








Biotechnology Bulletin ›› 2024, Vol. 40 ›› Issue (3): 322-332.doi: 10.13560/j.cnki.biotech.bull.1985.2023-0996
Previous Articles Next Articles
YANG Wei-jie1( ), YANG Zhou-lin1, ZHU Hao-dong1, WEI Yu1, LIU Jun1,2(
), YANG Zhou-lin1, ZHU Hao-dong1, WEI Yu1, LIU Jun1,2( ), LIU Xun1(
), LIU Xun1( )
)
Received:2023-10-23
Online:2024-03-26
Published:2024-04-08
Contact:
LIU Jun, LIU Xun
E-mail:18783502843@163.com;xunliu0123@suse.edu.cn;liujunbio@suse.edu.cn
YANG Wei-jie, YANG Zhou-lin, ZHU Hao-dong, WEI Yu, LIU Jun, LIU Xun. Study on the Properties and Functions of LchAD Protein, a Key Module of Lichenysin Synthase[J]. Biotechnology Bulletin, 2024, 40(3): 322-332.
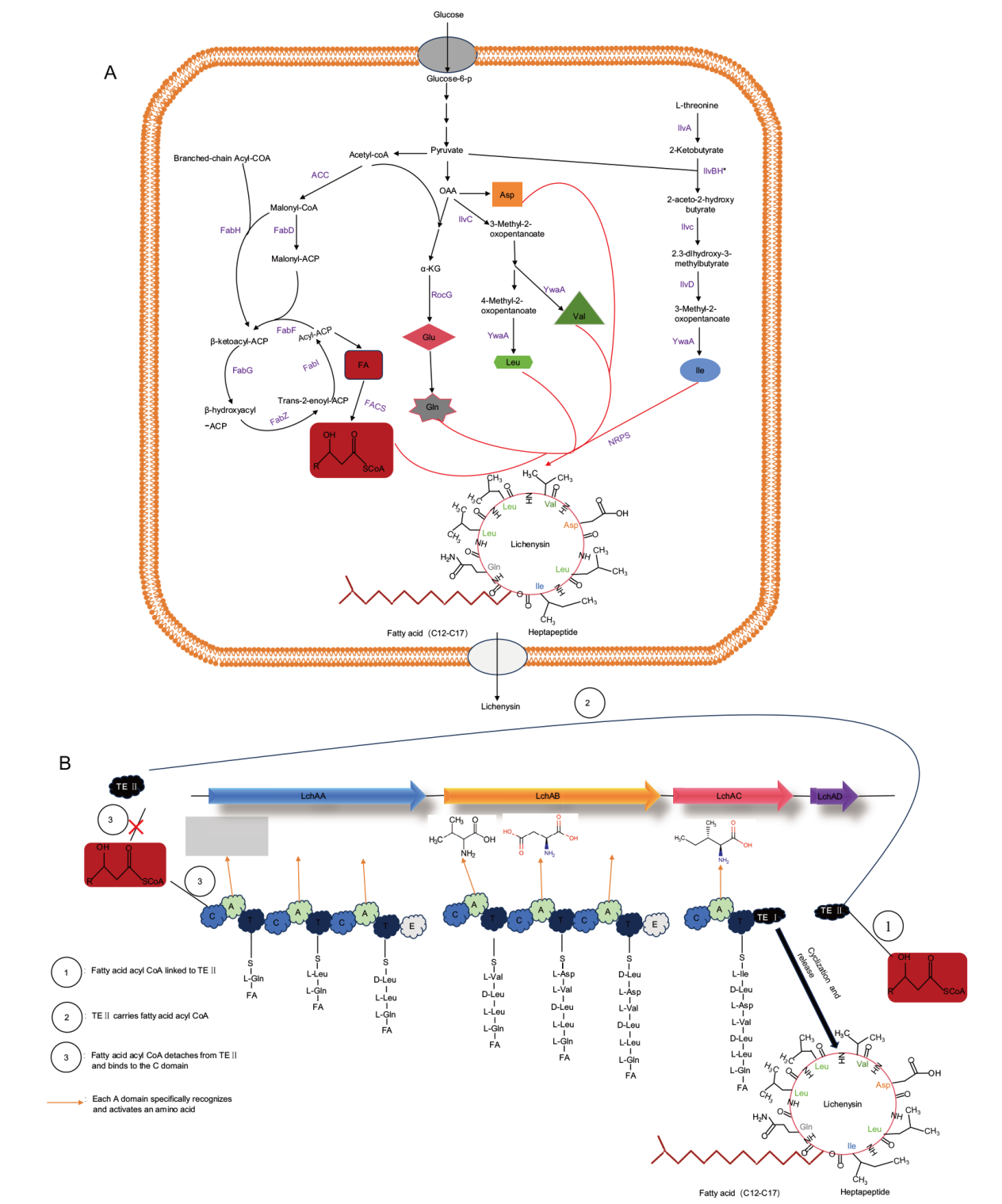
Fig. 1 Schematic diagram of lichenysin biosynthesis A: Production and secretion of lichenysin(ACC: acetyl CoA carboxylase complex; FabD: malonyl-CoA-ACP transacylase; FabH: 3-keto-acyl carrier protein synthase III; FabF: 3-oxoacyl-ACP-synthase II;FabG: β-ketoacyl-ACP reductase; FabI: enoyl-ACP reductases; FabZ: 3-hydroxyacyl-ACP dehydratase; IlvC: acetohydroxy-acid isomeroreductase; RocG: glutamic dehydrogenase; YwaA: branched chain amino acid transaminase; IlvA: L-threonine dehydratase; IlvBH*: regulatory subunits of