








生物技术通报 ›› 2023, Vol. 39 ›› Issue (10): 281-291.doi: 10.13560/j.cnki.biotech.bull.1985.2023-0307
收稿日期:2023-04-06
出版日期:2023-10-26
发布日期:2023-11-28
通讯作者:
郑菲,女,博士,讲师,研究方向:微生物代谢与酶工程;E-mail: zhengfei0718@bjfu.edu.cn作者简介:杨俊钊,女,硕士研究生,研究方向:资源环境微生物学;E-mail: YJZbio@bjfu.edu.cn
基金资助:
YANG Jun-zhao( ), ZHANG Xin-rui, SUN Qing-yang, ZHENG Fei(
), ZHANG Xin-rui, SUN Qing-yang, ZHENG Fei( )
)
Received:2023-04-06
Published:2023-10-26
Online:2023-11-28
摘要:
糖苷水解酶第七家族(GH7)包含内切和外切两类纤维素酶,其中对外切纤维素酶的研究较为成熟,但对内切纤维素酶的研究相对较少。本研究从嗜热真菌Myceliophthora thermophila的基因组中鉴定出一个新型GH7内切纤维素酶MtCel7b,其在60℃,pH 5.0时表现出最佳酶活力。在90℃孵育1 h后,MtCel7b仍能保留40%以上的活性。经过序列统计分析发现,MtCel7b loop B3存在长链型和短链型的进化差异,为了探究loop B3对内切纤维素酶结构和功能的影响,将MtCel7b的长链型loop B3进行截短,构建了B3cut突变体。结果显示,B3cut突变体在高温下的稳定性较野生型提高了约9%-44%,而其对3种纤维素底物的比活性降低了34%-74%。借助分子动力学模拟进一步分析显示,在突变体B3cut中,其loop B3的截短导致催化裂隙两端的loop A3和loop B1发生了显著位移,缩小了催化口袋的空间结构,加强了催化位点周围的氢键作用网络,从而导致酶在高温下更加稳定。本研究阐明了loop B3在GH7内切纤维素酶中的重要作用,为酶分子的改良工作提供了新的参考。
杨俊钊, 张新蕊, 孙清扬, 郑菲. Loop B3对GH7内切纤维素酶功能的影响机制[J]. 生物技术通报, 2023, 39(10): 281-291.
YANG Jun-zhao, ZHANG Xin-rui, SUN Qing-yang, ZHENG Fei. Affecting Mechanism of Loop B3 on the Function of GH7 Endoglucanase[J]. Biotechnology Bulletin, 2023, 39(10): 281-291.
