








生物技术通报 ›› 2025, Vol. 41 ›› Issue (1): 324-332.doi: 10.13560/j.cnki.biotech.bull.1985.2024-0540
文静1( ), 李倩倩1, 张明达1, 谭茗月1, 金博阳1, 沈秀丽2, 杜志强1(
), 李倩倩1, 张明达1, 谭茗月1, 金博阳1, 沈秀丽2, 杜志强1( )
)
收稿日期:2024-06-06
出版日期:2025-01-26
发布日期:2025-01-22
通讯作者:
杜志强,男,博士,教授,研究方向:无脊椎动物先天免疫;E-mail: nmdzq1981@163.com作者简介:文静,女,硕士研究生,研究方向:无脊椎动物先天免疫;E-mail: wj1483604284@163.com
基金资助:
WEN Jing1( ), LI Qian-qian1, ZHANG Ming-da1, TAN Ming-yue1, JIN Bo-yang1, SHEN XIU-li2, DU Zhi-qiang1(
), LI Qian-qian1, ZHANG Ming-da1, TAN Ming-yue1, JIN Bo-yang1, SHEN XIU-li2, DU Zhi-qiang1( )
)
Received:2024-06-06
Published:2025-01-26
Online:2025-01-22
摘要:
【目的】为揭示双氧化酶2(DUOX 2)在克氏原螯虾(Procambarus clarkii)肠道中对活性氧(reactive oxygen species,ROS)水平的调节功能,以及对体内抗菌肽基因表达的影响。【方法】首先通过实时荧光定量聚合酶链式反应(real-time quantitative PCR, RT-qPCR)检测金黄色葡萄球菌(Staphylococcus aureus)感染后的血细胞、肝胰腺、肠、鳃组织中Duox 2基因的表达。RNA干扰后金黄色葡萄球菌刺激克氏原螯虾,统计其存活率,以及对血淋巴中细菌清除能力涂板观察,并且利用荧光探针2', 7'-二氯二氢荧光素二乙酸酯(2',7'-dichlorodihydrofluorescein diacetate,DCFH-DA)测定克氏原螯虾肠道中的ROS水平。最后通过RT-qPCR检测肠道中抗菌肽相关基因(Pc-toll 1、Pc-toll 3、Pc-ALF 1、Pc-lysozyme)的表达。【结果】在金黄色葡萄球菌刺激下,克氏原螯虾的Duox 2基因表达在血细胞、肝胰腺、肠、鳃中显著升高。并且RNA干扰技术抑制Duox 2基因的表达后,克氏原螯虾对金黄色葡萄球菌的抵抗力下降,存活率降低,血淋巴中细菌数量增加。此外,Duox 2基因表达降低导致肠道ROS表达的上升水平受到抑制,并且抗菌肽相关基因的表达水平受到抑制,证实了Duox 2在调控克氏原螯虾免疫防御中的重要作用。【结论】Duox 2通过调节克氏原螯虾肠道内的ROS水平,对机体抗菌肽相关基因的表达产生影响,从而调控体内金黄色葡萄球菌的清除能力。Duox 2对克氏原螯虾抵御金黄色葡萄球菌感染的先天免疫反应有重要作用。
文静, 李倩倩, 张明达, 谭茗月, 金博阳, 沈秀丽, 杜志强. Duox 2调控克氏原螯虾肠组织抗细菌先天免疫的分子机制[J]. 生物技术通报, 2025, 41(1): 324-332.
WEN Jing, LI Qian-qian, ZHANG Ming-da, TAN Ming-yue, JIN Bo-yang, SHEN XIU-li, DU Zhi-qiang. Molecular Mechanism of Duox 2 Regulating Innate Immunity against Bacteria in Procambarus clarkii Intestine[J]. Biotechnology Bulletin, 2025, 41(1): 324-332.