engineering acetyl hydroxy acid synthase; IlvD: dihydroxyacid dehydratase; FACS: fatty acyl CoA ligases). B: Synthesis of lichenysin. Lichenysin synthase, consists of four modules, with TE I, the TE domain, connected to module 3. Module 4 is TE II, i.e. LchAD
| 菌株 Strain | 菌落形态 Colony morphology |
|---|---|
| YC1 | 奶白色,菌落表面干燥,边缘规则,扁平 |
| YC2 | 半透明、微黄色,菌落表面湿润,边缘不规则,扁平 |
| YC7 | 微红色、菌落表面不光滑但湿润,边缘不规则,扁平 |
Table 1 Colony morphology
| 菌株 Strain | 菌落形态 Colony morphology |
|---|---|
| YC1 | 奶白色,菌落表面干燥,边缘规则,扁平 |
| YC2 | 半透明、微黄色,菌落表面湿润,边缘不规则,扁平 |
| YC7 | 微红色、菌落表面不光滑但湿润,边缘不规则,扁平 |
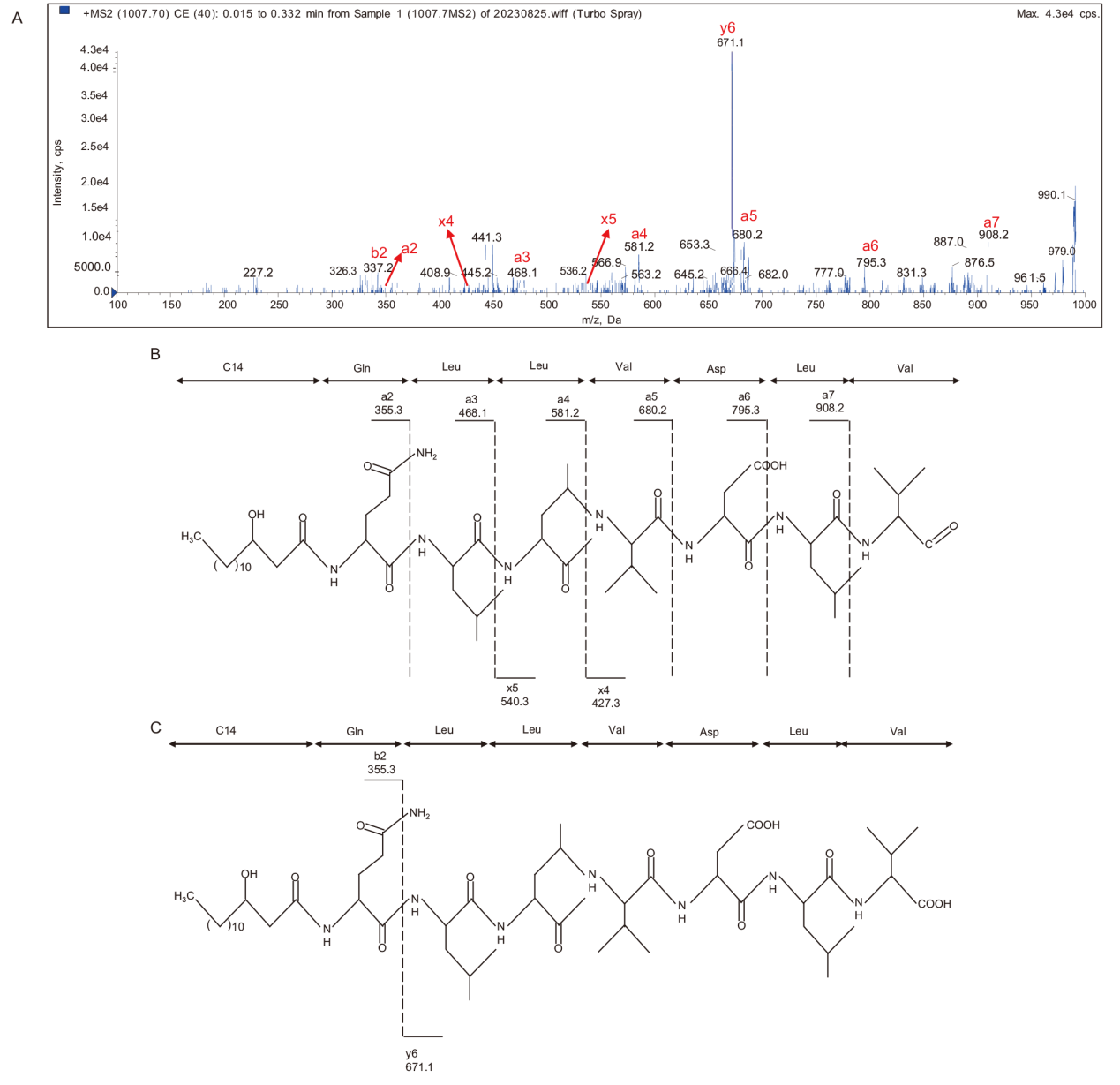
Fig. 3 Secondary mass spectrometry of ion peak with m/z of 1 007.4(A)and theoretical values of characteristic ion fragments generated by simple cleavage(B)and double hydrogen transfer cleavage(C)of this ion peak
| 实验项目Experiment item | 实验结果Experimental result |
|---|---|
| 革兰氏染色 | + |
| 芽孢染色 | + |
| 接触酶实验 | + |
| 厌氧生长实验 | + |
| 葡萄糖产酸实验 | + |
| 葡萄糖产气实验 | - |
| 硝酸盐还原实验 | + |
| 淀粉分解实验 | + |
| V-P实验 | + |
| 明胶分解实验 | + |
Table 2 Physiological and biochemical results
| 实验项目Experiment item | 实验结果Experimental result |
|---|---|
| 革兰氏染色 | + |
| 芽孢染色 | + |
| 接触酶实验 | + |
| 厌氧生长实验 | + |
| 葡萄糖产酸实验 | + |
| 葡萄糖产气实验 | - |
| 硝酸盐还原实验 | + |