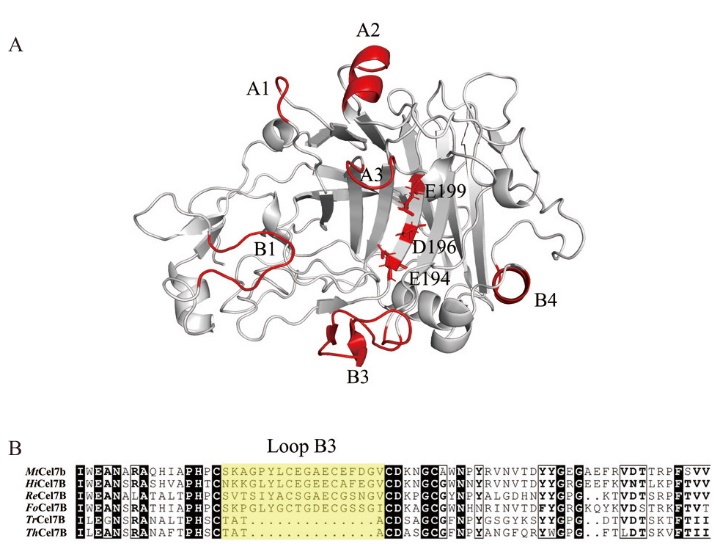
图1 MtCel7b结构模拟(A)及氨基酸序列对比(B) A:MtCel7b结构模拟图(红色区域为loop结构,E194、D196和E199为催化三联体);B:Loop B3区由黄色方框标记
Fig. 1 Structural simulation(A)and amino acid sequence alignment(B)of MtCel7b A: Modeled structure of MtCel7b, loop structure in red, E194, D196 and E199 are catalytic triplets. B: The Loop region are marked by a yellow box. The strain source and PDB ID of the alignment sequence are HiCel7B(H. insolens, 6YOZ); ReCel7B(R. emersonii CBS 394.64, 6SU8); FoCel7B(F. oxysporum, 1OVW); TrCel7B(T. reesei, 1EG1); ThCel7B(T. harzianum CBS 226.95, 5W0A)
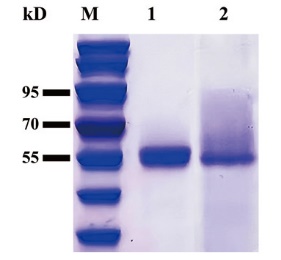
图2 野生型MtCel7b和突变体B3cut的SDS-PAGE分析 M:蛋白分子质量标准;1:野生型MtCel7b纯化蛋白;2:突变体B3cut纯化蛋白
Fig. 2 SDS-PAGE analysis of wild-type MtCel7b and mutant B3cut M: Protein molecular weight standard; 1: purified protein of wild-type MtCel7b; 2: purified protein of mutant B3cut
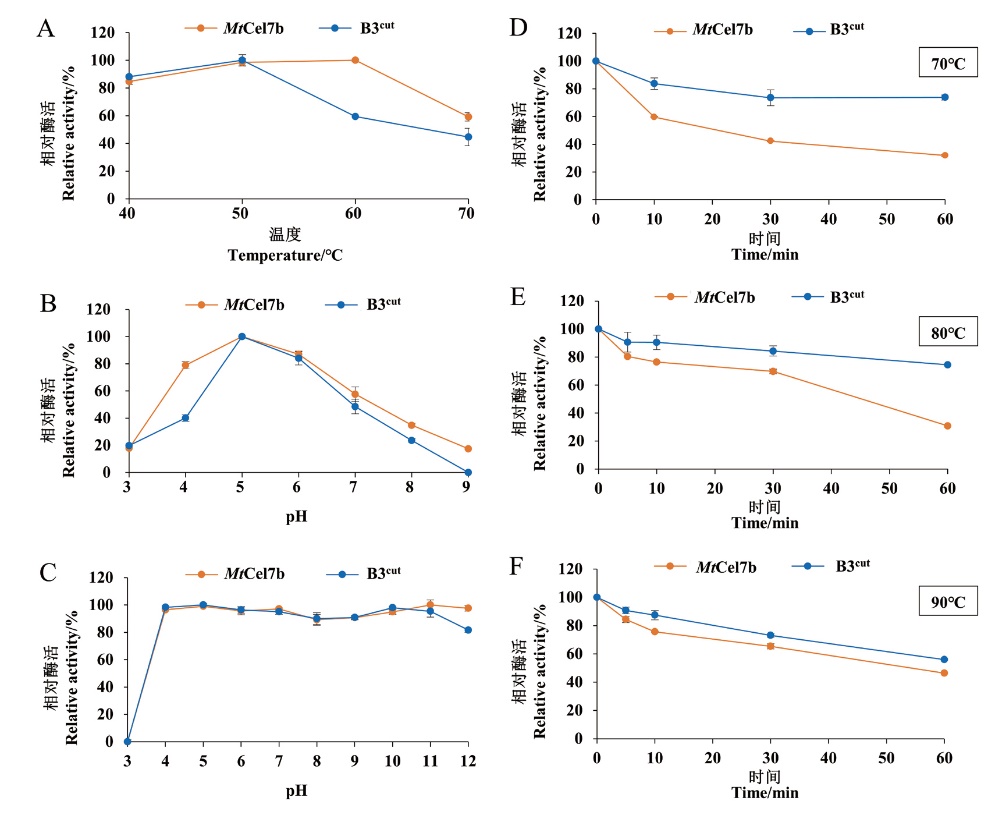
图3 野生型MtCel7b及突变体B3cut的酶学性质A:最适温度;B:最适pH;C:pH稳定性;D:70℃下温度稳定性;E:80℃下温度稳定性;F:90℃下温度稳定性
Fig. 3 Enzymatic properties of wild-type MtCel7b and mutant B3cut A: Optimal temperature-activity profile. B: Optimal pH. C: pH stability. D: Thermostability at 70℃. E: Thermostability at 80℃. F: Thermostability at 90℃