| 引物Primer | 引物序列Primer sequence(5'-3') |
|---|---|
| Pc-Duox 2-iF | GCGTAATACGACTCACTATAGGGTGATGAAAAATGCGTTT |
| Pc-Duox 2-iR | GCGTAATACGACTCACTATAGGAAAGATGTCTCCCTGAAG |
| Pc-GFP-iF | GCGTAATACGACTCACTATAGGCGAGCTGGACGGCGACGTAAAC |
| Pc-GFP-iR | GCGTAATACGACTCACTATAGGCTTGAAGTTCACCTTGATGCC |
| Pc-Duox 2-RT-F | GCTGCCCAAGTTTCACTA |
| Pc-Duox 2-RT-R | AAAGGAGGCTGCACCAGC |
| 18 S rRNA-RT-F | TCTTCTTAGAGGGATTAGCGG |
| 18 S rRNA-RT-R | AAGGGGATTGAACGGGTTA |
| Pc-toll 1-RT-F | GACTTGTCCAAAAACGATATACG |
| Pc-toll 1-RT-R | TGCGTTACAGTAGTGAGCGAA |
| Pc-toll 3-RT-F | TCATTTGGCATCTGGCTCAC |
| Pc-toll 3-RT-R | GCAGGTGGTGGCGTTGA |
| Pc-ALF 1-RT-F | CGAGAGGCTGTAGAGGATGC |
| Pc-ALF 1-RT-R | CCCAGTTTGTTGATGATGAG |
| Pc-lysozyme-RT-F | GTCAACCCACCCTCAATAAC |
| Pc-lysozyme-RT-R | CTTGTGAATCAGGGCGTA |
表1 引物列表
Table 1 Primer list
| 引物Primer | 引物序列Primer sequence(5'-3') |
|---|---|
| Pc-Duox 2-iF | GCGTAATACGACTCACTATAGGGTGATGAAAAATGCGTTT |
| Pc-Duox 2-iR | GCGTAATACGACTCACTATAGGAAAGATGTCTCCCTGAAG |
| Pc-GFP-iF | GCGTAATACGACTCACTATAGGCGAGCTGGACGGCGACGTAAAC |
| Pc-GFP-iR | GCGTAATACGACTCACTATAGGCTTGAAGTTCACCTTGATGCC |
| Pc-Duox 2-RT-F | GCTGCCCAAGTTTCACTA |
| Pc-Duox 2-RT-R | AAAGGAGGCTGCACCAGC |
| 18 S rRNA-RT-F | TCTTCTTAGAGGGATTAGCGG |
| 18 S rRNA-RT-R | AAGGGGATTGAACGGGTTA |
| Pc-toll 1-RT-F | GACTTGTCCAAAAACGATATACG |
| Pc-toll 1-RT-R | TGCGTTACAGTAGTGAGCGAA |
| Pc-toll 3-RT-F | TCATTTGGCATCTGGCTCAC |
| Pc-toll 3-RT-R | GCAGGTGGTGGCGTTGA |
| Pc-ALF 1-RT-F | CGAGAGGCTGTAGAGGATGC |