| 淀粉分解实验 | + |
| V-P实验 | + |
| 明胶分解实验 | + |
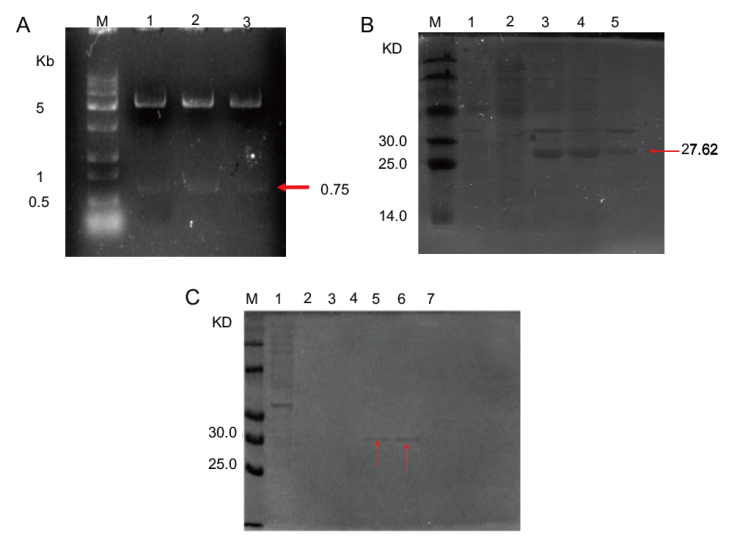
Fig. 5 Results of double digestion of recombinant plasmid pET-28a-LchAD(A)and LchAD protein expression(B)and purification(C) (A)M: DNA maker; 1, 2 and 3 are double restriction endonuclease digestion validation of recombinant plasmids.(B)M: Protein maker. 1: The supernatant of carrier pET-28a. 2: Recombinant plasmid supernatant without IPTG induction at 20℃. 3: Recombinant plasmid induced expression in whole cell fluid at 20℃. 4: The supernatant of the recombinant plasmid induced expression at 20℃.5: Precipitation of recombinant plasmid induced expression at 20℃.(C)M: Protein maker. 1: Flow through fluid. 2-7: Imidazole elution with a concentration of 10, 25, 50, 100, 250 and 500 mmol/L, respectively
| [1] |
Inoda S, Hirohashi Y, Torigoe T, et al. Cytotoxic T lymphocytes efficiently recognize human colon cancer stem-like cells[J]. Am J Pathol, 2011, 178(4): 1805-1813.
doi: 10.1016/j.ajpath.2011.01.004 pmid: 21435460 |
| [2] |
Wang XQ, Hu WW, Zhu LQ, et al. Bacillus subtilis and surfactin inhibit the transmissible gastroenteritis virus from entering the intestinal epithelial cells[J]. Biosci Rep, 2017, 37(2): BSR20170082.
doi: 10.1042/BSR20170082 URL |
