








生物技术通报 ›› 2026, Vol. 42 ›› Issue (4): 216-226.doi: 10.13560/j.cnki.biotech.bull.1985.2025-0820
江昕桦1,2,3( ), 方天宇1,2, 张晶晶1,2, 李相媛1,2, 张邦跃1,2, 廖晓珊4, 荣朵艳1,2(
), 方天宇1,2, 张晶晶1,2, 李相媛1,2, 张邦跃1,2, 廖晓珊4, 荣朵艳1,2( )
)
收稿日期:2025-07-30
出版日期:2026-04-26
发布日期:2026-04-30
通讯作者:
荣朵艳,女,博士,副教授,研究方向 :地钱基因功能及信号调控网络;E-mail: rongduoyan@163.com作者简介:江昕桦,女,硕士研究生,研究方向 :地钱基因功能;E-mail: 1978526350@qq.com
基金资助:
JIANG Xin-hua1,2,3( ), FANG Tian-yu1,2, ZHANG Jing-jing1,2, LI Xiang-yuan1,2, ZHANG Bang-yue1,2, LIAO Xiao-shan4, RONG Duo-yan1,2(
), FANG Tian-yu1,2, ZHANG Jing-jing1,2, LI Xiang-yuan1,2, ZHANG Bang-yue1,2, LIAO Xiao-shan4, RONG Duo-yan1,2( )
)
Received:2025-07-30
Published:2026-04-26
Online:2026-04-30
摘要:
目的 对地钱MpPP2A-C基因进行鉴定、表达量分析及与MpPP2A-A蛋白的相互作用研究,为阐明MpPP2A-C基因的功能提供理论依据。 方法 构建地钱和其他植物PP2A-C的系统发育树,并利用拟南芥PP2A-C基因序列进行同源比对,鉴定出地钱PP2A-C基因,对其进行生物信息学分析;通过RT-qPCR探究MpPP2A-C基因在地钱组织特异性及响应脱落酸的表达模式;采用酵母双杂交实验探究MpPP2A-C亚基与MpPP2A-A亚基的相互作用关系。 结果 地钱基因组中鉴定出3个MpPP2A-C基因(MpPP2A-C1、MpPP2A-C2、MpPP2A-C3),在苔藓植物中高度保守,且其编码蛋白具有保守的结构特征,但在理化性质和亚细胞定位上存在差异。MpPP2A-C基因在地钱顶端缺口、胞芽杯和叶状体中均有表达,其中MpPP2A-C1和MpPP2A-C3在顶端缺口处表达量最高,MpPP2A-C2在各组织中表达无显著差异。ABA处理显著抑制地钱叶状体生长,MpPP2A-C1表达量随ABA浓度升高而上升,MpPP2A-C2表达量无显著差异,MpPP2A-C3表达量则先升后降。酵母双杂交实验证实MpPP2A-A与MpPP2A-C1、MpPP2A-C3均存在相互作用。 结论 地钱MpPP2A-C具有高度保守性,能响应脱落酸,且与A亚基存在相互作用。这些结果将为未来研究PP2A-C基因功能奠定了基础。
江昕桦, 方天宇, 张晶晶, 李相媛, 张邦跃, 廖晓珊, 荣朵艳. 地钱MpPP2A-C基因的鉴定及功能分析[J]. 生物技术通报, 2026, 42(4): 216-226.
JIANG Xin-hua, FANG Tian-yu, ZHANG Jing-jing, LI Xiang-yuan, ZHANG Bang-yue, LIAO Xiao-shan, RONG Duo-yan. Identification and Functional Analysis of the MpPP2A-C Gene in Marchantia polymorpha[J]. Biotechnology Bulletin, 2026, 42(4): 216-226.
基因名称 Gene name | 基因ID Locus ID | 分子量 Molecular weight (Da) | 等电点 Point isoelectric | 脂肪系数 Aliphatic index | 不稳定系数 Instability index | 亲水性 GRAVY | 亚细胞定位 Subcellular localization |
|---|---|---|---|---|---|---|---|
| MpPP2A-C1 | Mp6g15310.1 | 35 243.00 | 5.08 | 82.48 | 44.06 | -0.332 | 细胞质 Cytoplasm 线粒体 Mitochondrion 细胞核 Nucleus |
| MpPP2A-C2 | Mp2g13820.1 | 34 746.72 | 5.35 | 89.08 | 44.75 | -0.210 | 细胞质 Cytoplasm 线粒体 Mitochondrion 细胞核 Nucleus |
| MpPP2A-C3 | Mp5g22560.1 | 34 719.48 | 5.07 | 82.28 | 35.67 | -0.240 | 细胞质 Cytoplasm 细胞核 Nucleus |
表1 地钱PP2A-C的基本信息