| Pc-ALF 1-RT-R | CCCAGTTTGTTGATGATGAG |
| Pc-lysozyme-RT-F | GTCAACCCACCCTCAATAAC |
| Pc-lysozyme-RT-R | CTTGTGAATCAGGGCGTA |

图1 金黄色葡萄球菌刺激后克氏原螯虾Duox 2基因的表达模式 不同字母代表差异显著(P < 0.05),误差线表示3个重复的标准差(SD),下同
Fig. 1 Expression patterns of Duox 2 gene in P. clarkii stimulated by S. aureus Different letters indicate significant differences(P < 0.05). The error bar indicates three repeated standard deviations(SD), the same below

图3 RNAi并且注射金黄色葡萄球菌之后克氏原螯虾血淋巴细菌清除实验的检测结果
Fig. 3 Results of the bacterial clearance assay in P. clarkii haemolymph after RNAi and injection of S. aureus
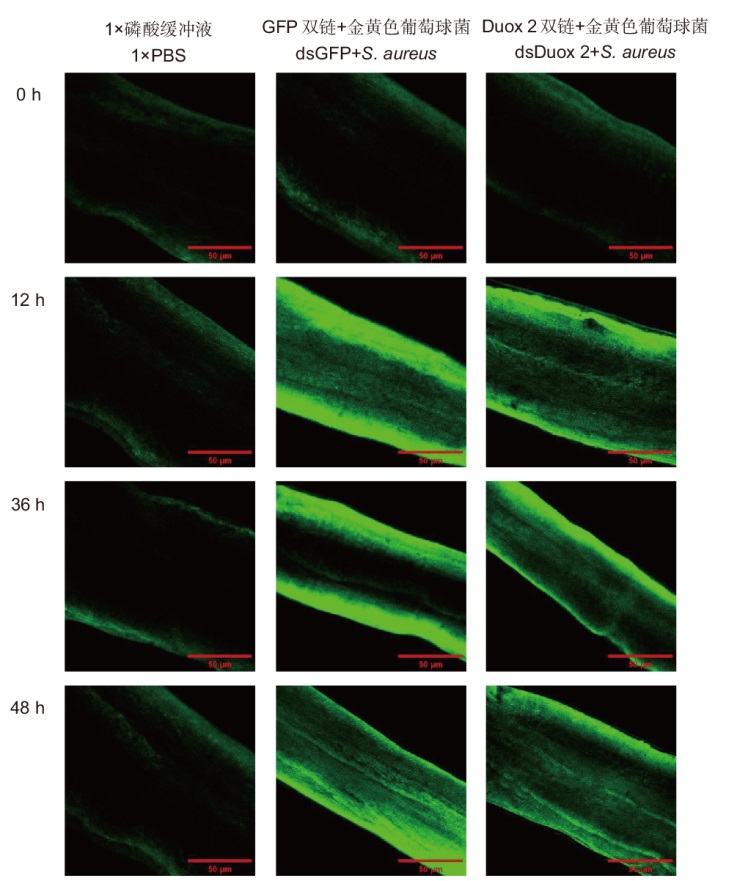
图4 RNAi并且注射金黄色葡萄球菌之后克氏原螯虾肠组织活性氧的检测结果
Fig. 4 Detection results of reactive oxygen species in P. clarkii intestinal tissue after RNAi and S. aureus injection
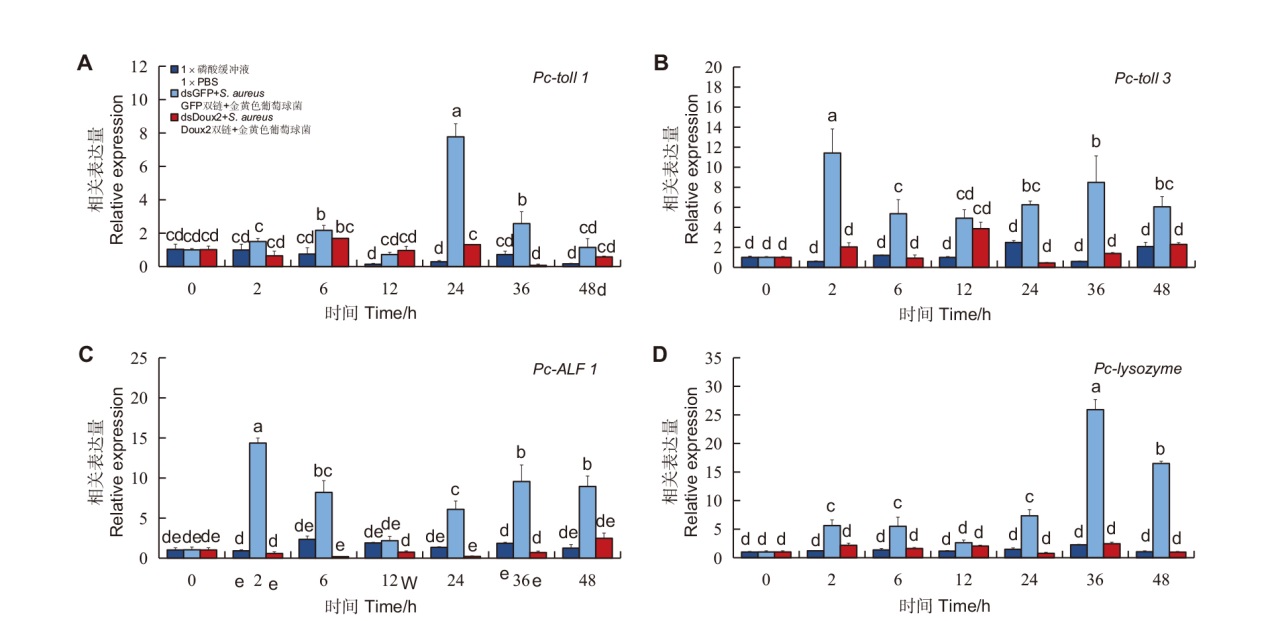
图5 RNAi并且注射金黄色葡萄球菌之后克氏原螯虾肠组织先天免疫相关抗菌肽基因的相对表达量
Fig. 5 Relative expressions of innate immune-related antimicrobial peptide genes in P. clarkii intestinal tissues after RNAi and S. aureus injection
| [1] | 邓灵, 赵康, 夏开, 等. 小龙虾(Procambarus clarkia)加工前后优势腐败菌的分离与鉴定[J]. 食品工业科技, 2020, 41(18): 100-104. |
| Deng L, Zhao K, Xia K, et al. Isolation and identification of specific spoilage organisms in crayfish(Procambarus clarkii)before and after processing[J]. Sci Technol Food Ind, 2020, 41(18): 100-104. | |
| [2] |
Arfatahery N, Davoodabadi A, Abedimohtasab T. Characterization of toxin genes and antimicrobial susceptibility of Staphylococcus aureus isolates in fishery products in Iran[J]. Sci Rep, 2016, 6: 34216.