| [3] |
Zhang YY, Liu C, Dong B, et al. Anti-inflammatory activity and mechanism of surfactin in lipopolysaccharide-activated macrophages[J]. Inflammation, 2015, 38(2): 756-764.
doi: 10.1007/s10753-014-9986-y pmid: 25331175 |
| [4] |
Chauhan V, Kanwar SS. Lipopeptide(s)associated with human microbiome as potent cancer drug[J]. Semin Cancer Biol, 2021, 70: 128-133.
doi: 10.1016/j.semcancer.2020.06.012 pmid: 32574814 |
| [5] |
Zhang R, Wu Q, Xu Y. Lichenysin, a cyclooctapeptide occurring in Chinese liquor Jiannanchun reduced the headspace concentration of phenolic off-flavors via hydrogen-bond interactions[J]. J Agric Food Chem, 2014, 62(33): 8302-8307.
doi: 10.1021/jf502053g URL |
| [6] | 郅岩, 吴群, 徐岩. 传统白酒中生物活性物质脂肽类化合物及其产生机制[J]. 酿酒科技, 2017(10): 17-23. |
| Zhi Y, Wu Q, Xu Y. Identification and formation mechanism of lipopeptides in Baijiu[J]. Liquor Mak Sci Technol, 2017(10): 17-23. | |
| [7] |
Madslien EH, Rønning HT, Lindbäck T, et al. Lichenysin is produced by most Bacillus licheniformis strains[J]. J Appl Microbiol, 2013, 115(4): 1068-1080.
doi: 10.1111/jam.12299 pmid: 23844764 |
| [8] |
Süssmuth RD, Mainz A. Nonribosomal peptide synthesis-principles and prospects[J]. Angew Chem Int Ed Engl, 2017, 56(14): 3770-3821.