Table 1 Basic information of PP2A-C in M. polymorpha
基因名称 Gene name | 基因ID Locus ID | 分子量 Molecular weight (Da) | 等电点 Point isoelectric | 脂肪系数 Aliphatic index | 不稳定系数 Instability index | 亲水性 GRAVY | 亚细胞定位 Subcellular localization |
|---|---|---|---|---|---|---|---|
| MpPP2A-C1 | Mp6g15310.1 | 35 243.00 | 5.08 | 82.48 | 44.06 | -0.332 | 细胞质 Cytoplasm 线粒体 Mitochondrion 细胞核 Nucleus |
| MpPP2A-C2 | Mp2g13820.1 | 34 746.72 | 5.35 | 89.08 | 44.75 | -0.210 | 细胞质 Cytoplasm 线粒体 Mitochondrion 细胞核 Nucleus |
| MpPP2A-C3 | Mp5g22560.1 | 34 719.48 | 5.07 | 82.28 | 35.67 | -0.240 | 细胞质 Cytoplasm 细胞核 Nucleus |
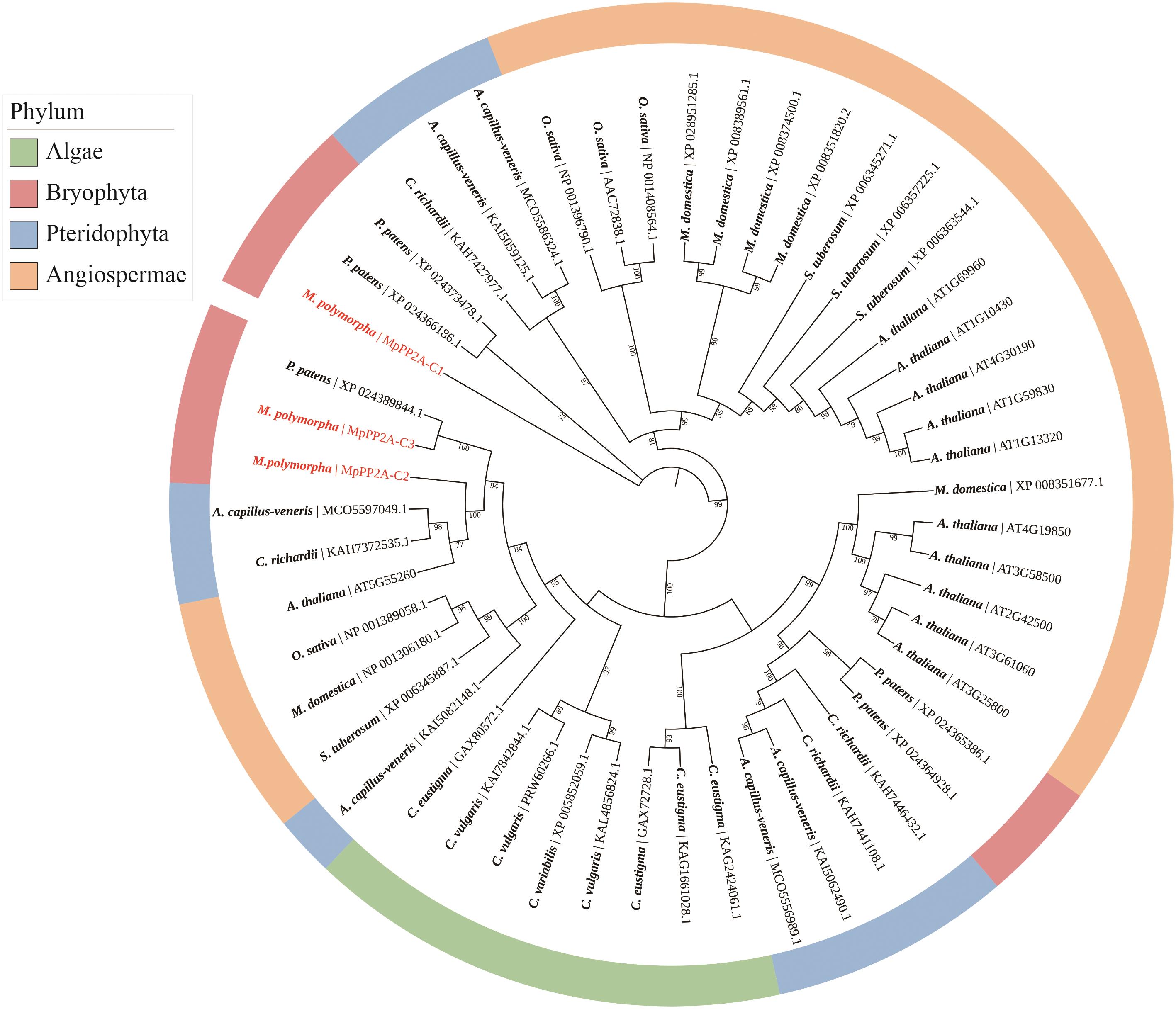
图1 地钱和其他物种PP2A-C氨基酸序列进化分析C. eustigma:衣藻;C. vulgaris:小球藻;M. polymorpha:地钱;P. patens:小立碗藓;A. capillus-veneris:铁线蕨;C. richardii:水蕨;A. thaliana:拟南芥;O. sativa:水稻;M. domestica:苹果;S. tuberosum:马铃薯
Fig. 1 Evolutionary analysis of PP2A-C amino acid sequences in M. polymorpha and other speciesC. Eustigma: Chlamydomonas eustigma; C. vulgaris: Chlorellavulgaris; M. Polymorpha: Marchantia polymorpha; P. patens: Physcomitrella patens; A. capillus-veneris: Adiantum capillus-veneris; C. richardii: Ceratopteris richardii; A. thaliana: Arabidopsis thaliana; O. sativa: Oryza sativa; M. domestica: Malus domestica; S. tuberosum: Solanum tuberosum
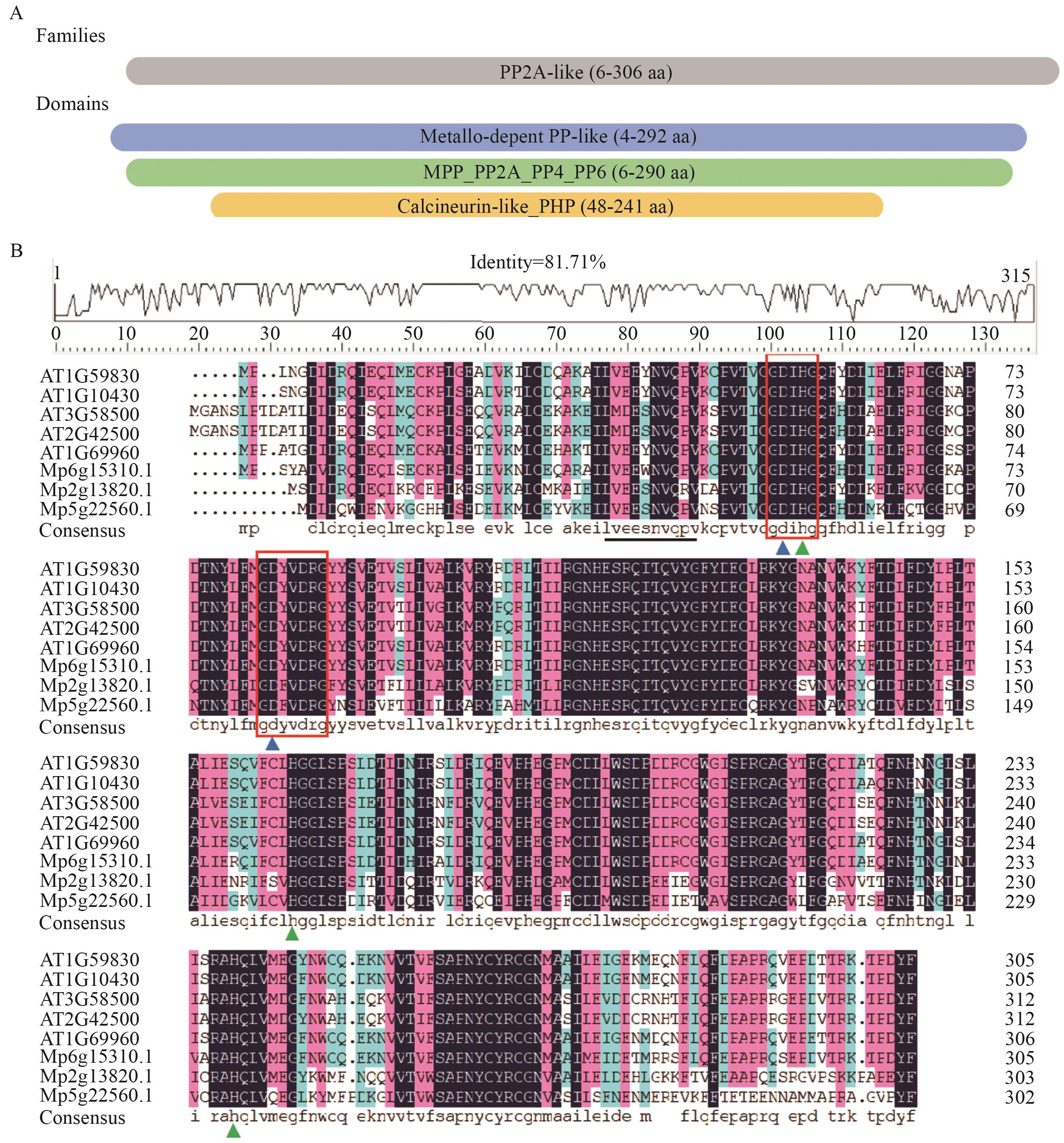
图2 拟南芥与地钱的PP2A-C蛋白保守结构域及序列比对A:MpPP2A-C1蛋白结构域示意图;PP2A-like:丝氨酸/苏氨酸蛋白磷酸酶PP2A样;Metallo-depent PP-like:金属依赖性蛋白磷酸酶样结构域;MPP_PP2A_PP4_PP6:金属依赖性蛋白磷酸酶2A/4/6结构域;Calcineurin-like_PHP:钙调神经磷酸酶样磷酸酯酶结构域。B:蛋白序列比对;红色方框标记催化核心结构域保守基序(GDXHG和GDXDRG);黑色下划线标记A亚基结合结构域保守基序(LVEEYNVQEV);蓝色三角形为金属离子结合位点Asp,绿色三角形为金属离子结合位点Glu
Fig. 2 Conserved domain and sequence alignment of PP2A-C proteins between A. thaliana and M. polymorphaA: Schematic diagram of structural domains in proteins. PP2A-like: Serine/Threonine protein phosphatase PP2A-like. Metallo-depent PP-like: Metallo-dependent phosphatase-like. MPP_PP2A_PP4_PP6: PP2A, PP4, and PP6 phosphoprotein phosphatases, metallophosphatase domain. Calcineurin-like_PHP: Calcineurin-like, phosphoesterase domain. B: Multiple sequence alignment of proteins. The conserved motifs of the catalytic core domain (GDXHG and GDXDRG) are indicated by red boxes; the conserved motif of the A-subunit binding domain (LVEEYNVQEV) is underlined in blue. The blue triangles indicate the metal ion binding site Asp, while the green triangles denote the metal ion binding site Glu
