








生物技术通报 ›› 2025, Vol. 41 ›› Issue (1): 347-356.doi: 10.13560/j.cnki.biotech.bull.1985.2024-0675
收稿日期:2024-07-15
出版日期:2025-01-26
发布日期:2025-01-22
通讯作者:
高伟霞,女,博士,副教授,研究方向:微生物;E-mail: gaoweixia@tust.edu.cn作者简介:裴旭娟,女,硕士研究生,研究方向:生物与医药;E-mail: 13652151929@163.com
基金资助:
PEI Xu-juan( ), DI Jing-yi, LIU Hao, GAO Wei-xia(
), DI Jing-yi, LIU Hao, GAO Wei-xia( )
)
Received:2024-07-15
Published:2025-01-26
Online:2025-01-22
摘要:
【目的】挖掘透明质酸分子量的影响元件,构建生产不同分子量大小的透明质酸兽疫链球菌工程菌。【方法】在利用不同碳源培养兽疫链球菌S12生产透明质酸时,发现10 g/L果糖为碳源发酵的透明质酸分子量为1.48×106 Da,比原始发酵培养基(50 g/L蔗糖为碳源)获得的透明质酸分子量2.10×106 Da降低了29.52%。随后将50 g/L的蔗糖和10 g/L的果糖为碳源的S12发酵液进行转录组学分析,发现除果糖代谢相关基因外,精氨酸脱亚胺酶途径的两个关键基因arcA(编码精氨酸脱亚胺酶)和argF(编码鸟氨酸氨基甲酰转移酶)转录水平分别提升了16.29倍和11.27倍。为了探究这两个基因对HA合成的影响,在S12中分别敲除和过表达基因arcA、argF。【结果】arcA过表达菌株在CDM培养基中合成HA分子量2.96×106 Da,比出发菌1.97×106 Da 提高50.25%,另外3个菌株HA分子量变化不大。进一步通过RT-qPCR发现arcA过表达菌株中谷氨酰胺(HA合成中的氨基供体)合成酶编码基因glnA转录水平上调2.0倍,这可能是导致arcA基因过表达HA分子量上升的原因之一。【结论】挖掘到精氨酸代谢相关两个基因对HA分子量具有调控作用,为其他菌株调控HA分子量提供了新靶标。
裴旭娟, 狄靖宜, 刘浩, 高伟霞. 基于转录组分析挖掘兽疫链球菌透明质酸分子量调控元件[J]. 生物技术通报, 2025, 41(1): 347-356.
PEI Xu-juan, DI Jing-yi, LIU Hao, GAO Wei-xia. Exploration of Regulatory Elements for Hyaluronic Acid Molecular Weight in Streptococcus zooepidemicus via Transcriptome Analysis[J]. Biotechnology Bulletin, 2025, 41(1): 347-356.
| Type | Laboratory number | Genotype/Phenotype | Description | Source |
|---|---|---|---|---|
| Plasmid | pLH67 | pSET4s::sacB | Temperature sensitive vector | Our Laboratory |
| pLH421 | pDL278 | Streptococcus shuttle vector, Specr | Our Laboratory | |
| pXJ00 | pLH421::PR22 | pDL278 carrying PR22 | Our Laboratory | |
| pXJ01 | pSET4s::sacB::arcA LR | arcA markerless deletion vector | This work | |
| pXJ02 | pLH421::PR22::arcA | pDL278 carrying PR22::arcA gene | This work | |
