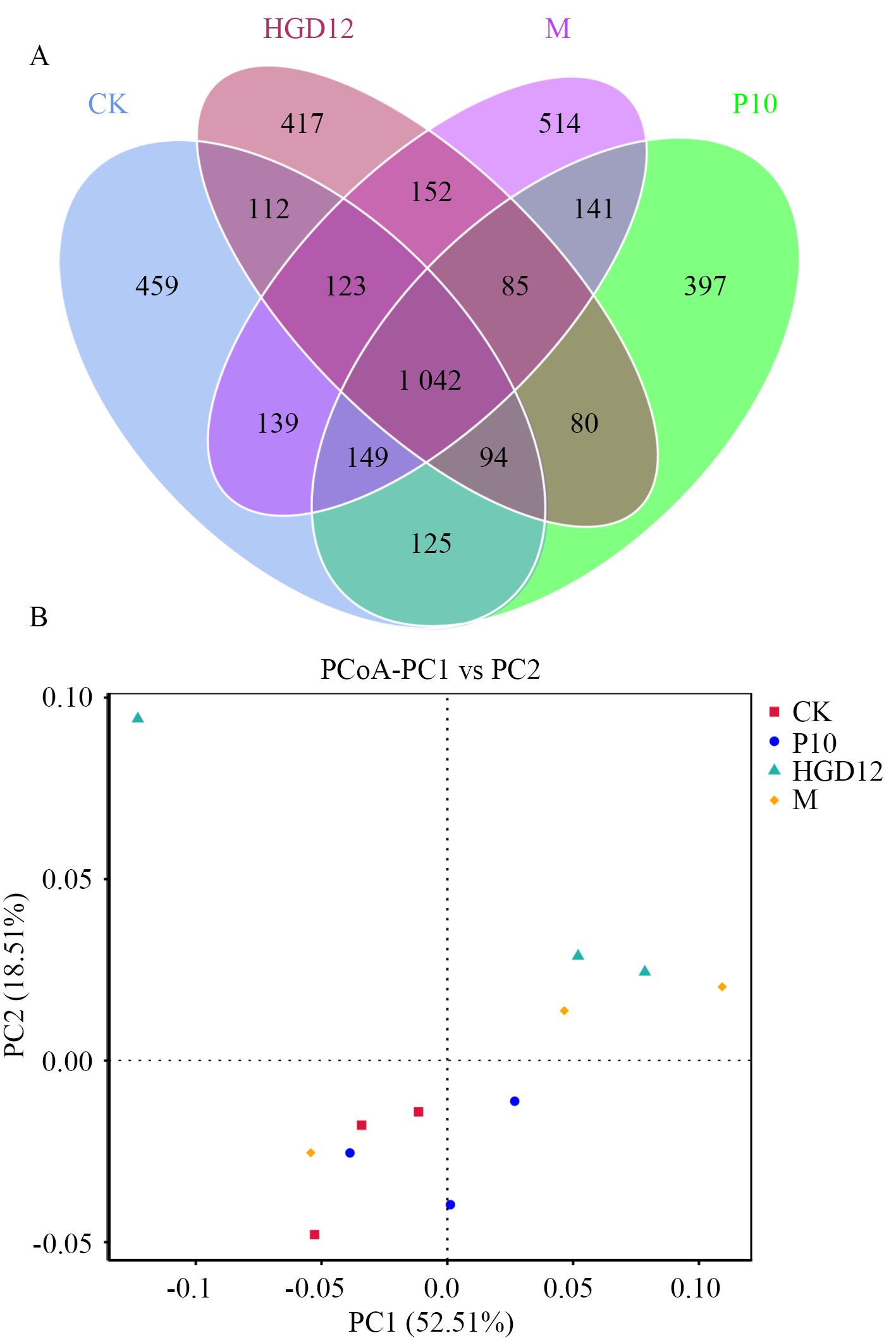
图4 不同处理基质细菌ASV的韦恩图(A)及PCoA分析(B)CK:对照;P10、HGD12分别代表P10或HGD12单菌添加组;M:P10+HGD12共发酵液添加组;下同
Fig. 4 Venn diagram (A) and PCoA analysis (B) of bacterial ASVs under different treatment substratesCK: Control. P10, HGD12 refers to the treatment of P10 and HGD12 strain, respectively. M: Treatment of P10+HGD12 co-fermented broth. The same below
| 多样性指数 Diversity index | Observed OTUs | Chao1 | Pielou | Shannon | Simpson | Coverage (%) |
|---|---|---|---|---|---|---|
| CK | 1 390.33±38.07a | 1 395.60±37.93a | 0.84±0.01a | 8.77±0.04a | 0.99±0.00a | 99.99 |
| P10 | 1 280.67±74.19bc | 1 283.94±75.44bc | 0.84±0.01a | 8.64±0.20a | 0.99±0.00a | 100.00 |
| HGD12 | 1 234.33±27.39c | 1 238.43±28.19c | 0.82±0.02a | 8.47±0.15a | 0.99±0.00a | 99.99 |
| P10+HGD12 | 1 377.00±37.40ab | 1 384.05±44.98ab | 0.83±0.01a | 8.63±0.17a | 0.99±0.00a | 99.99 |
表4 各处理基质中细菌群落的Alpha多样性指数
Table 4 Alpha diversity index of bacterial community in the substrate of each treatment
| 多样性指数 Diversity index | Observed OTUs | Chao1 | Pielou | Shannon | Simpson | Coverage (%) |
|---|---|---|---|---|---|---|
| CK | 1 390.33±38.07a | 1 395.60±37.93a | 0.84±0.01a | 8.77±0.04a | 0.99±0.00a | 99.99 |
| P10 | 1 280.67±74.19bc | 1 283.94±75.44bc | 0.84±0.01a | 8.64±0.20a | 0.99±0.00a | 100.00 |
| HGD12 | 1 234.33±27.39c | 1 238.43±28.19c | 0.82±0.02a | 8.47±0.15a | 0.99±0.00a | 99.99 |
| P10+HGD12 | 1 377.00±37.40ab | 1 384.05±44.98ab | 0.83±0.01a | 8.63±0.17a | 0.99±0.00a | 99.99 |
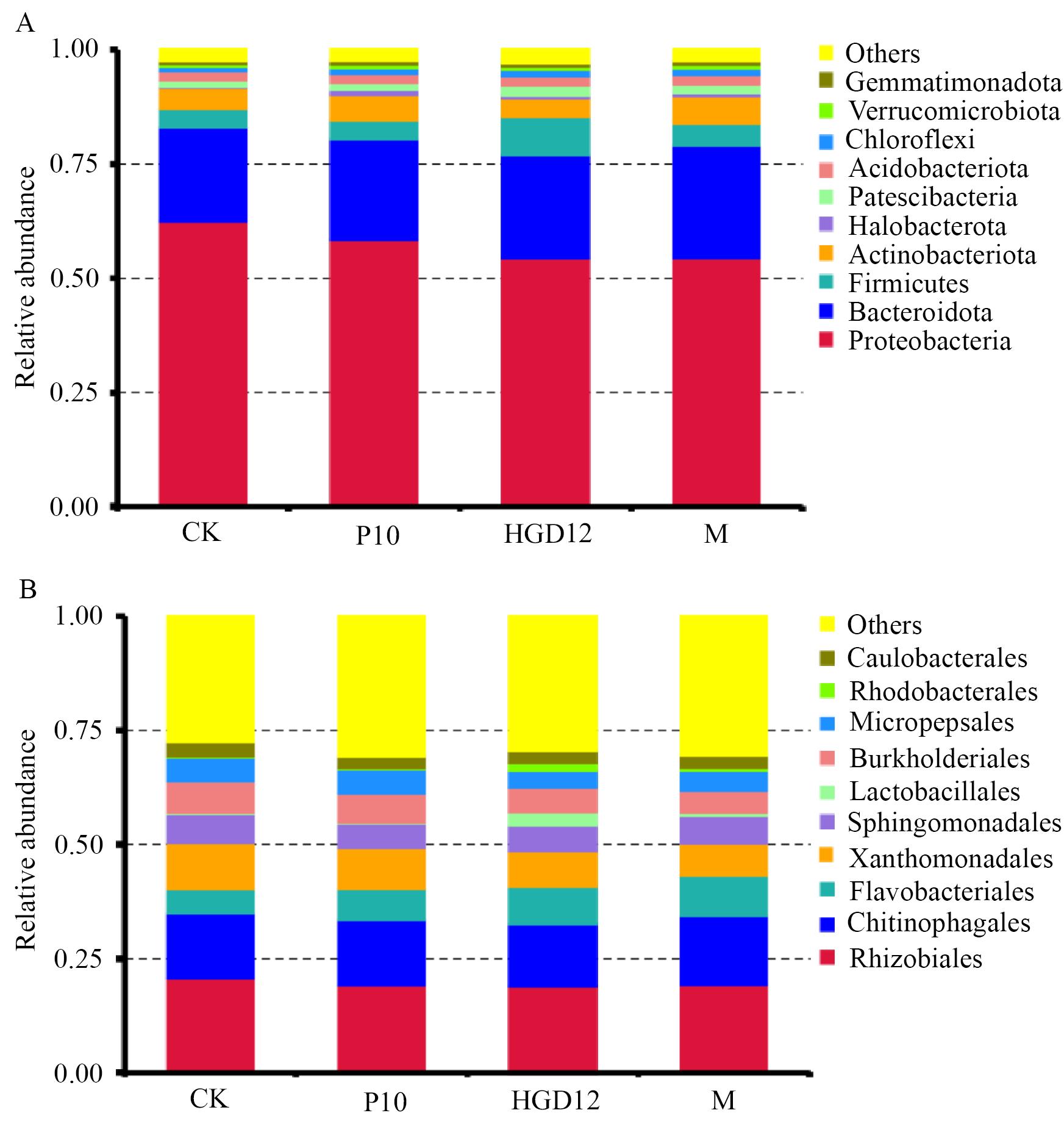
图5 不同处理下基质群落丰度排名前10的细菌类群A:门水平;B:目水平
Fig. 5 The top 10 bacterial community in the abundance ranking of different treatmentsA: Phylum level. B: Order level
| 多样性指数 Diversity index | Observed OTUs | Chao1 | Pielou | Shannon | Simpson | Coverage (%) |
|---|---|---|---|---|---|---|
| CK | 385.33±23.46a | 387.64±22.99a | 0.63±0.01a | 5.41±0.15a | 0.94±0.01a | 100 |
| P10 | 355.33±70.15a | 355.84±70.82a | 0.59±0.03a | 5.03±0.25a | 0.92±0.01ab | 100 |
| HGD12 | 374.67±84.63a | 374.94±84.84a | 0.58±0.03a | 4.94±0.25a | 0.88±0.01b | 100 |
| P10+HGD12 | 373.67±22.81a | 373.90±22.68a | 0.59±0.04a | 5.03±0.18a | 0.9±0.01b | 100 |