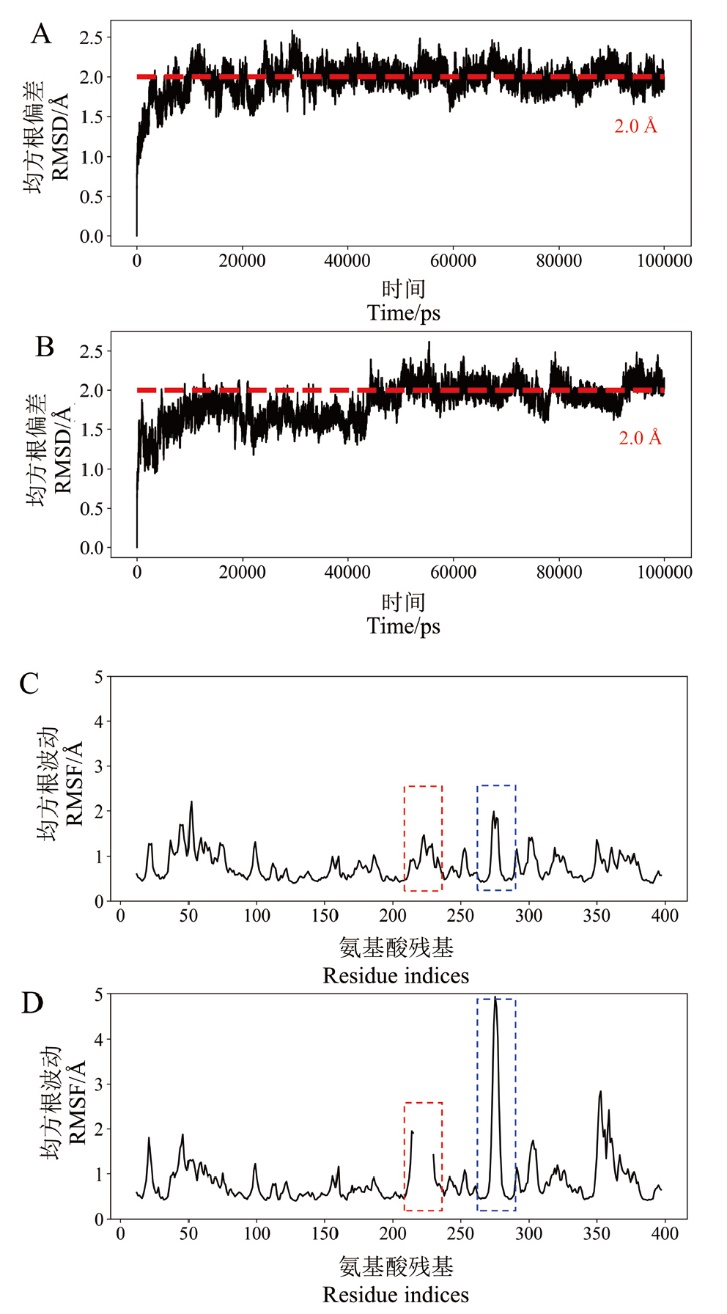
图6 野生型MtCel7b及突变体B3cut的均方根偏差和均方根波动图 A:野生型MtCel7b的均方根偏差图;B:突变体B3cut的均方根偏差图;C:野生型MtCel7b的均方根波动图;D:突变体B3cut的均方根波动图
Fig. 6 RMSD and RMSF of wild-type MtCel7b and mutant B3cut A: RMSD of wild-type MtCel7b; B: RMSD of mutant B3cut; C: RMSF of wild-type MtCel7b; D: RMSF of mutant B3cut

图7 野生型MtCel7b及突变体B3cut分子动力学模拟的轨迹主成分分析及平均结构图 A、B:野生型MtCel7b及突变体B3cut分子动力学模拟轨迹的主成分分析;C、D:野生型MtCel7b及突变体B3cut的蛋白质分子表面图;E、F:野生型MtCel7b及突变体B3cut催化裂隙周围氨基酸残基
Fig. 7 Trajectory principal component analysis plots and average structure of molecular dynamics simulations of wild-type MtCel7b and mutant B3cut A and B: Principal component analysis of the molecular dynamic simulation trajectory of wild-type MtCel7b and mutant B3cut. C and D: Protein surface of wild-type MtCel7b and mutant B3cut. E and F: Amino acid residues around the catalytic cleft of wild-type MtCel7b and mutant B3cut
| [1] |
Chen Y, Huang JW, Chen CC, et al. Crystallization and preliminary X-ray diffraction analysis of an endo-1,4-β-D-glucanase from Aspergillus aculeatus F-50[J]. Acta Crystallogr F Struct Biol Commun, 2015, 71(Pt 4): 397-400.