蛋白质名称 Protein name | 氨基酸数量 Number of amino acids (aa) | 蛋白质二级结构 Secondary structure of protein | 蛋白质三级结构 Tertiary structure of protein | |||||
|---|---|---|---|---|---|---|---|---|
α-螺旋 Alpha helix(%) | 延伸链 Extended strand(%) | 不规则卷曲 Random coil(%) | 相似性 Identity (%) | 全局模型质量评估 GMQE | 基于距离约束的定性模型能量分析全局分数 QMEANDisCo Global | |||
| MpPP2A-C1 | 306 | 37.25 | 15.03 | 47.71 | 82.89 | 0.91 | 0.89±0.05 | |
| MpPP2A-C2 | 304 | 39.47 | 14.80 | 45.72 | 65.35 | 0.88 | 0.83±0.05 | |
| MpPP2A-C3 | 303 | 38.28 | 16.18 | 45.54 | 58.14 | 0.88 | 0.83±0.05 | |
表2 地钱PP2A-C蛋白质二、三级结构基本信息
Table 2 Basic information on the secondary and tertiary structures of PP2A-C in M. polymorpha
蛋白质名称 Protein name | 氨基酸数量 Number of amino acids (aa) | 蛋白质二级结构 Secondary structure of protein | 蛋白质三级结构 Tertiary structure of protein | |||||
|---|---|---|---|---|---|---|---|---|
α-螺旋 Alpha helix(%) | 延伸链 Extended strand(%) | 不规则卷曲 Random coil(%) | 相似性 Identity (%) | 全局模型质量评估 GMQE | 基于距离约束的定性模型能量分析全局分数 QMEANDisCo Global | |||
| MpPP2A-C1 | 306 | 37.25 | 15.03 | 47.71 | 82.89 | 0.91 | 0.89±0.05 | |
| MpPP2A-C2 | 304 | 39.47 | 14.80 | 45.72 | 65.35 | 0.88 | 0.83±0.05 | |
| MpPP2A-C3 | 303 | 38.28 | 16.18 | 45.54 | 58.14 | 0.88 | 0.83±0.05 | |
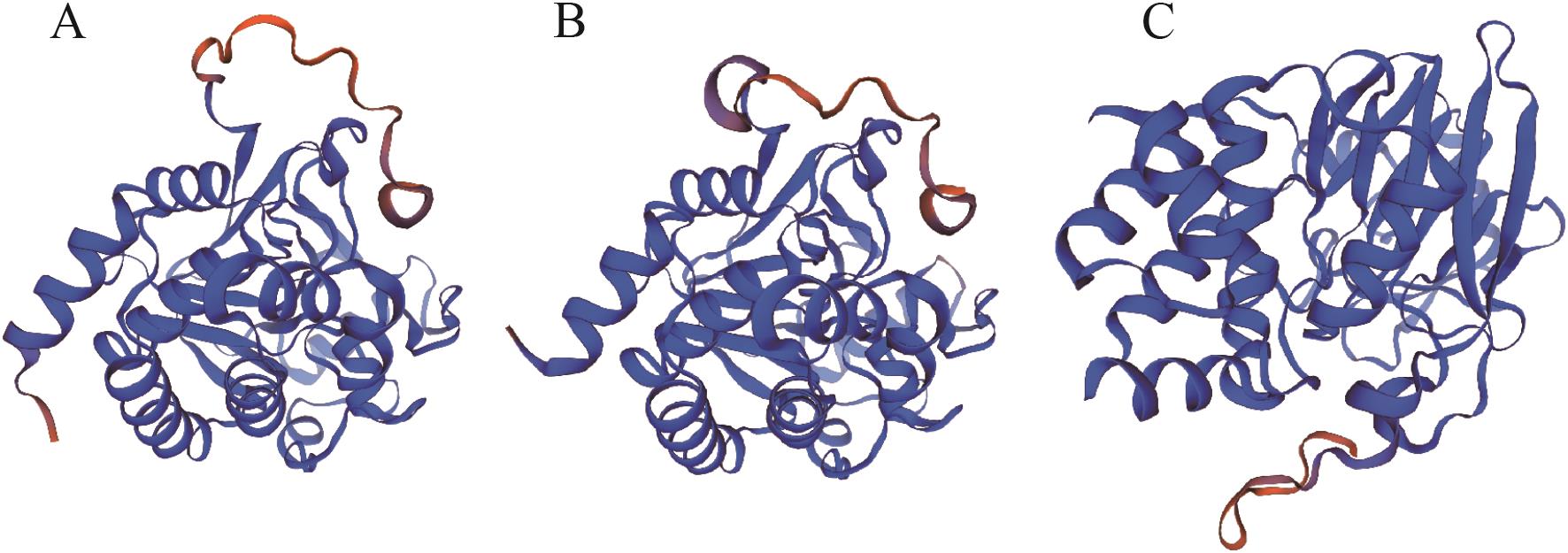
图3 MpPP2A-C蛋白质的三级结构A:MpPP2A-C1;B:MpPP2A-C2;C:MpPP2A-C3。均以可信度梯度进行配色,蓝色表示高置信度(可信),红色表示低置信度(不可信)
Fig. 3 Tertiary structure of MpPP2A-C proteinA: MpPP2A-C1. B: MpPP2A-C2. C: MpPP2A-C3. All structures are colored by the confidence gradient, with blue indicating high confidence (trustworthy) and red indicating low confidence (unreliable)
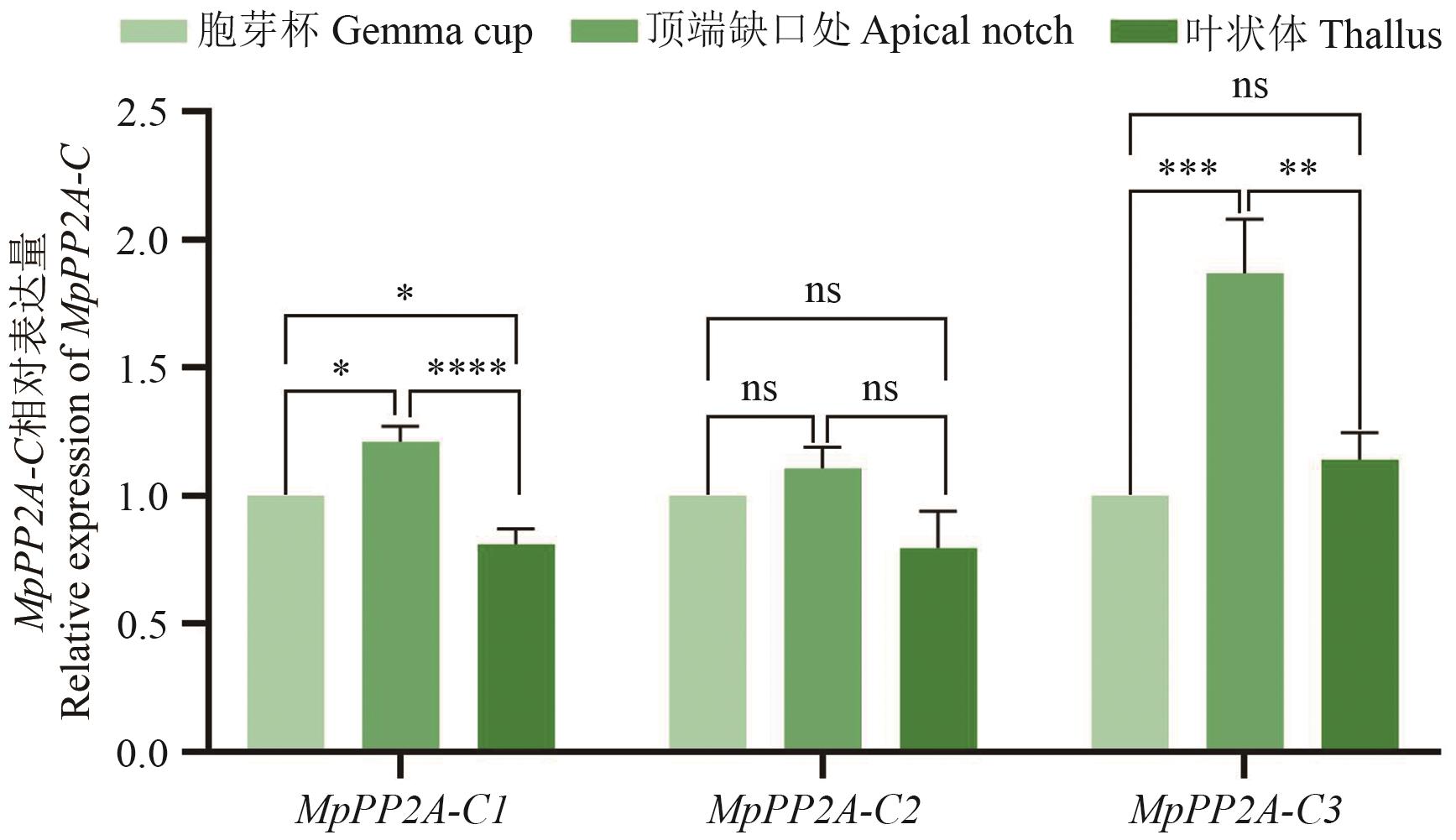
图4 MpPP2A-C基因在地钱不同组织部位中表达量的差异数值为平均值±标准误,****P<0.000 1 (Student’s t-test)
Fig. 4 Differences of expressions of MpPP2A-C in different tissue parts of M. polymorphaValues are the average±standard error, ****P<0.000 1 (Student's t-test)
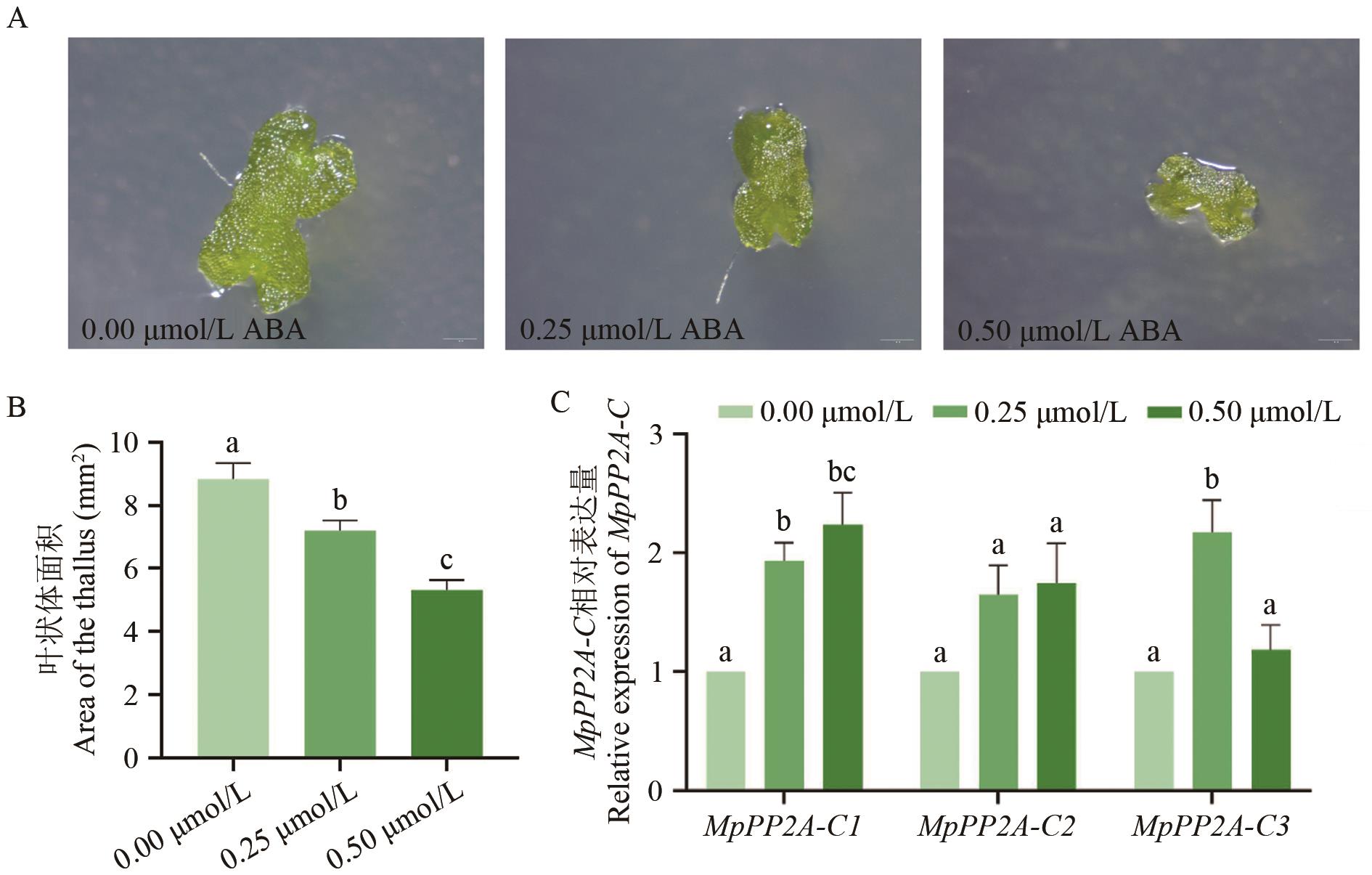
图5 MpPP2A-C基因在不同浓度脱落酸处理下的表型及表达量分析A:不同浓度ABA处理下的地钱叶状体表型对比,标尺长度为1 mm;B:不同浓度ABA处理下的地钱叶状体面积统计;C:MpPP2A-C基因在不同浓度ABA处理下的表达水平。数值为平均值±标准误,小写字母表示组间显著性差异(P<0.05);统计分析采用单因素方差分析结合Duncan多重比较法