表5 各处理基质中真菌群落的Alpha多样性指数
Table 5 Alpha diversity index of fungal community in the substrate of each treatment
| 多样性指数 Diversity index | Observed OTUs | Chao1 | Pielou | Shannon | Simpson | Coverage (%) |
|---|---|---|---|---|---|---|
| CK | 385.33±23.46a | 387.64±22.99a | 0.63±0.01a | 5.41±0.15a | 0.94±0.01a | 100 |
| P10 | 355.33±70.15a | 355.84±70.82a | 0.59±0.03a | 5.03±0.25a | 0.92±0.01ab | 100 |
| HGD12 | 374.67±84.63a | 374.94±84.84a | 0.58±0.03a | 4.94±0.25a | 0.88±0.01b | 100 |
| P10+HGD12 | 373.67±22.81a | 373.90±22.68a | 0.59±0.04a | 5.03±0.18a | 0.9±0.01b | 100 |
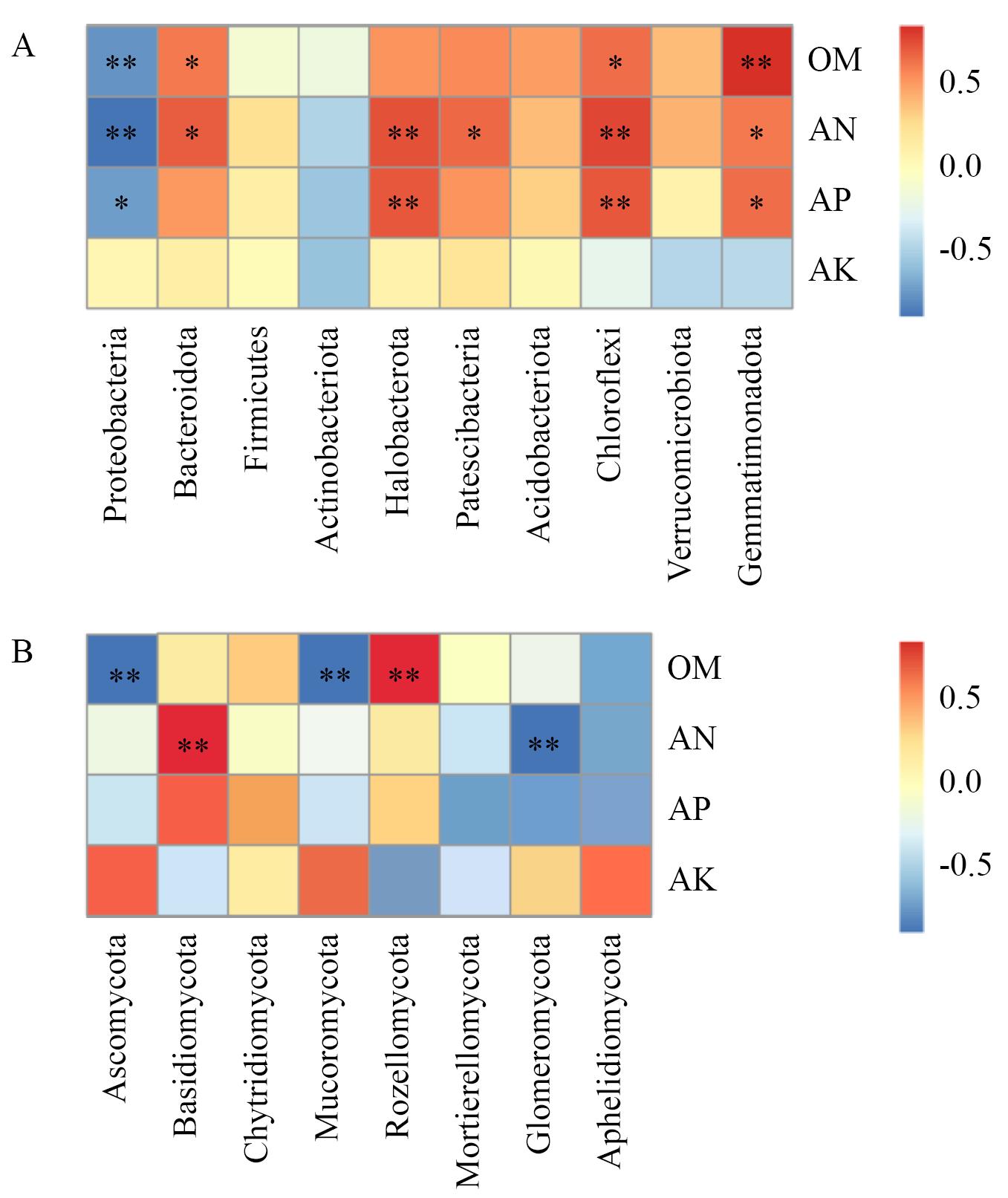
图8 环境因子与微生物门在不同处理的Spearman分析A:细菌;B:真菌;OM、AN、AP和AK分别代表有机质、碱解氮、有效磷和速效钾;*P<0.05,**P<0.01
Fig. 8 Spearman analysis of environmental factors and microbial phylum at different treatmentsA: Bacterial. B: Fungal. OM, AN, AP and AK refers to organic matter, alkaline-hydrolyzed nitrogen, available phosphorus and available potassium, respectively. * P<0.05, ** P<0.01
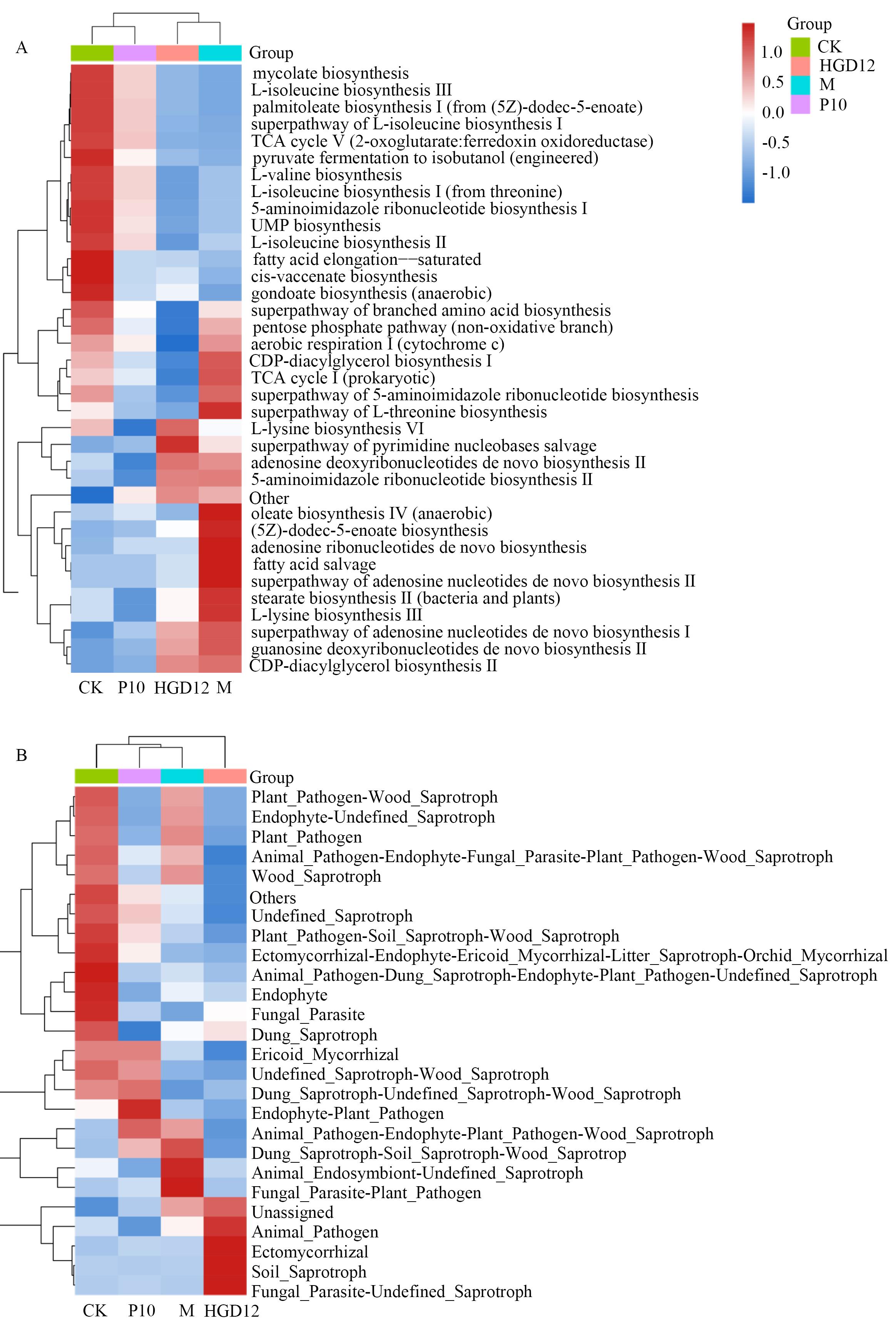
图9 根际微生物群落功能注释聚类热图A:细菌群落;B:真菌群落。热图中对应的值为每一行功能信息的相对丰度经过标准化处理后得到的Z值,红色表示高丰度、蓝色表示低丰度
Fig. 9 Cluster heatmap of functional annotation of rhizosphere microbial communitiesA: Bacterial community. B: Fungal community. The corresponding values in the heatmap are the Z values obtained by standardizing the relative abundance of each row of functional information, with red indicating high abundance and blue indicating low abundance
| [1] | 邹学校, 胡博文, 熊程, 等. 中国辣椒育种60年回顾与展望 [J]. 园艺学报, 2022, 49(10): 2099-2118. |
| Zou XX, Hu BW, Xiong C, et al. Review and prospects of pepper breeding for the past 60 years in China [J]. Acta Hortic Sin, 2022, 49(10): 2099-2118. | |
| [2] | Folta KM. Breeding new varieties for controlled environments [J]. Plant Biol, 2019, 21(S1): 6-12. |
| [3] | Sarangi SK, Maji B, Singh S, et al. Improved nursery management further enhances the productivity of stress-tolerant rice varieties in coastal rainfed Lowlands [J]. Field Crops Res, 2015, 174: 61-70. |
| [4] | Zhang AA, Shang QM. Transcriptome analysis of early lateral root formation in tomato [J]. Plants, 2024, 13(12): 1620. |