| pXJ03 | pSET4s::sacB::argF LR | argF markerless deletion vector | This work | |
| pXJ04 | pLH421::PR22::argF | pDL278 carrying PR22::argF gene | This work | |
| Strains | S12 | ATCC 39920 | Wild-type strain | Our Laboratory |
| S3896 | ΔarcA | arcA markerless deletion mutant | This work | |
| S3897 | arcA/OP | S12 containing plasmid pXJ02 | This work | |
| S3898 | ΔargF | argF markerless deletion mutant | This work | |
| S3899 | argF/OP | S12 containing plasmid pXJ04 | This work |
表1 实验所用质粒及菌株
Table 1 Plasmids and strains used in this study
| Type | Laboratory number | Genotype/Phenotype | Description | Source |
|---|---|---|---|---|
| Plasmid | pLH67 | pSET4s::sacB | Temperature sensitive vector | Our Laboratory |
| pLH421 | pDL278 | Streptococcus shuttle vector, Specr | Our Laboratory | |
| pXJ00 | pLH421::PR22 | pDL278 carrying PR22 | Our Laboratory | |
| pXJ01 | pSET4s::sacB::arcA LR | arcA markerless deletion vector | This work | |
| pXJ02 | pLH421::PR22::arcA | pDL278 carrying PR22::arcA gene | This work | |
| pXJ03 | pSET4s::sacB::argF LR | argF markerless deletion vector | This work | |
| pXJ04 | pLH421::PR22::argF | pDL278 carrying PR22::argF gene | This work | |
| Strains | S12 | ATCC 39920 | Wild-type strain | Our Laboratory |
| S3896 | ΔarcA | arcA markerless deletion mutant | This work | |
| S3897 | arcA/OP | S12 containing plasmid pXJ02 | This work | |
| S3898 | ΔargF | argF markerless deletion mutant | This work | |
| S3899 | argF/OP | S12 containing plasmid pXJ04 | This work |
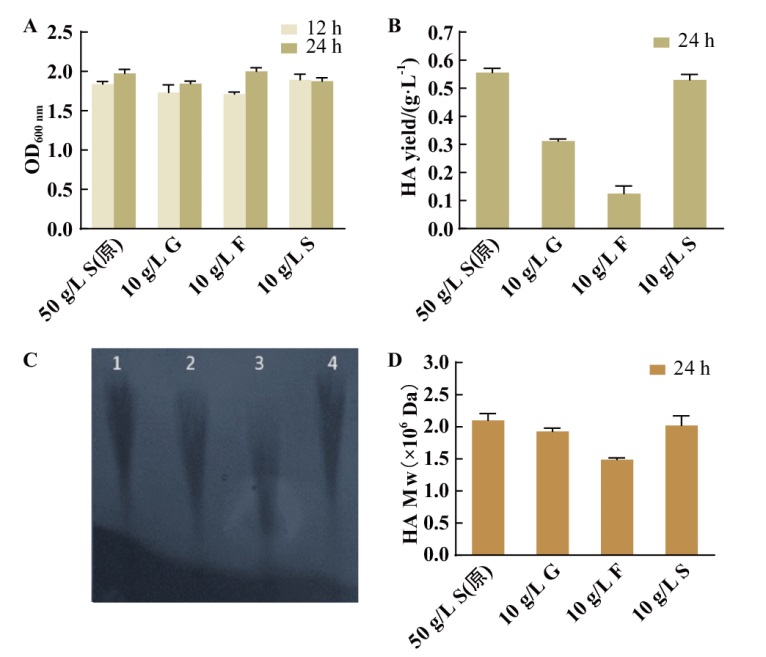
图2 野生型菌株在不同碳源下产HA的表征 A:野生型菌株摇瓶生长情况;B:野生型菌株摇瓶发酵24 h HA的产量;C:24 h HA分子量凝胶图,1:原培养基(50 g/L蔗糖);2:10 g/L葡萄糖;3:10 g/L果糖;4:10 g/L蔗糖; D: HPGPC 检测不同碳源下发酵S12菌株 HA分子量;图中50 g/L S(原):50 g/L 蔗糖(原始培养基);10 g/L G:10 g/L 葡萄糖;10 g/L F: 10 g/L 果糖;10 g/L S: 10 g/L 蔗糖