Fig. 5 Phenotype and expression analysis of MpPP2A-C gene treated with abscisic acid at different concentrationsA: The comparison of the surface type of the phyllodes of the plant under different concentrations of ABA, and the scale length is 1 mm. B: Statistics of the phyllosoma area under different concentrations of ABA treatment. C: Expressions of MpPP2A-C gene under different ABA concentrations. Values are the average ± standard error, the lowercase letters indicate significant differences between groups (P<0.05). Statistical analysis was performed using one-way analysis of variance (ANOVA) combined with Duncan’s multiple comparison test
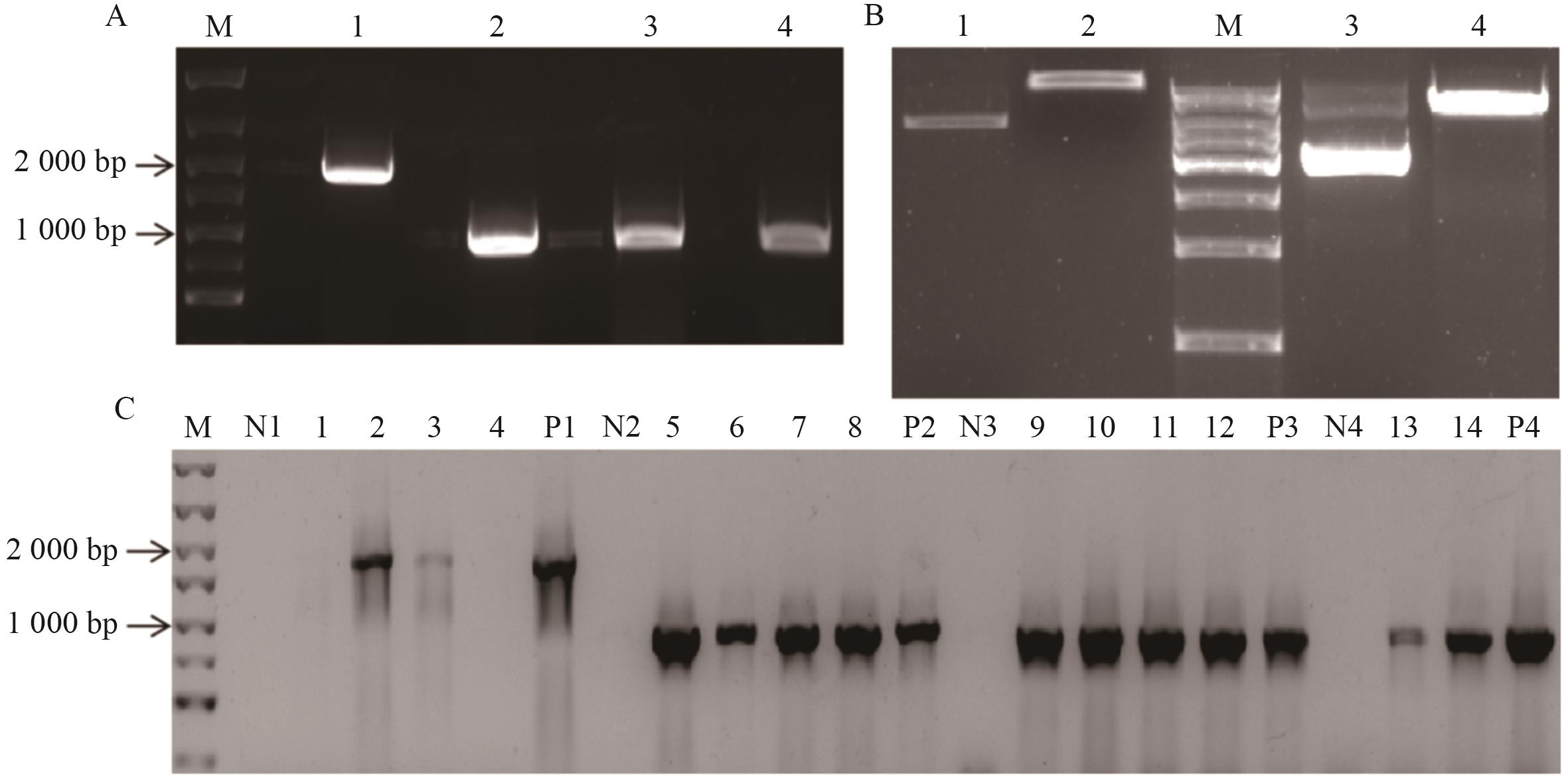
图6 酵母双杂交载体的构建A:MpPP2A-A/C1/C2/C3基因目的片段的扩增(M:DNA Marker;1-4:MpPP2A-A/C1/C2/C3基因的PCR扩增产物);B:pGADT7和pGBKT7质粒的酶切(M:DNA Marker;1:未酶切pGADT7质粒;2:酶切后pGADT7质粒;3:未酶切pGBKT7质粒;4:酶切后pGBKT7质粒);C:pGADT7-MpPP2A-A/C1载体和pGBKT7-MpPP2A-C2/C3载体的构建(M:DNA Marker;1-4:DH5α-pGADT7-MpPP2A-A菌液;N1:阴性对照;P1:阳性对照(MpPP2A-A基因目的片段);5-8:DH5α-pGADT7-MpPP2A-C1菌液;N2:阴性对照;P2:阳性对照(MpPP2A-C1基因目的片段);9-12:DH5α-pGBKT7-MpPP2A-C2菌液;N3:阴性对照;P3:阳性对照(MpPP2A-C2基因目的片段);13-14:DH5α-pGBKT7-MpPP2A-C3菌液;N4:阴性对照;P4:阳性对照(MpPP2A-C2基因目的片段)
Fig. 6 Construction of yeast two-hybrid vectorsA: Amplification of the target fragment of the MpPP2A-A/C1/C2/C3 gene (M: DNA Marker; 1-4: PCR amplified products of the MpPP2A-A/C1/C2/C3 genes). B: Enzymatic digestion of pGADT7 and pGBKT7 plasmids (M: DNA Marker; 1: Undigested pGADT7 plasmid; 2: pGADT7 plasmid after enzymatic digestion; 3: Undigested pGBKT7 plasmid; 4: pGBKT7 plasmid after enzymatic digestion). C: Construction of the pGADT7-MpPP2A-A/C1 vector and pGBKT7-MpPP2A-C2/C3 vectors (M: DNA Marker; 1-4: DH5α-pGADT7-MpPP2A-A bacterial liquid, N1: Negative control, P1: Positive control (the target fragment of the MpPP2A-A); 5-8: DH5α-pGADT7-MpPP2A-C1 bacterial liquid, N2: Negative control, P2: Positive control (the target fragment of the MpPP2A-C1); 9-12: DH5α-pGBKT7-MpPP2A-C2 bacterial liquid. N3: Negative control. P3: Positive control (the target fragment of the MpPP2A-C2); 13-14: DH5α-pGBKT7-MpPP2A-C3 bacterial liquid. N4: Negative control. P4: Positive control (the target fragment of the MpPP2A-C3)