| [5] | Liu Y, Ren XX, Jeong BR. Night temperature affects the growth, metabolism, and photosynthetic gene expression in Astragalus membranaceus and Codonopsis lanceolata plug seedlings [J]. Plants, 2019, 8(10): 407. |
| [6] | Park EW, Hwang JH, Hwang HS, et al. Effect of temperature and seedling cultivation period on the quality of plug seedlings of four medicinal plant [J]. Hortic Environ Biotechnol, 2025, 66(2): 319-330. |
| [7] | Epelbaum A, Pearce CM, Yuan S, et al. Effects of stocking density and substratum on the survival, growth, burrowing behaviour and shell morphology of juvenile basket cockle, Clinocardium nuttallii: implications for nursery seed production and field outplanting [J]. Aquac Res, 2011, 42(7): 975-986. |
| [8] | Wu YQ, Chen RZ, Wang FL, et al. Enhanced seedling growth effect of cocopeat by supplementation with earthworm manure as nutrient amendment [J]. Pol J Environ Stud, 2025, 34(4): 3853-3861. |
| [9] | Álvarez-Pérez JM, González-García S, Cobos R, et al. Use of endophytic and rhizosphere Actinobacteria from grapevine plants to reduce nursery fungal graft infections that lead to young grapevine decline [J]. Appl Environ Microbiol, 2017, 83(24): e01564-17. |
| [10] | Li XW, Xia HT, Wang JW, et al. Nutrient uptake and assimilation in fragrant rosewood (Dalbergia odorifera T.C. Chen) seedlings in growing media with un-composted spent mushroom residue [J]. PLoS One, 2021, 16(4): e0249534. |
| [11] | 李瑞敏, 商美妮, 李荣, 等. 木霉生物育苗基质中功能菌数量对辣椒促生效应的影响 [J]. 南京农业大学学报, 2021, 44(5): 869-875. |
| Li RM, Shang MN, Li R, et al. Effects of the amount of Trcichoderma in the bio-nursey substrates on the growth promotion of pepper [J]. J Nanjing Agric Univ, 2021, 44(5): 869-875. | |
| [12] | Prodhan MY, Rahman MB, Rahman A, et al. Characterization of growth-promoting activities of consortia of chlorpyrifos mineralizing endophytic bacteria naturally harboring in rice plants-a potential bio-stimulant to develop a safe and sustainable agriculture [J]. Microorganisms, 2023, 11(7): 1821. |