Fig. 2 Characterization of HA production by wild-type strains under different carbon sources A: Growth of wild-type strain in shaking bottle. B: Yield of hyaluronic acid from wild-type strain in shaking flask for 24 h. C: Molecular weight gel map of 24 h HA, 1: original medium(50 g/L sucrose); 2: 10 g/L glucose; 3: 10 g/L fructose; 4: 10 g/L sucrose). D: HPGPC to detect HA molecular weight of fermented S12 strain under different carbon sources. In the figure, 50 g/L S(original medium): 50 g/L sucrose(original medium); 10 g/L G: 10 g/L glucose; 10 g/L F: 10 g/L fructose; 10 g/L S: 10 g/L sucrose
| Gene | Gene ID | 对数期(FPKM) | Regulate | |
|---|---|---|---|---|
| S12(50 g /L蔗糖)S12(50 g/L sucrose) | S12(10 g /L果糖)S12(10 g/L fructose) | |||
| arcA | SeseC_RS02770 | 102 | 1 700 | Up |
| argF | SeseC _RS02780 | 160 | 1 900 | Up |
| scrA | SeseC _RS01805 | 1 907.5 | 20 | Down |
| fruR2 | SeseC _RS05815 | 1.72 | 175 | Up |
| SeseC _RS05805 | SeseC _RS05805 | 20.25 | 364.3 | Up |
| SeseC _RS01810 | SeseC _RS01810 | 344 | 8.2 | Down |
表2 转录组差异明显的关键基因
Table 2 Key genes with distinct transcriptome differences
| Gene | Gene ID | 对数期(FPKM) | Regulate | |
|---|---|---|---|---|
| S12(50 g /L蔗糖)S12(50 g/L sucrose) | S12(10 g /L果糖)S12(10 g/L fructose) | |||
| arcA | SeseC_RS02770 | 102 | 1 700 | Up |
| argF | SeseC _RS02780 | 160 | 1 900 | Up |
| scrA | SeseC _RS01805 | 1 907.5 | 20 | Down |
| fruR2 | SeseC _RS05815 | 1.72 | 175 | Up |
| SeseC _RS05805 | SeseC _RS05805 | 20.25 | 364.3 | Up |
| SeseC _RS01810 | SeseC _RS01810 | 344 | 8.2 | Down |
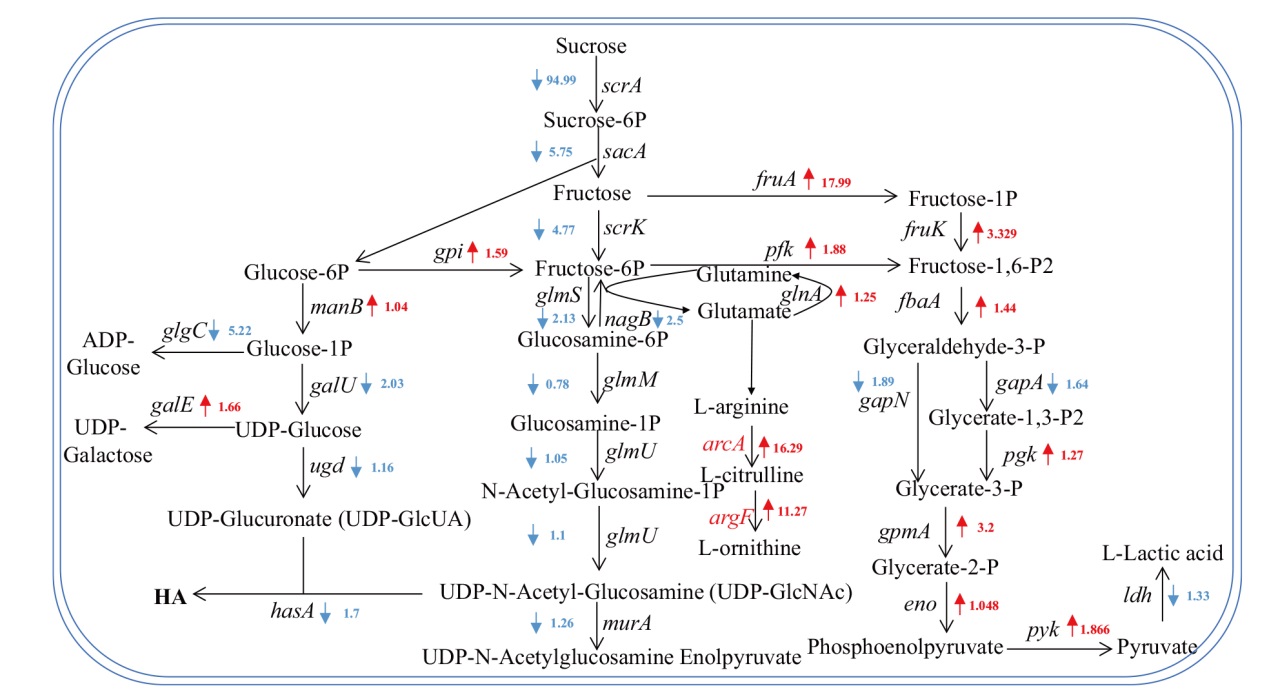
图6 兽疫链球菌中蔗糖代谢途径相关基因的转录水平倍数示意图 红色表示基因转录水平升高,蓝色表示基因转录水平降低