doi: 10.1038/srep34216 pmid: 27694813 |
| [3] | Shandilya S, Kumar S, Kumar JN, et al. Interplay of gut microbiota and oxidative stress: perspective on neurodegeneration and neuroprotection[J]. J Adv Res, 2021, 38: 223-244. |
| [4] | Poljsak B, Šuput D, Milisav I. Achieving the balance between ROS and antioxidants: when to use the synthetic antioxidants[J]. Oxid Med Cell Longev, 2013, 2013: 956792. |
| [5] | Yang HT, Yang MC, Sun JJ, et al. Catalase eliminates reactive oxygen species and influences the intestinal microbiota of shrimp[J]. Fish Shellfish Immunol, 2015, 47(1): 63-73. |
| [6] | De Deken X, Corvilain B, Dumont JE, et al. Roles of DUOX-mediated hydrogen peroxide in metabolism, host defense, and signaling[J]. Antioxid Redox Signal, 2014, 20(17): 2776-2793. |
| [7] |
Hazime H, Ducasa GM, Santander AM, et al. Intestinal epithelial inactivity of dual oxidase 2 results in microbiome-mediated metabolic syndrome[J]. Cell Mol Gastroenterol Hepatol, 2023, 16(4): 557-572.
doi: 10.1016/j.jcmgh.2023.06.009 pmid: 37369278 |
| [8] |
Sommer F, Bäckhed F. The gut microbiota engages different signaling pathways to induce Duox2 expression in the ileum and colon epithelium[J]. Mucosal Immunol, 2015, 8(2): 372-379.
doi: 10.1038/mi.2014.74 pmid: 25160818 |
| [9] | Lee KA, Kim B, You H, et al. Uracil-induced signaling pathways for DUOX-dependent gut immunity[J]. Fly, 2015, 9(3): 115-120. |
| [10] | Yang QH, Sun ZQ, Zhou YL, et al. SpATF2 participates in maintaining the homeostasis of hemolymph microbiota by regulating dual oxidase expression in mud crab[J]. Fish Shellfish Immunol, 2020, 104: 252-261. |
| [11] | Liu ZG, Zhang HY, Lemaitre B, et al. Duox activation in Drosophila Malpighian tubules stimulates intestinal epithelial renewal through a countercurrent flow[J]. Cell Rep, 2024, 43(4): 114109. |
| [12] |
Dias RO, Cardoso C, Pimentel AC, et al. The roles of mucus-forming mucins, peritrophins and peritrophins with mucin domains in the insect midgut[J]. Insect Mol Biol, 2018, 27(1): 46-60.
doi: 10.1111/imb.12340 pmid: 28833767 |
| [13] | Zeng T, Jaffar S, Xu YJ, et al. The intestinal immune defense system in insects[J]. Int J Mol Sci, 2022, 23(23): 15132. |
| [14] | Zhou YL, Wang LZ, Gu WB, et al. Identification and functional analysis of immune deficiency(IMD)from Scylla paramamosain: the first evidence of IMD signaling pathway involved in immune defense against bacterial infection in crab species[J]. Fish Shellfish Immunol, 2018, 81: 150-160. |
| [15] | Li HY, Li QY, Wang S, et al. Stimulator of interferon genes defends against bacterial infection via IKKβ-mediated Relish activation in shrimp[J]. Front Immunol, 2022, 13: 977327. |
| [16] | Ha EM, Lee KA, Seo YY, et al. Coordination of multiple dual oxidase-regulatory pathways in responses to commensal and infectious microbes in drosophila gut[J]. Nat Immunol, 2009, 10(9): 949-957. |
| [17] | Donkó Á, Péterfi Z, Sum A, et al. Dual oxidases[J]. Phil Trans R Soc B, 2005, 360(1464): 2301-2308. |
| [18] | Sun ZQ, Hao SF, Gong Y, et al. Dual oxidases participate in the regulation of hemolymph microbiota homeostasis in mud crab Scylla paramamosain[J]. Dev Comp Immunol, 2018, 89: 111-121. |
| [19] | Inada M, Kihara K, Kono T, et al. Deciphering of the Dual oxidase(Nox family)gene from kuruma shrimp, Marsupenaeus japonicus: full-length cDNA cloning and characterization[J]. Fish Shellfish Immunol, 2013, 34(2): 471-485. |
| [20] |
Schweikl H, Godula M, Petzel C, et al. Critical role of superoxide anions and hydroxyl radicals in HEMA-induced apoptosis[J]. Dent Mater, 2017, 33(1): 110-118.