| [13] | Shahid MA, Balal RM, Khan N, et al. Selenium impedes cadmium and arsenic toxicity in potato by modulating carbohydrate and nitrogen metabolism [J]. Ecotoxicol Environ Saf, 2019, 180: 588-599. |
| [14] | 戚秀秀, 魏畅, 刘晓丹, 等. 根际促生菌应用于基质对水稻幼苗生长的影响 [J]. 土壤, 2020, 52(5): 1025-1032. |
| Qi XX, Wei C, Liu XD, et al. Effects of plant growth-promoting rhizobacteria added in seedling substrate on rice growth [J]. Soils, 2020, 52(5): 1025-1032. | |
| [15] | 牛文研, 李小杰, 宋守晔, 等. 两株根际促生细菌的筛选鉴定及其对烟草的促生作用 [J]. 中国烟草科学, 2024, 45(6): 46-54. |
| Niu WY, Li XJ, Song SY, et al. Screening and identification of two plant growth-promoting rhizobacteria and the effects of growth promoting on tobacco [J]. Chin Tob Sci, 2024, 45(6): 46-54. | |
| [16] | Zhang X, Peng JX, Hao XD, et al. Serratia marcescens LYGN1 reforms the rhizosphere microbial community and promotes cucumber and pepper growth in plug seedling cultivation [J]. Plants, 2024, 13(5): 592. |
| [17] | 张慧, 卢文才, 王冬, 等. 一株高产吲哚乙酸的Bacillus cereus YT2-1C的鉴定及促生作用 [J]. 生物技术通报, 2025, 41(5): 300-309. |
| Zhang H, Lu WC, Wang D, et al. Identification of Bacillus cereus YT2-1C with high indoleacetic acid yield and its growth-promoting effect [J]. Biotechnol Bull, 2025, 41(5): 300-309. | |
| [18] | 马连杰, 卢文才, 刘倩, 等. 四株高产吲哚乙酸菌株的筛选鉴定及促生应用研究 [J]. 中国土壤与肥料, 2025(5): 222-230. |
| Ma LJ, Lu WC, Liu Q, et al. Screening, identification and growth-promoting application research of four high IAA-producing strains [J]. Soil Fertil Sci China, 2025(5): 222-230. | |
| [19] | Nikolić I, Berić T, Dimkić I, et al. Biological control of Pseudomonas syringae pv. aptata on sugar beet with Bacillus pumilus SS-10.7 and Bacillus amyloliquefaciens (SS-12.6 and SS-38.4) strains [J]. J Appl Microbiol, 2019, 126(1): 165-176. |
| [20] | 李静, 操一凡, 丁佳兴, 等. 含复合菌群生物育苗基质的研制及其育苗效果 [J]. 南京农业大学学报, 2018, 41(4): 676-684. |
| Li J, Cao YF, Ding JX, et al. Development of bio-nursery substrates containing PGPR flora and evaluation of their seedlings growth promoting effect [J]. J Nanjing Agric Univ, 2018, 41(4): 676-684. | |
| [21] | Sibponkrung S, Kondo T, Tanaka K, et al. Co-inoculation of Bacillus velezensis strain S141 and Bradyrhizobium strains promotes nodule growth and nitrogen fixation [J]. Microorganisms, 2020, 8(5): 678. |