doi: 10.1002/anie.v56.14 URL |
| [9] |
Koglin A, Löhr F, Bernhard F, et al. Structural basis for the selectivity of the external thioesterase of the surfactin synthetase[J]. Nature, 2008, 454(7206): 907-911.
doi: 10.1038/nature07161 |
| [10] |
Du LC, Lou LL. PKS and NRPS release mechanisms[J]. Nat Prod Rep, 2010, 27(2): 255-278.
doi: 10.1039/b912037h pmid: 20111804 |
| [11] |
乔龙亮, 朱鹏, 庞建虎. II型硫脂酶与次生代谢产物合成[J]. 中国生物化学与分子生物学报, 2017, 33(11): 1115-1124.
doi: 10.13865/j.cnki.cjbmb.2017.11.06 |
| Qiao LL, Zhu P, Pang JH. Type II thioesterases and their role in the synthesis of secondary metabolites[J]. Chin J Biochem Mol Biol, 2017, 33(11): 1115-1124. | |
| [12] |
Peschke M, Brieke C, Heimes M, et al. The thioesterase domain in glycopeptide antibiotic biosynthesis is selective for cross-linked aglycones[J]. ACS Chem Biol, 2018, 13(1): 110-120.
doi: 10.1021/acschembio.7b00943 pmid: 29192758 |
| [13] |
Schneider A, Marahiel MA. Genetic evidence for a role of thioesterase domains, integrated in or associated with peptide synthetases, in non-ribosomal peptide biosynthesis in Bacillus subtilis[J]. Arch Microbiol, 1998, 169(5): 404-410.
pmid: 9560421 |
| [14] | Wu H, Liang JD, Gou LX, et al. Recycling of overactivated acyls by a type II thioesterase during calcimycin biosynthesis in Streptomyces chartreusis NRRL 3882[J]. Appl Environ Microbiol, 2018, 84(12): e00587-e00518. |
| [15] | Kotowska M, Ciekot J, Pawlik K. Type II thioesterase ScoT is required for coelimycin production by the modular polyketide synthase Cpk of Streptomyces coelicolor A3(2)[J]. Acta Biochim Pol, 2014, 61(1): 141-147. |
| [16] |
Claxton HB, Akey DL, Silver MK, et al. Structure and functional analysis of RifR, the type II thioesterase from the rifamycin biosynthetic pathway[J]. J Biol Chem, 2009, 284(8): 5021-5029.
doi: 10.1074/jbc.M808604200 pmid: 19103602 |
| [17] |
Whicher JR, Florova G, Sydor PK, et al. Structure and function of the RedJ protein, a thioesterase from the prodiginine biosynthetic pathway in Streptomyces coelicolor[J]. J Biol Chem, 2011, 286(25): 22558-22569.
doi: 10.1074/jbc.M110.213512 URL |
| [18] |
Kotowska M, Pawlik K. Roles of type II thioesterases and their application for secondary metabolite yield improvement[J]. Appl Microbiol Biotechnol, 2014, 98(18): 7735-7746.
doi: 10.1007/s00253-014-5952-8 pmid: 25081554 |
| [19] |
Yakimov MM, Fredrickson HL, Timmis KN. Effect of heterogeneity of hydrophobic moieties on surface activity of lichenysin A, a lipopeptide biosurfactant from Bacillus licheniformis BAS50[J]. Biotechnol Appl Biochem, 1996, 23(1): 13-18.
pmid: 8867891 |
| [20] | 钟桂芳, 张帆, 郭辉祥, 等. 高温大曲中产四甲基吡嗪细菌的筛选及鉴定[J]. 中国酿造, 2020, 39(8): 107-111. |
| Zhong GF, Zhang F, Guo HX, et al. Screening and identification of tetramethylpyrazine-producing strains from high-temperature Daqu[J]. China Brew, 2020, 39(8): 107-111. | |
| [21] | 梁慧珍, 卢延想, 刘正, 等. 高温大曲中高产吡嗪类物质芽孢杆菌的筛选与应用[J]. 中国酿造, 2022, 41(1): 116-122. |
| Liang HZ, Lu YX, Liu Z, et al. Screening and application of high-yield pyrazines Bacillus from high temperature Daqu[J]. China Brew, 2022, 41(1): 116-122. | |
| [22] |
Qiu YM, Xiao F, Wei XT, et al. Improvement of lichenysin production in Bacillus licheniformis by replacement of native promoter of lichenysin biosynthesis operon and medium optimization[J]. Appl Microbiol Biotechnol, 2014, 98(21): 8895-8903.