doi: S0109-5641(16)30620-0 pmid: 27887776 |
| [21] | He HH, Chi YM, Yuan K, et al. Functional characterization of a reactive oxygen species modulator 1 gene in Litopenaeus vannamei[J]. Fish Shellfish Immunol, 2017, 70: 270-279. |
| [22] | Zhang L, Wang K, Lei YL, et al. Redox signaling: potential arbitrator of autophagy and apoptosis in therapeutic response[J]. Free Radic Biol Med, 2015, 89: 452-465. |
| [23] | de Almeida AJPO, de Oliveira JCPL, da Silva Pontes LV, et al. ROS: basic concepts, sources, cellular signaling, and its implications in aging pathways[J]. Oxid Med Cell Longev, 2022, 2022: 1225578. |
| [24] | Ray PD, Huang BW, Tsuji Y. Reactive oxygen species(ROS)homeostasis and redox regulation in cellular signaling[J]. Cell Signal, 2012, 24(5): 981-990. |
| [25] | Dryden M. Reactive oxygen species: a novel antimicrobial[J]. Int J Antimicrob Agents, 2018, 51(3): 299-303. |
| [26] | Wu XF, Yang MY, Kim JS, et al. Reactivity differences enable ROS for selective ablation of bacteria[J]. Angew Chem Int Ed Engl, 2022, 61(17): e202200808. |
| [27] | Nie JJ, Yu ZX, Yao DF, et al. Litopenaeus vannamei sirtuin 6 homolog(LvSIRT6)is involved in immune response by modulating hemocytes ROS production and apoptosis[J]. Fish Shellfish Immunol, 2020, 98: 271-284. |
| [28] | Benguettat O, Jneid R, Soltys J, et al. The DH31/CGRP enteroendocrine peptide triggers intestinal contractions favoring the elimination of opportunistic bacteria[J]. PLoS Pathog, 2018, 14(9): e1007279. |
| [29] |
Mukherjee S, Hooper LV. Antimicrobial defense of the intestine[J]. Immunity, 2015, 42(1): 28-39.
doi: 10.1016/j.immuni.2014.12.028 pmid: 25607457 |
| [30] | Tassanakajon A, Somboonwiwat K, Supungul P, et al. Discovery of immune molecules and their crucial functions in shrimp immunity[J]. Fish Shellfish Immunol, 2013, 34(4): 954-967. |
| [31] | Yang L, Qiu LM, Fang Q, et al. Cellular and humoral immune interactions between Drosophila and its parasitoids[J]. Insect Sci, 2021, 28(5): 1208-1227. |
| [32] | Yu SC, Luo FZ, Xu YY, et al. Drosophila innate immunity involves multiple signaling pathways and coordinated communication between different tissues[J]. Front Immunol, 2022, 13: 905370. |
| [33] | Ding D, Sun XJ, Yan M, et al. The ECSIT mediated Toll3-dorsal-ALFs pathway inhibits bacterial amplification in kuruma shrimp[J]. Front Immunol, 2022, 13: 807326. |