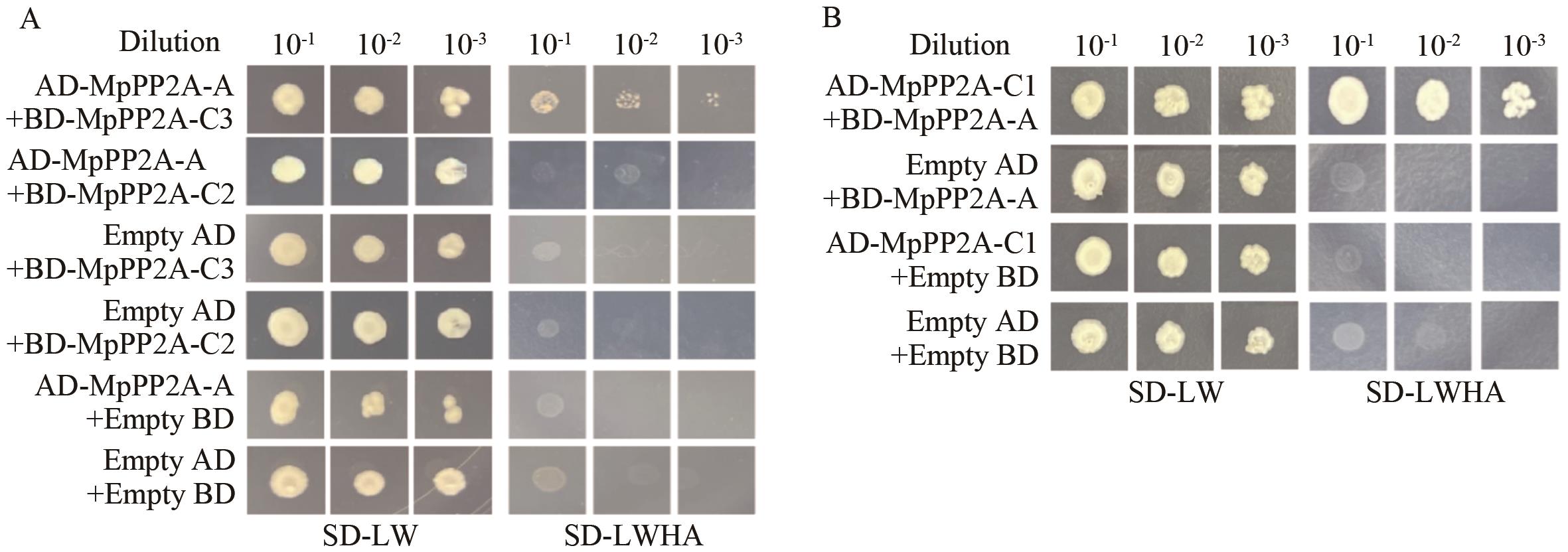
图7 缺陷培养基菌落生长情况A:AD-MpPP2A-A与BD-MpPP2A-C2/C3的互作验证;B:AD-MpPP2A-C1和BD-MpPP2A-A的互作验证;SD-LW:二缺培养基(缺失亮氨酸和色氨酸);SD-LWHA:四缺培养基(缺失亮氨酸、色氨酸、组氨酸和腺嘌呤)
Fig. 7 Growth of colonies in the defective mediumA: Verification of the interaction between AD-MpPP2A-A and BD-MpPP2A-C2/C3. B: Verification of the interaction between AD-MpPP2A-C1 and BD-A. SD-LW: Dideficiency medium (lacking leucine and tryptophan). SD-LWHA: Four-Deficiency medium (lacking leucine, tryptophan, histidine and adenine)
| [1] | Qu L, Liu MY, Zheng LL, et al. Data-independent acquisition-based global phosphoproteomics reveal the diverse roles of casein kinase 1 in plant development [J]. Sci Bull, 2023, 68(18): 2077-2093. |
| [2] | Duan XX, Zhang YY, Huang XH, et al. GreenPhos, a universal method for in-depth measurement of plant phosphoproteomes with high quantitative reproducibility [J]. Mol Plant, 2024, 17(1): 199-213. |
| [3] | Zhang N, Liu HT. Switch on and off: Phospho-events in light signaling pathways [J]. J Integr Plant Biol, 2025, 67(7): 1756-1770. |
| [4] | Zhang WJ, Zhou YW, Zhang Y, et al. Protein phosphorylation: a molecular switch in plant signaling [J]. Cell Rep, 2023, 42(7): 112729. |
| [5] | Tan ST, Luschnig C, Friml J. Pho-view of auxin: reversible protein phosphorylation in auxin biosynthesis, transport and signaling [J]. Mol Plant, 2021, 14(1): 151-165. |
| [6] | Cohen PTW, Brewis ND, Hughes V, et al. Protein serine/threonine phosphatases; an expanding family [J]. FEBS Lett, 1990, 268(2): 355-359. |
| [7] | 刘卫军, 沈瑛, 丁健. 蛋白磷酸酶2A的结构、功能和活性调节 [J]. 生物化学与生物物理学报, 2003(2): 105-112. |
| Liu WJ, Shen Y, Ding J. Protein phosphatase 2A: its structure, function and activity regulation [J]. Acta Biochim Biophys Sin, 2003(2): 105-112. | |
| [8] | 韩琦, 潘超颖. 蛋白磷酸酶2A在真菌细胞中的研究进展 [J]. 微生物学通报, 2021, 48(6): 2214-2221. |
| Han Q, Pan CY. Research progress of fungal protein phosphatase 2A [J]. Microbiol China, 2021, 48(6): 2214-2221. | |
| [9] | Jiang TY, Cui XW, Zeng TM, et al. PTEN deficiency facilitates gemcitabine efficacy in cancer by modulating the phosphorylation of PP2Ac and DCK [J]. Sci Transl Med, 2023, 15(704): eadd7464. |