doi: 10.1107/S2053230X15003659 URL |
| [2] |
Bhat MK, Bhat S. Cellulose degrading enzymes and their potential industrial applications[J]. Biotechnol Adv, 1997, 15(3/4): 583-620.
doi: 10.1016/S0734-9750(97)00006-2 URL |
| [3] |
Kitamoto N, Go M, Shibayama T, et al. Molecular cloning, purification and characterization of two endo-1, 4-beta-glucanases from Aspergillus oryzae KBN616[J]. Appl Microbiol Biotechnol, 1996, 46(5/6): 538-544.
doi: 10.1007/s002530050857 URL |
| [4] |
Takashima S, Nakamura A, Hidaka M, et al. Cloning, sequencing, and expression of the cellulase genes of Humicola grisea var. thermoidea[J]. J Biotechnol, 1996, 50(2/3): 137-147.
doi: 10.1016/0168-1656(96)01555-6 URL |
| [5] |
Luo HY, Yang J, Yang PL, et al. Gene cloning and expression of a new acidic family 7 endo-beta-1, 3-1, 4-glucanase from the acidophilic fungus Bispora sp. MEY-1[J]. Appl Microbiol Biotechnol, 2010, 85(4): 1015-1023.
doi: 10.1007/s00253-009-2119-0 URL |
| [6] |
Bernardi AV, de Gouvêa PF, Gerolamo LE, et al. Functional characterization of GH7 endo-1,4-β-glucanase from Aspergillus fumigatus and its potential industrial application[J]. Protein Expr Purif, 2018, 150: 1-11.
doi: 10.1016/j.pep.2018.04.016 URL |
| [7] |
Kleywegt GJ, Zou JY, Divne C, et al. The crystal structure of the catalytic core domain of endoglucanase I from Trichoderma reesei at 3.6 Å resolution, and a comparison with related enzymes[J]. J Mol Biol, 1997, 272(3): 383-397.
pmid: 9325098 |
| [8] |
Sulzenbacher G, Driguez H, Henrissat B, et al. Structure of the Fusarium oxysporum endoglucanase I with a nonhydrolyzable substrate analogue: substrate distortion gives rise to the preferred axial orientation for the leaving group[J]. Biochemistry, 1996, 35(48): 15280-15287.
pmid: 8952478 |
| [9] |
MacKenzie LF, Sulzenbacher G, Divne C, et al. Crystal structure of the family 7 endoglucanase I(Cel7B)from Humicola insolens at 2.2 Å resolution and identification of the catalytic nucleophile by trapping of the covalent glycosyl-enzyme intermediate[J]. Biochem J, 1998, 335(Pt 2)(Pt 2):409-416.
doi: 10.1042/bj3350409 URL |
| [10] |
Sonoda MT, Godoy AS, Pellegrini VOA, et al. Structure and dynamics of Trichoderma harzianum Cel7B suggest molecular architecture adaptations required for a wide spectrum of activities on plant cell wall polysaccharides[J]. Biochim Biophys Acta Gen Subj, 2019, 1863(6): 1015-1026.