| [34] | Li HY, Yin B, Wang S, et al. RNAi screening identifies a new Toll from shrimp Litopenaeus vannamei that restricts WSSV infection through activating Dorsal to induce antimicrobial peptides[J]. PLoS Pathog, 2018, 14(9): e1007109. |
| [35] | Wang Z, Chen YH, Dai YJ, et al. A novel vertebrates Toll-like receptor counterpart regulating the anti-microbial peptides expression in the freshwater crayfish, Procambarus clarkii[J]. Fish Shellfish Immunol, 2015, 43(1): 219-229. |
| [36] | Zhang HQ, Cheng WZ, Zheng LB, et al. Identification of a group D anti-lipopolysaccharide factor(ALF)from kuruma prawn(Marsupenaeus japonicus)with antibacterial activity against Vibrio parahaemolyticus[J]. Fish Shellfish Immunol, 2020, 102: 368-380. |
| [37] | Marra A, Hanson MA, Kondo S, et al. Drosophila antimicrobial peptides and lysozymes regulate gut microbiota composition and abundance[J]. mBio, 2021, 12(4): e0082421. |
| [1] | 武志健, 刘广洋, 林志豪, 盛彬, 陈鸽, 许晓敏, 王军伟, 徐东辉. 蔬菜种子萌发的纳米调控及其机制研究进展[J]. 生物技术通报, 2025, 41(1): 14-24. |
| [2] | 王柯然, 闫俊杰, 刘建凤, 高玉林. RNAi技术在马铃薯害虫防控中的应用和风险[J]. 生物技术通报, 2024, 40(9): 4-10. |
| [3] | 周冉, 王兴平, 李彦霞, 罗仍卓么. 金黄色葡萄球菌型乳房炎奶牛乳腺组织的lncRNA差异表达分析[J]. 生物技术通报, 2024, 40(8): 320-328. |
| [4] | 金博阳, 秦仕宇, 张明达, 李倩倩, 文静, 沈秀丽, 杜志强. 小龙虾prx 6基因在对抗金黄色葡萄球菌感染中的分子作用机制研究[J]. 生物技术通报, 2024, 40(7): 314-322. |
| [5] | 蔡志成, 王媛媛, 桑晓涵, 曾丽仙, 邓文韬, 王佳媚. 低温等离子体活化溶液在抑菌及清除生物被膜中的研究进展[J]. 生物技术通报, 2024, 40(6): 95-104. |
| [6] | 李慧, 文钰芳, 王悦, 纪超, 石国优, 罗英, 周勇, 李志敏, 吴晓玉, 杨有新, 刘建萍. 盐胁迫下辣椒CaPIF4的表达特性与功能分析[J]. 生物技术通报, 2024, 40(4): 148-158. |
| [7] | 邹修为, 岳佳妮, 李志宇, 戴良英, 李魏. 水稻热激转录因子HsfA2b调控非生物胁迫抗性的功能分析[J]. 生物技术通报, 2024, 40(2): 90-98. |
| [8] | 邹永烽, 包志明, 曹攀辉, 张嘉媛, 郭介宇, 苏现斌, 徐宇, 许志强, 郭慧. 克氏原螯虾响应高温胁迫的生理代谢机制[J]. 生物技术通报, 2024, 40(11): 321-334. |
| [9] | 陈晓萌, 张雪静, 张欢, 张宝江, 苏艳. 重组牛乳源金黄色葡萄球菌GapC蛋白优势B细胞抗原表位的预测和筛选[J]. 生物技术通报, 2023, 39(5): 306-313. |
| [10] | 张龙喜, 吕琳, 张欢欢, 周金成, 车午男, 董辉. RNAi技术在寄生蜂中的应用研究进展[J]. 生物技术通报, 2023, 39(12): 99-108. |
| [11] | 周恒, 谢彦杰. 植物氧化胁迫信号应答的研究进展[J]. 生物技术通报, 2023, 39(11): 36-43. |
| [12] | 赵佳, 赵飞燕, 沈馨, 高广琦, 孙志宏. 乳酸菌抗氧化活性及其应用研究进展[J]. 生物技术通报, 2023, 39(11): 182-190. |
| [13] | 程深伟, 张克强, 梁军锋, 刘福元, 郜兴亮, 杜连柱. 畜禽养殖粪污中典型致病菌的三重微滴式数字PCR定量检测方法的建立[J]. 生物技术通报, 2022, 38(9): 271-280. |
| [14] | 徐红云, 张明意. GRAS转录因子AtSCL4负调控拟南芥应答渗透胁迫[J]. 生物技术通报, 2022, 38(6): 129-135. |
| [15] | 刘晓玫, 王东鑫, 张春, 魏双施. AAV介导的RNAi对SARS-CoV-2 S基因表达的抑制作用[J]. 生物技术通报, 2022, 38(3): 188-193. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||