| [10] | Cai XB, Lee S, Gómez Jaime AP, et al. PHOSPHATASE 2A dephosphorylates PHYTOCHROME-INTERACTING FACTOR3 to modulate photomorphogenesis in Arabidopsis [J]. Plant Cell, 2024, 36(10): 4457-4471. |
| [11] | 丘韶鑫, 杨发恒, 何经纬, 等. 植物中PP2A功能研究进展 [J]. 植物生理学报, 2024, 60(3): 430-440. |
| Qiu SX, Yang FH, He JW, et al. Research advances on function of PP2A in plants [J]. Plant Physiol J, 2024, 60(3): 430-440. | |
| [12] | Durian G, Jeschke V, Rahikainen M, et al. PROTEIN PHOSPHATASE 2A-B'γ controls Botrytis cinerea resistance and developmental leaf senescence [J]. Plant Physiol, 2020, 182(2): 1161-1181. |
| [13] | Shimamura M. Marchantia polymorpha: taxonomy, phylogeny and morphology of a model system [J]. Plant Cell Physiol, 2016, 57(2): 230-256. |
| [14] | Naramoto S, Hata Y, Fujita T, et al. The bryophytes Physcomitrium patens and Marchantia polymorpha as model systems for studying evolutionary cell and developmental biology in plants [J]. Plant Cell, 2022, 34(1): 228-246. |
| [15] | Ishizaki K, Nishihama R, Yamato KT, et al. Molecular genetic tools and techniques for Marchantia polymorpha research [J]. Plant Cell Physiol, 2016, 57(2): 262-270. |
| [16] | Kubota A, Ishizaki K, Hosaka M, et al. Efficient Agrobacterium-mediated transformation of the liverwort Marchantia polymorpha using regenerating thalli [J]. Biosci Biotechnol Biochem, 2013, 77(1): 167-172. |
| [17] | Chang CR, Bowman JL, Meyerowitz EM. Field guide to plant model systems [J]. Cell, 2016, 167(2): 325-339. |
| [18] | Sugano SS, Shirakawa M, Takagi J, et al. CRISPR/Cas9-mediated targeted mutagenesis in the liverwort Marchantia polymorpha L [J]. Plant Cell Physiol, 2014, 55(3): 475-481. |
| [19] | 郝格格, 孙忠富, 张录强, 等. 脱落酸在植物逆境胁迫研究中的进展 [J]. 中国农学通报, 2009, 25(18): 212-215. |
| Hao GG, Sun ZF, Zhang LQ, et al. A research overview of the plant resistance to adverse environment by using abscisic acid [J]. Chin Agric Sci Bull, 2009, 25(18): 212-215. | |
| [20] | Kwak JM, Moon JH, Murata Y, et al. Disruption of a guard cell-expressed protein phosphatase 2A regulatory subunit, RCN1, confers abscisic acid insensitivity in Arabidopsis [J]. Plant Cell, 2002, 14(11): 2849-2861. |