Fig. 6 Schematic diagram of transcriptional levels of genes related to sucrose metabolism pathway in Streptococcus zooepi-demicus Red indicates elevated gene transcription levels, and blue indicates decreased gene transcription levels
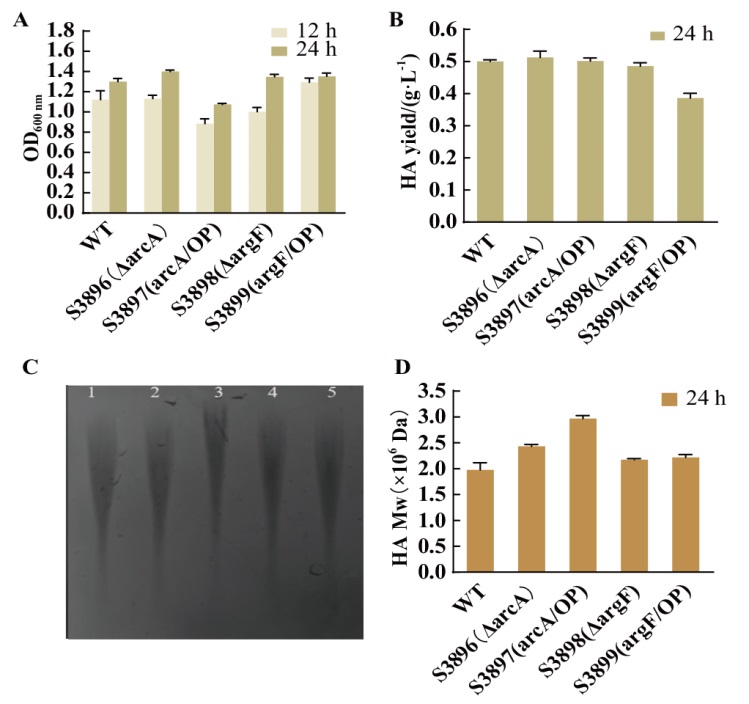
图7 四个构建菌株与野生型菌株在CDM培养基中产HA的表征 A:各菌株摇瓶生长情况;B:各菌株摇瓶发酵24 h 的HA产量; C:24 h 的HA分子量凝胶图(1:兽疫链球菌野生型 S12;2:ΔarcA菌株;3:arcA/OP菌株;4:ΔargF菌株;5:argF/OP菌株);D:HPGPC 检测各菌株产HA分子量
Fig. 7 Characterization of HA in CDM medium of four constructed strains and wild-type strains A: Growth of each strain in shaking flask. B: HA production of each strain in shaking flask for 24 h. C: Molecular weight gel map of HA at 24 h (1: Streptococcus zooepidemicus wild-type S12; 2: ΔarcA strain; 3: arcA/OP strain; 4: ΔargF strain; 5: argF/OP strain). D: HPGPC to detect the molecular weight of HA produced by each strain
| [1] | Zheng XL, Wang BT, Tang X, et al. Absorption, metabolism, and functions of hyaluronic acid and its therapeutic prospects in combination with microorganisms: a review[J]. Carbohydr Polym, 2023, 299: 120153. |
| [2] | Yao ZY, Qin JF, Gong JS, et al. Versatile strategies for bioproduction of hyaluronic acid driven by synthetic biology[J]. Carbohydr Polym, 2021, 264: 118015. |
| [3] | Snetkov P, Zakharova K, Morozkina S, et al. Hyaluronic acid: the influence of molecular weight on structural, physical, physico-chemical, and degradable properties of biopolymer[J]. Polymers, 2020, 12(8): 1800. |
| [4] |
Puré E, Assoian RK. Rheostatic signaling by CD44 and hyaluronan[J]. Cell Signal, 2009, 21(5): 651-655.
doi: 10.1016/j.cellsig.2009.01.024 pmid: 19174187 |
| [5] |
Kang Z, Zhou ZX, Wang Y, et al. Bio-based strategies for producing glycosaminoglycans and their oligosaccharides[J]. Trends Biotechnol, 2018, 36(8): 806-818.