| [22] | Han YJ, Tian L, Luo Y, et al. Inoculation with PGPR co-fermented broths promotes growth, yields, and quality of pepper by increasing soil nutrients and changing soil bacterial community [J]. J Appl Microbiol, 2024, 135(11): lxae289. |
| [23] | 黄文茂, 易伦, 彭思云, 等. PGPR复合菌剂对辣椒生长及根际 土壤微生物结构的影响 [J]. 中国土壤与肥料, 2020(1): 195-201. |
| Huang WM, Yi L, Peng SY, et al. Effect of PGPR compound bacterial agents on growth of chilli and changes of soil microbial structure [J]. Soil Fertil Sci China, 2020(1): 195-201. | |
| [24] | 黄文茂, 詹永发, 王欢, 等. PGPR菌剂对辣椒的促生效应及根际土壤细菌的响应研究 [J]. 中国土壤与肥料, 2020(5): 152-160. |
| Huang WM, Zhan YF, Wang H, et al. Growth-promoting effects of PGPR microbial agent on chilli and response of rhizosphere soil bacteria [J]. Soil Fertil Sci China, 2020(5): 152-160. | |
| [25] | Han DD, Wang LY, Luo YP. Isolation, identification, and the growth promoting effects of two antagonistic actinomycete strains from the rhizosphere of Mikania micrantha Kunth [J]. Microbiol Res, 2018, 208: 1-11. |
| [26] | Wang AN, Zhang YF, Wang GL, et al. Soil physicochemical properties and microorganisms jointly regulate the variations of soil carbon and nitrogen cycles along vegetation restoration on the Loess Plateau, China [J]. Plant Soil, 2024, 494(1): 413-436. |
| [27] | 郭宇泽, 丁雪敏, 姚岚, 等. 马西利亚菌B260的分离鉴定及促进育苗的效果 [J]. 生物技术通报, 2019, 35(9): 144-149. |
| Guo YZ, Ding XM, Yao L, et al. Isolation and identification of Massilia sp. B260 and its effect on seedling raising [J]. Biotechnol Bull, 2019, 35(9): 144-149. | |
| [28] | Tang A, Haruna AO, Majid NMA, et al. Effects of selected functional bacteria on maize growth and nutrient use efficiency [J]. Microorganisms, 2020, 8(6): 854. |
| [29] | Karuppiah V, Sun JN, Li TT, et al. Co-cultivation of Trichoderma asperellum GDFS1009 and Bacillus amyloliquefaciens 1841 causes differential gene expression and improvement in the wheat growth and biocontrol activity [J]. Front Microbiol, 2019, 10: 1068. |
| [30] | 韩丽珍, 周静, 王欢. 两株对花生促生的芽孢杆菌的鉴定及溶磷特性研究 [J]. 基因组学与应用生物学, 2019, 38(9): 4066-4076. |