doi: 10.1007/s00253-014-5978-y URL |
| [23] | 王成俊, 李玲珊, 范梅, 等. 酱香型大曲中产4-乙基愈创木酚芽孢杆菌的筛选、鉴定及特性研究[J]. 酿酒科技, 2023(4): 45-52. |
| Wang CJ, Li LS, Fan M, et al. Screening, identification and characterization of 4-ethylguaiacol producing Bacillus strains from Jiangxiang daqu[J]. Liquor Mak Sci Technol, 2023(4): 45-52. | |
| [24] |
Ayyaz K, Zaheer A, Rasul G, et al. Isolation and identification by 16S rRNA sequence analysis of plant growth-promoting azospirilla from the rhizosphere of wheat[J]. Braz J Microbiol, 2016, 47(3): 542-550.
doi: 10.1016/j.bjm.2015.11.035 pmid: 27133558 |
| [25] |
Ngo G, Centola M, Krasnoselska G, et al. LptC from Anabaena sp. PCC 7120: expression, purification and crystallization[J]. Protein Expr Purif, 2020, 175: 105689.
doi: 10.1016/j.pep.2020.105689 URL |
| [26] |
Jumper J, Evans R, Pritzel A, et al. Highly accurate protein structure prediction with AlphaFold[J]. Nature, 2021, 596(7873): 583-589.
doi: 10.1038/s41586-021-03819-2 |
| [27] |
Wu Q, Zhang R, Peng SQ, et al. Transcriptional characteristics associated with lichenysin biosynthesis in Bacillus licheniformis from Chinese Maotai-flavor liquor making[J]. J Agric Food Chem, 2015, 63(3): 888-893.
doi: 10.1021/jf5036806 URL |
| [28] | 李海岩, 麻新妍, 薛永常. 非核糖体肽合成酶硫酯酶结构域研究进展[J]. 中国抗生素杂志, 2021, 46(12): 1073-1077. |
| Li HY, Ma XY, Xue YC. The research progress of thioesterase domain in non-ribosomal peptide synthetase[J]. Chin J Antibiot, 2021, 46(12): 1073-1077. | |
| [29] | 聂凌鸿, 樊璐, 季方. 大曲中细菌和酵母菌的分离及其Biolog微生物系统分析鉴定[J]. 安徽农业科学, 2012, 40(2): 1036-1038, 1122. |
| Nie LH, Fan L, Ji F. Study on color-protection technique of Tricholoma matsutake in flexible packages[J]. J Anhui Agric Sci, 2012, 40(2): 1036-1038, 1122. | |
| [30] | 周平, 罗惠波, 黄丹, 等. 中高温大曲中一株耐热细菌的分离鉴定及其风味代谢产物分析[J]. 食品工业科技, 2016, 37(24): 215-220. |
| Zhou P, Luo HB, Huang D, et al. Separation and identification of thermoduric bacteria strains in medium/high temperature Daqu and the analysis of the flavor metabolites[J]. Sci Technol Food Ind, 2016, 37(24): 215-220. | |
| [31] | 柏永昊, 张明春, 缪礼鸿. 芽孢杆菌对白酒发酵过程中正丙醇含量的影响[J]. 酿酒科技, 2013(11): 26-29. |
| Bai YH, Zhang MC, Miao LH. Effects of the addition of Bacillus NX3-5Y during liquor fermentation on the content of n-propan-ol[J]. Liquor Mak Sci Technol, 2013(11): 26-29. | |
| [32] | 明红梅, 董瑞丽, 许德富, 等. 浓香型大曲中优势菌的分离及初步鉴定[J]. 酿酒科技, 2013(12): 57-60. |
| Ming HM, Dong RL, Xu DF, et al. Separation and preliminary identification of dominant microbes in Nong-flavor daqu[J]. Liquor Mak Sci Technol, 2013(12): 57-60. | |
| [33] | Rønning HT, Madslien EH, Asp TN, et al. Identification and quantification of lichenysin - a possible source of food poisoning[J]. Food Addit Contam, 2015, 32(12): 2120-2130. |
| [34] |
Suharti, Mahardika G, Raissa, et al. Cloning, heterologous expression, and characterization of a novel thioesterase from natural sample[J]. Heliyon, 2021, 7(3): e06542.