doi: S0167-7799(18)30094-5 pmid: 29674113 |
| [6] | Cheng FY, Gong QY, Yu HM, et al. High-titer biosynthesis of hyaluronic acid by recombinant Corynebacterium glutamicum[J]. Biotechnol J, 2016, 11(4): 574-584. |
| [7] |
Ke CL, Sun LP, Qiao DL, et al. Antioxidant acitivity of low molecular weight hyaluronic acid[J]. Food Chem Toxicol, 2011, 49(10): 2670-2675.
doi: 10.1016/j.fct.2011.07.020 pmid: 21787831 |
| [8] | Kim M, Yang H, Kim H, et al. Novel cosmetic patches for wrinkle improvement: retinyl retinoate- and ascorbic acid-loaded dissolving microneedles[J]. Int J Cosmet Sci, 2014, 36(3): 207-212. |
| [9] |
Waddell DD, Bert JM. The use of hyaluronan after arthroscopic surgery of the knee[J]. Arthroscopy, 2010, 26(1): 105-111.
doi: 10.1016/j.arthro.2009.05.009 pmid: 20117634 |
| [10] | Henrotin Y, Bannuru R, Malaise M, et al. Hyaluronan derivative HYMOVIS® increases cartilage volume and type II collagen turnover in osteoarhritic knee: data from MOKHA study[J]. BMC Musculoskelet Disord, 2019, 20(1): 293. |
| [11] |
Litwiniuk M, Krejner A, Speyrer MS, et al. Hyaluronic acid in inflammation and tissue regeneration[J]. Wounds, 2016, 28(3): 78-88.
pmid: 26978861 |
| [12] |
Frenkel JS. The role of hyaluronan in wound healing[J]. Int Wound J, 2014, 11(2): 159-163.
doi: 10.1111/j.1742-481X.2012.01057.x pmid: 22891615 |
| [13] | Yamasaki K, Drolle E, Nakagawa H, et al. Impact of a low molecular weight hyaluronic acid derivative on contact lens wettability[J]. Cont Lens Anterior Eye, 2021, 44(3): 101334. |
| [14] | Huang WC, Chen SJ, Chen TL. Production of hyaluronic acid by repeated batch fermentation[J]. Biochem Eng J, 2008, 40(3): 460-464. |
| [15] | Hartman AH, Liu HL, Melville SB. Construction and characterization of a lactose-inducible promoter system for controlled gene expression in Clostridium perfringens[J]. Appl Environ Microbiol, 2011, 77(2): 471-478. |
| [16] | Chen SJ, Chen JL, Huang WC, et al. Fermentation process development for hyaluronic acid production by Streptococcus zooepidemicus ATCC 39920[J]. Korean J Chem Eng, 2009, 26(2): 428-432. |
| [17] | Liu L, Yang HQ, Zhang DX, et al. Enhancement of hyaluronic acid production by batch culture of Streptococcus zooepidemicus with n-dodecane as an oxygen vector[J]. J Microbiol Biotechnol, 2009, 19(6): 596-603. |
| [18] | Johns MR, Goh LT, Oeggerli A. Effect of pH, agitation and aeration on hyaluronic acid production by Streptococcus zooepidemicus[J]. Biotechnol Lett, 1994, 16(5): 507-512. |
| [19] | Kim JH, Yoo SJ, Oh DK, et al. Selection of a Streptococcus equi mutant and optimization of culture conditions for the production of high molecular weight hyaluronic acid[J]. Enzyme Microb Technol, 1996, 19(6): 440-445. |
| [20] | Chen WY, Marcellin E, Steen JA, et al. The role of hyaluronic acid precursor concentrations in molecular weight control in Streptococ-cus zooepidemicus[J]. Mol Biotechnol, 2014, 56(2): 147-156. |
| [21] | Chen WY, Marcellin E, Hung J, et al. Hyaluronan molecular weight is controlled by UDP-N-acetylglucosamine concentration in Strep-tococcus zooepidemicus[J]. J Biol Chem, 2009, 284(27): 18007-18014. |
| [22] | Jeeva P, Jayaprakash SR, Jayaraman G. Hyaluronic acid production is enhanced by harnessing the heme-induced respiration in recombinant Lactococcus lactis cultures[J]. Biochem Eng J, 2022, 182: 108428. |
| [23] |
Sheng JZ, Ling PX, Zhu XQ, et al. Use of induction promoters to regulate hyaluronan synthase and UDP-glucose-6-dehydrogenase of Streptococcus zooepidemicus expression in Lactococcus lac-tis: a case study of the regulation mechanism of hyaluronic acid polymer[J]. J Appl Microbiol, 2009, 107(1): 136-144.