| Han LZ, Zhou J, Wang H. Identification and phosphorus-dissolving characteristics of two Bacillus strains promoted on peanut growth [J]. Genom Appl Biol, 2019, 38(9): 4066-4076. | |
| [31] | Han LZ, Zhang H, Xu Y, et al. Biological characteristics and salt-tolerant plant growth-promoting effects of an ACC deaminase-producing Burkholderia pyrrocinia strain isolated from the tea rhizosphere [J]. Arch Microbiol, 2021, 203(5): 2279-2290. |
| [32] | Bagheri N, Ahmadzadeh M, Mariotte P, et al. Behavior and interactions of the plant growth-promoting bacteria Azospirillum oryzae NBT506 and Bacillus velezensis UTB96 in a co-culture system [J]. World J Microbiol Biotechnol, 2022, 38(6): 101. |
| [33] | Qiu LP, Kong WB, Zhu HS, et al. Halophytes increase rhizosphere microbial diversity, network complexity and function in inland saline ecosystem [J]. Sci Total Environ, 2022, 831: 154944. |
| [34] | Guo P, Li C, Liu J, et al. Predation has a significant impact on the complexity and stability of microbial food webs in subalpine lakes [J]. Microbiol Spectr, 2023, 11(6): e0241123. |
| [35] | Kruczyńska A, Kuźniar A, Podlewski J, et al. Bacteroidota structure in the face of varying agricultural practices as an important indicator of soil quality-a culture independent approach [J]. Agric Ecosyst Environ, 2023, 342: 108252. |
| [36] | Carrión VJ, Perez-Jaramillo J, Cordovez V, et al. Pathogen-induced activation of disease-suppressive functions in the endophytic root microbiome [J]. Science, 2019, 366(6465): 606-612. |
| [37] | Mujakić I, Cabello-Yeves PJ, Villena-Alemany C, et al. Multi-environment ecogenomics analysis of the cosmopolitan Phylum Gemmatimonadota [J]. Microbiol Spectr, 2023, 11(5) |
| [38] | Venkatachalam S, Jabir T, Vipindas PV, et al. Ecological significance of Candidatus ARS69 and Gemmatimonadota in the Arctic glacier foreland ecosystems [J]. Appl Microbiol Biotechnol, 2024, 108(1): 128. |
| [39] | Daims H, Lücker S, Wagner M. A new perspective on microbes formerly known as nitrite-oxidizing bacteria [J]. Trends Microbiol, 2016, 24(9): 699-712. |