doi: 10.1016/j.heliyon.2021.e06542 URL |
| [35] | 陈维斌. 恰塔努加链霉菌L10中II型硫酯酶生物学功能研究[D]. 杭州: 浙江大学, 2015. |
| Chen WB. The study of the functions of type II Thioesterases in Streptomyces chattanoosensis L10[D]. Hangzhou: Zhejiang University, 2015. | |
| [36] |
Swarbrick CMD, Nanson JD, Patterson EI, et al. Structure, function, and regulation of thioesterases[J]. Prog Lipid Res, 2020, 79: 101036.
doi: 10.1016/j.plipres.2020.101036 URL |
| [37] |
Pourmasoumi F, De S, Peng HY, et al. Proof-reading thioesterase boosts activity of engineered nonribosomal peptide synthetase[J]. ACS Chem Biol, 2022, 17(9): 2382-2388.
doi: 10.1021/acschembio.2c00341 pmid: 36044980 |
| [38] |
Krätzschmar J, Krause M, Marahiel MA. Gramicidin S biosynthesis operon containing the structural genes grsA and grsB has an open reading frame encoding a protein homologous to fatty acid thioesterases[J]. J Bacteriol, 1989, 171(10): 5422-5429.
pmid: 2477357 |
| [39] |
Berditsch M, Trapp M, Afonin S, et al. Antimicrobial peptide gramicidin S is accumulated in granules of producer cells for storage of bacterial phosphagens[J]. Sci Rep, 2017, 7: 44324.
doi: 10.1038/srep44324 pmid: 28295017 |
| [40] |
Coronel-León J, Marqués AM, Bastida J, et al. Optimizing the production of the biosurfactant lichenysin and its application in biofilm control[J]. J Appl Microbiol, 2016, 120(1): 99-111.
doi: 10.1111/jam.12992 pmid: 26519210 |
| [41] | Jacques P. Surfactin and other lipopeptides from Bacillus spp.[M]// Biosurfactants. Berlin, Heidelberg: Springer, 2011: 57-91. |
| [42] |
Curran SC, Pereira JH, Baluyot MJ, et al. Structure and function of BorB, the type II thioesterase from the borrelidin biosynthetic gene cluster[J]. Biochemistry, 2020, 59(16): 1630-1639.
doi: 10.1021/acs.biochem.0c00126 pmid: 32250597 |
| [1] | CHEN Xiao-song, LIU Chao-jie, ZHENG Jia, QIAO Zong-wei, LUO Hui-bo, ZOU Wei. Analyzing the Growth and Caproic Acid Metabolism Mechanism of Rummeliibacillus suwonensis 3B-1 by Tandem Mass Tag-based Quantitative Proteomics [J]. Biotechnology Bulletin, 2024, 40(3): 135-145. |
| [2] | GONG Li-li, YU Hua, YANG Jie, CHEN Tian-chi, ZHAO Shuang-ying, WU Yue-yan. Identification and Analysis of Grape(Vitis vinifera L.)CYP707A Gene Family and Functional Verification to Fruit Ripening [J]. Biotechnology Bulletin, 2024, 40(2): 160-171. |
| [3] | ZHANG Lu-yang, HAN Wen-long, XU Xiao-wen, YAO Jian, LI Fang-fang, TIAN Xiao-yuan, ZHANG Zhi-qiang. Identification and Expression Analysis of the Tobacco TCP Gene Family [J]. Biotechnology Bulletin, 2023, 39(6): 248-258. |
| [4] | LI Jing-rui, WANG Yu-bo, XIE Zi-wei, LI Chang, WU Xiao-lei, GONG Bin-bin, GAO Hong-bo. Identification and Expression Analysis of PIN Gene Family in Melon Under High Temperature Stress [J]. Biotechnology Bulletin, 2023, 39(5): 192-204. |