| [21] | Pernas M, García-Casado G, Rojo E, et al. A protein phosphatase 2A catalytic subunit is a negative regulator of abscisic acid signalling [J]. Plant J, 2007, 51(5): 763-778. |
| [22] | Saint-Marcoux D, Proust H, Dolan L, et al. Identification of reference genes for real-time quantitative PCR experiments in the liverwort Marchantia polymorpha [J]. PLoS One, 2015, 10(3): e0118678. |
| [23] | Fu Y, Jia XF, Yuan JW, et al. Fam72a functions as a cell-cycle-controlled gene during proliferation and antagonizes apoptosis through reprogramming PP2A substrates [J]. Dev Cell, 2023, 58(5): 398-415.e7. |
| [24] | Shen C, Li H, Shu L, et al. Ancient large-scale gene duplications and diversification in bryophytes illuminate the plant terrestrialization [J]. New Phytol, 2025, 245(5): 2292-2308. |
| [25] | Rensing SA, Lang D, Zimmer AD, et al. The Physcomitrella Genome reveals evolutionary insights into the conquest of land by plants [J]. Science, 2008, 319(5859): 64-69. |
| [26] | Shao ZY, Zhao B, Kotla P, et al. Phosphorylation status of Bβ subunit acts as a switch to regulate the function of phosphatase PP2A in ethylene-mediated root growth inhibition [J]. New Phytol, 2022, 236(5): 1762-1778. |
| [27] | Yue K, Sandal P, Williams EL, et al. PP2A-3 interacts with ACR4 and regulates formative cell division in the Arabidopsis root [J]. Proc Natl Acad Sci U S A, 2016, 113(5): 1447-1452. |
| [28] | Zhu YF, Huang PC, Guo PC, et al. CDK8 is associated with RAP2.6 and SnRK2.6 and positively modulates abscisic acid signaling and drought response in Arabidopsis [J]. New Phytol, 2020, 228(5): 1573-1590. |
| [29] | Li FQ, Wang JY, Wang PC, et al. Dephosphorylation of bZIP59 by PP2A ensures appropriate shade avoidance response in Arabidopsis [J]. Dev Cell, 2025, 60(4): 551-566.e6. |
| [30] | Wang ZN, Song AX, Tao BL, et al. The phosphatase PP1 sustains global transcription by promoting RNA polymerase Ⅱ pause release [J]. Mol Cell, 2024, 84(24): 4824-4842.e7. |
| [31] | Umezawa T, Sugiyama N, Mizoguchi M, et al. Type 2C protein phosphatases directly regulate abscisic acid-activated protein kinases in Arabidopsis [J]. Proc Natl Acad Sci U S A, 2009, 106(41): 17588-17593. |