doi: 10.1016/j.bbagen.2019.03.013 URL |
| [11] |
Schiano-di-Cola C, Kołaczkowski B, Sørensen TH, et al. Structural and biochemical characterization of a family 7 highly thermostable endoglucanase from the fungus Rasamsonia emersonii[J]. FEBS J, 2020, 287(12): 2577-2596.
doi: 10.1111/febs.15151 pmid: 31755197 |
| [12] | 高小晓, 孟虹, 李蓉, 等. 糖苷水解酶7家族蛋白在纤维素降解中作用的研究进展[J]. 微生物学杂志, 2020, 40(6): 113-117. |
| Gao XX, Meng H, Li R, et al. Advances in cellulose degradation by glycoside hydrolase family 7 proteins[J]. J Microbiol, 2020, 40(6): 113-117. | |
| [13] |
Taylor CB, Payne CM, Himmel ME, et al. Binding site dynamics and aromatic-carbohydrate interactions in processive and non-processive family 7 glycoside hydrolases[J]. J Phys Chem B, 2013, 117(17): 4924-4933.
doi: 10.1021/jp401410h URL |
| [14] |
Borisova AS, Eneyskaya EV, Jana S, et al. Correlation of structure, function and protein dynamics in GH7 cellobiohydrolases from Trichoderma atroviride, T. reesei and T. harzianum[J]. Biotechnol Biofuels, 2018, 11: 5.
doi: 10.1186/s13068-017-1006-7 pmid: 29344086 |
| [15] | Yang H, Shi PJ, Liu Y, et al. Loop 3 of fungal endoglucanases of glycoside hydrolase family 12 modulates catalytic efficiency[J]. Appl Environ Microbiol, 2017, 83(6): e03123-e03116. |
| [16] |
Zheng F, Tu T, Wang XY, et al. Enhancing the catalytic activity of a novel GH5 cellulase GtCel5 from Gloeophyllum trabeum CBS 900.73 by site-directed mutagenesis on loop 6[J]. Biotechnol Biofuels, 2018, 11: 76.
doi: 10.1186/s13068-018-1080-5 pmid: 29588661 |
| [17] |
Kawamoto D, Takashima T, Fukamizo T, et al. A conserved loop structure of GH19 chitinases assists the enzyme function from behind the core-functional region[J]. Glycobiology, 2022, 32(4): 356-364.
doi: 10.1093/glycob/cwab117 URL |
| [18] |
Divne C, Ståhlberg J, Teeri TT, et al. High-resolution crystal structures reveal how a cellulose chain is bound in the 50 Å long tunnel of cellobiohydrolase I from Trichoderma reesei[J]. J Mol Biol, 1998, 275(2): 309-325.
doi: 10.1006/jmbi.1997.1437 pmid: 9466911 |
| [19] |
von Ossowski I, Ståhlberg J, Koivula A, et al. Engineering the exo-loop of Trichoderma reesei cellobiohydrolase, Cel7A. A comparison with Phanerochaete chrysosporium Cel7D[J]. J Mol Biol, 2003, 333(4): 817-829.
doi: 10.1016/s0022-2836(03)00881-7 pmid: 14568538 |
| [20] |
Karlsson J, Momcilovic D, Wittgren B, et al. Enzymatic degradation of carboxymethyl cellulose hydrolyzed by the endoglucanases Cel5A, Cel7B, and Cel45A from Humicola insolens and Cel7B, Cel12A and Cel45Acore from Trichoderma reesei[J]. Biopolymers, 2002, 63(1): 32-40.
pmid: 11754346 |
| [21] |
Dym O, Mevarech M, Sussman JL. Structural features that stabilize halophilic malate dehydrogenase from an archaebacterium[J]. Science, 1995, 267(5202): 1344-1346.
doi: 10.1126/science.267.5202.1344 pmid: 17812611 |
| [22] |
Karan R, Mathew S, Muhammad R, et al. Understanding high-salt and cold adaptation of a polyextremophilic enzyme[J]. Microorganisms, 2020, 8(10): 1594.
doi: 10.3390/microorganisms8101594 URL |
| [23] |
Zhao B, Al Rasheed H, Ali I, et al. Efficient enzymatic saccharification of alkaline and ionic liquid-pretreated bamboo by highly active extremozymes produced by the co-culture of two halophilic fungi[J]. Bioresour Technol, 2021, 319: 124115.