doi: 10.1111/j.1365-2672.2009.04185.x pmid: 19302304 |
| [24] |
Jokela TA, Jauhiainen M, Auriola S, et al. Mannose inhibits hyaluronan synthesis by down-regulation of the cellular pool of UDP-N-acetylhexosamines[J]. J Biol Chem, 2008, 283(12): 7666-7673.
doi: 10.1074/jbc.M706001200 pmid: 18201970 |
| [25] | Jagannath S, Ramachandran KB. Influence of competing metabolic processes on the molecular weight of hyaluronic acid synthesized by Streptococcus zooepidemicus[J]. Biochem Eng J, 2010, 48(2): 148-158. |
| [26] |
Weigel PH, DeAngelis PL. Hyaluronan synthases: a decade-plus of novel glycosyltransferases[J]. J Biol Chem, 2007, 282(51): 36777-36781.
doi: 10.1074/jbc.R700036200 pmid: 17981795 |
| [27] | Wang J, He W, Wang T, et al. Sucrose-modified iron nanoparticles for highly efficient microbial production of hyaluronic acid by Streptococcus zooepidemicus[J]. Colloids Surf B Biointerfaces, 2021, 205: 111854-111862. |
| [28] | Marques WL, Raghavendran V, Stambuk BU, et al. Sucrose and Saccharomyces cerevisiae: a relationship most sweet[J]. FEMS Yeast Res, 2016, 16(1): fov107. |
| [29] |
van de Rijn I, Kessler RE. Growth characteristics of group A streptococci in a new chemically defined medium[J]. Infect Immun, 1980, 27(2): 444-448.
doi: 10.1128/iai.27.2.444-448.1980 pmid: 6991416 |
| [30] | Armstrong DC, Cooney MJ, Johns MR. Growth and amino acid requirements of hyaluronic-acid-producing Streptococcus zooepidemi-cus[J]. Appl Microbiol Biotechnol, 1997, 47(3): 309-312. |
| [31] | 孙晓燕. 兽疫链球菌基因敲除系统的建立及hasE功能研究[D]. 天津: 天津科技大学, 2014. |
| Sun XY. Establishment of gene knockout system of Streptococcus zooepidemicus and study on HasE function[D]. Tianjin: Tianjin University of Science & Technology, 2014. | |
| [32] | Oueslati N, Leblanc P, Harscoat-Schiavo C, et al. CTAB turbidimetric method for assaying hyaluronic acid in complex environments and under cross-linked form[J]. Carbohydr Polym, 2014, 112: 102-108. |
| [33] |
Funderburgh JL, Chandler JW. An agarose gel electrophoretic method for analysis of sulfated glycosaminoglycans of cultured cells[J]. Anal Biochem, 1978, 91(2): 464-472.
pmid: 9762132 |
| [34] |
Tlapak-Simmons VL, Baggenstoss BA, Kumari K, et al. Kinetic characterization of the recombinant hyaluronan synthases from Streptococcus pyogenes and Streptococcus equisimilis[J]. J Biol Chem, 1999, 274(7): 4246-4253.
doi: 10.1074/jbc.274.7.4246 pmid: 9933624 |
| [35] |
Marcellin E, Chen WY, Nielsen LK. Understanding plasmid effect on hyaluronic acid molecular weight produced by Streptococcus equi subsp. zooepidemicus[J]. Metab Eng, 2010, 12(1): 62-69.
doi: 10.1016/j.ymben.2009.09.001 pmid: 19782148 |
| [36] | Badle SS, Jayaraman G, Ramachandran KB. Ratio of intracellular precursors concentration and their flux influences hyaluronic acid molecular weight in Streptococcus zooepidemicus and recombinant Lactococcus lactis[J]. Bioresour Technol, 2014, 163: 222-227. |
| [37] | Xu MJ, Rao ZM, Dou WF, et al. Site-directed mutagenesis and feedback-resistant N-acetyl-L-glutamate kinase(NAGK)increase Corynebacterium crenatum L-arginine production[J]. Amino Acids, 2012, 43(1): 255-266. |
| [38] | Ikeda M, Mitsuhashi S, Tanaka K, et al. Reengineering of a Corynebacterium glutamicum L-arginine and L-citrulline producer[J]. Appl Environ Microbiol, 2009, 75(6): 1635-1641. |
| [39] |
Wray LV Jr, Ferson AE, Rohrer K, et al. TnrA, a transcription factor required for global nitrogen regulation in Bacillus subtilis[J]. Proc Natl Acad Sci USA, 1996, 93(17): 8841-8845.