| [40] | Ju WL, Jin XL, Liu L, et al. Rhizobacteria inoculation benefits nutrient availability for phytostabilization in copper contaminated soil: Drivers from bacterial community structures in rhizosphere [J]. Appl Soil Ecol, 2020, 150: 103450. |
| [41] | 姚佳妮, 代金霞, 刘爽, 等. 基于宏转录组的宁夏荒漠灌丛根际土壤微生物群落组成和功能多样性 [J]. 草业科学, 2025, 42(6): 1331-1342. |
| Yao JN, Dai JX, Liu S, et al. Macrotranscriptome-based rhizosphere soil microbial community composition and functional diversity in the desert shrublands of Ningxia [J]. Pratacultural Sci, 2025, 42(6): 1331-1342. | |
| [42] | Zhang X, Song X, Wang TT, et al. The responses to long-term nitrogen addition of soil bacterial, fungal, and archaeal communities in a desert ecosystem [J]. Front Microbiol, 2022, 13: 1015588. |
| [43] | Goldenberg-Vilar A, Morán-Luis M, Vieites DR, et al. Biogeographical distribution of river microbial communities in Atlantic Catchments [J]. Environ Microbiol Rep, 2025, 17(1): e70065. |
| [44] | Zeilinger S, Gupta VK, Dahms TES, et al. Friends or foes Emerging insights from fungal interactions with plants [J]. FEMS Microbiol Rev, 2016, 40(2): 182-207. |
| [45] | Peterson D, Li T, Calvo AM, et al. Categorization of orthologous gene clusters in 92 ascomycota genomes reveals functions important for phytopathogenicity [J]. J Fungi, 2021, 7(5): 337. |
| [46] | Wang QF, Zhou DP, Chu CB, et al. Responses of arbuscular mycorrhizal fungi to rice-upland crop rotations in an 8-year paddy ecosystem [J]. Soil Sci Soc Am J, 2023, 87(3): 516-527. |
| [47] | 项小燕, 阮思睿, 张颖, 等. 大别山五针松根际微生物和内生菌群落特征及功能多样性 [J]. 广西植物, 2025, 45(1): 31-43. |
| Xiang XY, Ruan SR, Zhang Y, et al. Community characteristics and functional diversity of the rhizosphere microorganism and endophyte from Pinus dabeshanensis [J]. Guihaia, 2025, 45(1): 31-43. | |
| [48] | Fang K, Xu GC, Chen X, et al. Distribution pattern and assembly process of fungal communities along altitude gradient in sediments of the Yellow River Basin [J]. J Fungi, 2025, 11(3): 214. |
| [49] | Li MY, Wang JL, Yao T, et al. Bacterial diversity and community structure in the rhizosphere of four halophytes [J]. Curr Microbiol, 2021, 78(7): 2720-2732. |