| [32] | Ahmad Z, Tian DY, Li Y, et al. Characterization, evolution, expression and functional divergence of the DMP gene family in plants [J]. Int J Mol Sci, 2024, 25(19): 10435. |
| [33] | Lyons SP, Greiner EC, Cressey LE, et al. Regulation of PP2A, PP4, and PP6 holoenzyme assembly by carboxyl-terminal methylation [J]. Sci Rep, 2021, 11: 23031. |
| [34] | Wu CG, Chen H, Guo F, et al. PP2A-B' holoenzyme substrate recognition, regulation and role in cytokinesis [J]. Cell Discov, 2017, 3: 17027. |
| [35] | Li H, Wang JL, Kuan TA, et al. Pathogen protein modularity enables elaborate mimicry of a host phosphatase [J]. Cell, 2023, 186(15): 3196-3207.e17. |
| [1] | 李天源, 亓新亮, 刘珊, 张建成, 王鹏飞, 张帅, 贾璐婷, 穆霄鹏. 欧李SPL基因家族的鉴定及在果实发育过程中的表达分析[J]. 生物技术通报, 2026, 42(3): 362-373. |
| [2] | 陈常璐, 杨志芳, 曹嵩晓, 李杨青, 叶靖烽, 吕海艳, 陈姗姗, 陈昊. 脱落酸与乙烯协同调控薄皮甜瓜果实软化的机制[J]. 生物技术通报, 2026, 42(3): 302-311. |
| [3] | 刘保财, 胡学博, 张武君, 赵云青, 黄颖桢, 陈菁瑛. 石仙桃内参基因筛选与应用[J]. 生物技术通报, 2026, 42(2): 158-168. |
| [4] | 倪莹, 李雷, 汪进萱, 马波, 孟昕, 冷平生, 吴静, 胡增辉. 紫丁香So4CL的克隆及功能分析[J]. 生物技术通报, 2026, 42(1): 139-149. |
| [5] | 陈静欢, 房国楠, 朱文豪, 叶广继, 苏旺, 贺苗苗, 杨生龙, 周云. 马铃薯种质资源淀粉表征及相关基因表达分析[J]. 生物技术通报, 2026, 42(1): 170-183. |
| [6] | 曾厅, 张兰, 罗睿. 转录因子MpR2R3-MYB17调控地钱胞芽发育的功能研究[J]. 生物技术通报, 2026, 42(1): 208-217. |
| [7] | 陈强, 于璎霏, 张颖, 张冲. 茉莉酸甲酯对薄皮甜瓜‘绿宝石’采后冷害的调控[J]. 生物技术通报, 2025, 41(9): 105-114. |
| [8] | 程雪, 付颖, 柴晓娇, 王红艳, 邓欣. 谷子LHC基因家族鉴定及非生物胁迫表达分析[J]. 生物技术通报, 2025, 41(8): 102-114. |
| [9] | 任睿斌, 司二静, 万广有, 汪军成, 姚立蓉, 张宏, 马小乐, 李葆春, 王化俊, 孟亚雄. 大麦条纹病菌GH17基因家族的鉴定及表达分析[J]. 生物技术通报, 2025, 41(8): 146-154. |
| [10] | 曾丹, 黄园, 王健, 张艳, 刘庆霞, 谷荣辉, 孙庆文, 陈宏宇. 铁皮石斛bZIP转录因子家族全基因组鉴定及表达分析[J]. 生物技术通报, 2025, 41(8): 197-210. |
| [11] | 李新妮, 李俊怡, 马雪华, 何卫, 李佳丽, 于佳, 曹晓宁, 乔治军, 刘思辰. 谷子果胶甲酯酶抑制子PMEI基因家族鉴定及其对非生物胁迫的响应分析[J]. 生物技术通报, 2025, 41(7): 150-163. |
| [12] | 韩燚, 侯昌林, 唐露, 孙璐, 谢晓东, 梁晨, 陈小强. 大麦HvERECTA基因的克隆及功能分析[J]. 生物技术通报, 2025, 41(7): 106-116. |
| [13] | 郭秀娟, 冯宇, 吴瑞香, 王利琴, 杨建春. Ca2+处理对胡麻种子萌发影响的转录组分析[J]. 生物技术通报, 2025, 41(7): 139-149. |
| [14] | 曲珊, 赵月, 李雅华, 郑桂玲, 咸洪泉. 调控棘孢木霉几丁质酶Tachi2基因的转录因子和蛋白的互作研究[J]. 生物技术通报, 2025, 41(5): 310-319. |
| [15] | 刘卓君, 柴文婷, 任毅乐, 王新宇, 朱立勋, 赵珊珊, 杨博慧, 范佳利, 李新凤, 赵威军, 吕晋慧, 张春来. 高粱TGA基因响应孢堆黑粉菌(Sporisorium reilianum)侵染表达和DNA变异分析[J]. 生物技术通报, 2025, 41(5): 90-103. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||