doi: 10.1016/j.biortech.2020.124115 URL |
| [24] | 刘欣, 魏雪, 王凤忠, 等. 极端酶研究进展及其在食品工业中的应用现状[J]. 生物产业技术, 2017(4): 62-69. |
| Liu X, Wei X, Wang FZ, et al. Research progress of extremozymes and its application in food industry[J]. Biotechnol Bus, 2017(4): 62-69. | |
| [25] |
You S, Li J, Zhang F, et al. Loop engineering of a thermostable GH10 xylanase to improve low-temperature catalytic performance for better synergistic biomass-degrading abilities[J]. Bioresour Technol, 2021, 342: 125962.
doi: 10.1016/j.biortech.2021.125962 URL |
| [1] | 张晶, 张浩睿, 曹云, 黄红英, 曲萍, 张志萍. 嗜热纤维素降解菌研究进展[J]. 生物技术通报, 2023, 39(6): 73-87. |
| [2] | 杨俊钊, 张新蕊, 赵国柱, 郑菲. 新型GH5家族多结构域纤维素酶的结构与功能研究[J]. 生物技术通报, 2023, 39(4): 71-80. |
| [3] | 陈泉冰, 曹伟洁, 李春, 吕波. GH79家族糖苷水解酶分子进化关系和蛋白结构研究[J]. 生物技术通报, 2023, 39(1): 104-114. |
| [4] | 吴娇, 余桂珍, 袁航, 刘娴, 高艳秀, 龚明, 邹竹荣. 融合超嗜热菌Pyrococcus furiosus红素氧还蛋白可提高靶蛋白的热稳定性[J]. 生物技术通报, 2021, 37(10): 110-119. |
| [5] | 刘登, 刘均洪. 嗜热性木质纤维素酶在纤维素乙醇生产中的研究进展[J]. 生物技术通报, 2020, 36(8): 185-193. |
| [6] | 曾静, 郭建军, 袁林, 杨罡, 陈俊. 极端嗜热α-淀粉酶ApkA的高温活性和热稳定性的优化研究[J]. 生物技术通报, 2017, 33(8): 192-198. |
| [7] | 郝梦圆, 曲晓军, 崔艳华. CRISPRs基因编辑技术研究进展[J]. 生物技术通报, 2017, 33(6): 45-53. |
| [8] | 张多多, 郑菲, 罗会颖, 李中媛, 罗学刚. 嗜热子囊菌JCM12803的α-半乳糖苷酶基因tcgal27A在毕赤酵母中的表达[J]. 生物技术通报, 2017, 33(6): 207-213. |
| [9] | 赵雪淞, 王冬旭, 刘鑫, 石倩倩, 史帅. 金属离子对中等嗜热混合菌活性的影响[J]. 生物技术通报, 2017, 33(12): 151-155. |
| [10] | 储引娣, 苏小运. 嗜热厌氧细菌Caldicellulosiruptor bescii降解木质纤维素研究进展[J]. 生物技术通报, 2017, 33(10): 33-39. |
| [11] | 曾静, 郭建军, 邱小忠, 王贤卓, 袁林. 极端嗜热微生物及其高温适应机制的研究进展[J]. 生物技术通报, 2015, 31(9): 30-37. |
| [12] | 田辉, 梁宏彰, 霍贵成, Evivie Smith Etareri. 嗜热链球菌的特性与应用研究进展[J]. 生物技术通报, 2015, 31(9): 38-48. |
| [13] | 刘毅, 姚粟, 李辉, 刘洋, 刘勇, 刘波, 程池. 三种DNA指纹图谱技术在菌株分型中的应用[J]. 生物技术通报, 2015, 31(6): 81-86. |
| [14] | 凌小芳;谢天;杨静;. 嗜热厌氧杆菌X514生长及代谢的初步研究[J]. , 2012, 0(09): 131-136. |
| [15] | 凌小芳;谢天;杨静;. 响应面法优化嗜热厌氧杆菌X514产乙醇合成培养基[J]. , 2012, 0(08): 168-174. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||