| [5] | GUO San-bao, SONG Mei-ling, LI Ling-xin, YAO Zi-zhao, GUI Ming-ming, HUANG Sheng-he. Cloning and Analysis of Chalcone Synthase Gene and Its Promoter from Euphorbia maculata [J]. Biotechnology Bulletin, 2023, 39(4): 148-156. |
| [6] | WANG Yi-qing, WANG Tao, WEI Chao-ling, DAI Hao-min, CAO Shi-xian, SUN Wei-jiang, ZENG Wen. Identification and Interaction Analysis of SMAS Gene Family in Tea Plant(Camellia sinensis) [J]. Biotechnology Bulletin, 2023, 39(4): 246-258. |
| [7] | YANG Lan, ZHANG Chen-xi, FAN Xue-wei, WANG Yang-guang, WANG Chun-xiu, LI Wen-ting. Gene Cloning, Expression Pattern, and Promoter Activity Analysis of Chicken BMP15 [J]. Biotechnology Bulletin, 2023, 39(4): 304-312. |
| [8] | CHEN Qiang, ZHOU Ming-kang, SONG Jia-min, ZHANG Chong, WU Long-kun. Identification and Analysis of LBD Gene Family and Expression Analysis of Fruit Development in Cucumis melo [J]. Biotechnology Bulletin, 2023, 39(3): 176-183. |
| [9] | PING Huai-lei, GUO Xue, YU Xiao, SONG Jing, DU Chun, WANG Juan, ZHANG Huai-bi. Cloning and Expression of PdANS in Paeonia delavayi and Correlation with Anthocyanin Content [J]. Biotechnology Bulletin, 2023, 39(3): 206-217. |
| [10] | XING Yuan, SONG Jian, LI Jun-yi, ZHENG Ting-ting, LIU Si-chen, QIAO Zhi-jun. Identification of AP Gene Family and Its Response Analysis to Abiotic Stress in Setaria italica [J]. Biotechnology Bulletin, 2023, 39(11): 238-251. |
| [11] | CHEN Chu-yi, YANG Xiao-mei, CHEN Sheng-yan, CHEN Bin, YUE Li-ran. Expression Analysis of the ZF-HD Gene Family in Chrysanthemum nankingense Under Drought and ABA Treatment [J]. Biotechnology Bulletin, 2023, 39(11): 270-282. |
| [12] | YANG Min, LONG Yu-qing, ZENG Juan, ZENG Mei, ZHOU Xin-ru, WANG Ling, FU Xue-sen, ZHOU Ri-bao, LIU Xiang-dan. Cloning and Function Analysis of Gene UGTPg17 and UGTPg36 in Lonicera macranthoides [J]. Biotechnology Bulletin, 2023, 39(10): 256-267. |
| [13] | GUO Zhi-hao, JIN Ze-xin, LIU Qi, GAO Li. Bioinformatics Analysis, Subcellular Localization and Toxicity Verification of Effector g11335 in Tilletia contraversa Kühn [J]. Biotechnology Bulletin, 2022, 38(8): 110-117. |
| [14] | YU Qiu-lin, MA Jing-yi, ZHAO Pan, SUN Peng-fang, HE Yu-mei, LIU Shi-biao, GUO Hui-hong. Cloning and Functional Analysis of Gynostemma pentaphyllum GpMIR156a and GpMIR166b [J]. Biotechnology Bulletin, 2022, 38(7): 186-193. |
| [15] | CHEN Jia-min, LIU Yong-jie, MA Jin-xiu, LI Dan, GONG Jie, ZHAO Chang-ping, GENG Hong-wei, GAO Shi-qing. Expression Pattern Analysis of Histone Methyltransferase Under Drought Stress in Hybrid Wheat [J]. Biotechnology Bulletin, 2022, 38(7): 51-61. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||