pmid: 8799114 |
| [1] | 寇焙森, 程萌萌, 郭雪琴, 葛彬, 刘迪, 陆海, 李慧. 组蛋白去乙酰化酶抑制剂TSA处理对杨树茎生长发育的影响[J]. 生物技术通报, 2025, 41(1): 240-251. |
| [2] | 岳丽昕, 王清华, 刘泽洲, 孔素萍, 高莉敏. 基于转录组和WGCNA筛选大葱雄性不育相关基因[J]. 生物技术通报, 2024, 40(9): 212-224. |
| [3] | 聂祝欣, 郭瑾, 乔子洋, 李微薇, 张学燕, 刘春阳, 王静. 黑果枸杞不同发育时期果实花色苷合成的转录组分析[J]. 生物技术通报, 2024, 40(8): 106-117. |
| [4] | 周麟, 黄顺满, 苏文坤, 姚响, 屈燕. 滇山茶bHLH基因家族鉴定及花色形成相关基因筛选[J]. 生物技术通报, 2024, 40(8): 142-151. |
| [5] | 王睿, 戚继. 整合组织学图像信息增强空间转录组细胞聚类的分辨率[J]. 生物技术通报, 2024, 40(8): 39-46. |
| [6] | 高萌萌, 赵天宇, 焦馨悦, 林春晶, 关哲允, 丁孝羊, 孙妍妍, 张春宝. 大豆细胞质雄性不育系及其恢复系的比较转录组分析[J]. 生物技术通报, 2024, 40(7): 137-149. |
| [7] | 廖杨梅, 赵国春, 翁学煌, 贾黎明, 陈仲. 无患子雄性不育品种‘琦蕊’不同发育时期雄花转录组分析[J]. 生物技术通报, 2024, 40(7): 197-206. |
| [8] | 白志元, 徐菲, 杨午, 王明贵, 杨玉花, 张海平, 张瑞军. 大豆细胞质雄性不育弱恢复型杂种F1育性转变的转录组分析[J]. 生物技术通报, 2024, 40(6): 134-142. |
| [9] | 秦健, 李振月, 何浪, 李俊玲, 张昊, 杜荣. 肌源性细胞分化的单细胞转录谱变化及细胞间通讯分析[J]. 生物技术通报, 2024, 40(6): 330-342. |
| [10] | 吴迪, 游小凤, 郑亦铮, 林楠, 张燕燕, 魏艺聪. 草珊瑚中类胡萝卜素合成的内源激素调控机制分析[J]. 生物技术通报, 2024, 40(5): 203-214. |
| [11] | 郭纯, 宋桂梅, 闫艳, 邸鹏, 王英平. 西洋参bZIP基因家族全基因组鉴定和表达分析[J]. 生物技术通报, 2024, 40(4): 167-178. |
| [12] | 钟匀, 林春, 刘正杰, 董陈文华, 毛自朝, 李兴玉. 芦笋皂苷合成相关糖基转移酶基因克隆及原核表达分析[J]. 生物技术通报, 2024, 40(4): 255-263. |
| [13] | 杨淇, 魏子迪, 宋娟, 童堃, 杨柳, 王佳涵, 刘海燕, 栾维江, 马轩. 水稻组蛋白H1三突变体的创建和转录组学分析[J]. 生物技术通报, 2024, 40(4): 85-96. |
| [14] | 谢倩, 江来, 贺进, 刘玲玲, 丁明月, 陈清西. 不同鲜食品质橄榄果实转录组测序及酚类代谢途径相关调控基因挖掘[J]. 生物技术通报, 2024, 40(3): 215-228. |
| [15] | 梁佳林, 赵爽, 李幸儿, 赵成周, 李萍. 尼泊尔黄堇Aux/IAA基因家族的鉴定与UVB处理下表达模式分析[J]. 生物技术通报, 2024, 40(12): 182-192. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||