| [50] | Nakei MD, Venkataramana PB, Ndakidemi PA. Evaluating Rhizobium and Bradyrhizobium species as potential biocontrol agents for root rot fungi in soybean seedlings [J]. Technol Agron, 2024, 4(1) : e010. |
| [51] | 韩配配, 秦璐, 李银水, 等. 不同营养元素缺乏对甘蓝型油菜苗期生长和根系形态的影响 [J]. 中国油料作物学报, 2016, 38(1): 88-97. |
| Han PP, Qin L, Li YS, et al. Effects of different nutrient deficiencies on growth and root morphological changes of rapeseed seedlings (Brassica napus L.) [J]. Chin J Oil Crop Sci, 2016, 38(1): 88-97. |
| [1] | 裴红霞, 汪露瑶, 李生梅, 高晶霞. 基于SCoT、SRAP和SSR分子标记的220份辣椒种质资源遗传多样性分析[J]. 生物技术通报, 2025, 41(8): 165-174. |
| [2] | 段敏杰, 李怡斐, 王春萍, 黄任中, 黄启中, 张世才. 辣椒果实颜色性状与SSR分子标记的关联分析及指纹图谱构建[J]. 生物技术通报, 2025, 41(7): 81-94. |
| [3] | 彭绍智, 王登科, 张祥, 戴雄泽, 徐昊, 邹学校. 辣椒CaFD1基因克隆、表达特征及功能验证[J]. 生物技术通报, 2025, 41(5): 153-164. |
| [4] | 王田田, 常雪瑞, 黄婉洋, 黄嘉欣, 苗如意, 梁燕平, 王静. 辣椒GASA基因家族的鉴定及分析[J]. 生物技术通报, 2025, 41(4): 166-175. |
| [5] | 夏馨媛, 薛道晟, 李鑫静, 龙俊杰, 陆开形, 丁沃娜, 李梦莎. 稻油轮作土壤多功能促生菌的鉴定及其对油菜生长和根际细菌群落的影响[J]. 生物技术通报, 2025, 41(4): 289-301. |
| [6] | 马耀武, 张麒宇, 杨淼, 蒋诚, 张振宇, 张伊琳, 李梦莎, 许嘉阳, 张斌, 崔光周, 姜瑛. 烟草根际促生菌的筛选鉴定及促生性能研究[J]. 生物技术通报, 2025, 41(3): 271-281. |
| [7] | 王静, 常雪瑞, 贾旭, 黄嘉欣, 王田田, 梁燕平. 辣椒CaUBC38基因的克隆及功能分析[J]. 生物技术通报, 2025, 41(10): 242-252. |
| [8] | 刘克寒, 杨升辉, 黄巧云, 崔文靖. 黑龙江大豆根瘤菌及根际促共生菌株的筛选及应用[J]. 生物技术通报, 2025, 41(1): 252-262. |
| [9] | 张婷, 万雨欣, 徐伟慧, 王志刚, 陈文晶, 胡云龙. 一株玉米根际促生菌Leclercia adecarboxylata LN01促生效果研究及其基因组分析[J]. 生物技术通报, 2025, 41(1): 263-275. |
| [10] | 刘倩, 马连杰, 张慧, 王冬, 范茂, 廖敦秀, 赵正武, 卢文才. 辣椒炭疽病生防菌株TN2的筛选鉴定与抑菌效果[J]. 生物技术通报, 2025, 41(1): 287-297. |
| [11] | 吴慧琴, 王延宏, 刘涵, 司政, 刘雪晴, 王静, 阳宜, 成妍. 辣椒UGT基因家族的鉴定及表达分析[J]. 生物技术通报, 2024, 40(9): 198-211. |
| [12] | 刘文志, 贺丹, 李鹏, 傅应林, 张译心, 温华杰, 于文清. 多粘类芽胞杆菌新菌株X-11及其对番茄和水稻的促生效应[J]. 生物技术通报, 2024, 40(9): 249-259. |
| [13] | 常雪瑞, 王田田, 王静. 辣椒E2基因家族的鉴定及分析[J]. 生物技术通报, 2024, 40(6): 238-250. |
| [14] | 李慧, 文钰芳, 王悦, 纪超, 石国优, 罗英, 周勇, 李志敏, 吴晓玉, 杨有新, 刘建萍. 盐胁迫下辣椒CaPIF4的表达特性与功能分析[J]. 生物技术通报, 2024, 40(4): 148-158. |
| [15] | 毛立杰, 梁晓, 刘迎, 伍春玲, 韩晓燕, 陈青. CMV通过影响效应因子MpC002的表达干预桃蚜种群增长的机制[J]. 生物技术通报, 2024, 40(4): 271-277. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||