








生物技术通报 ›› 2024, Vol. 40 ›› Issue (12): 136-144.doi: 10.13560/j.cnki.biotech.bull.1985.2024-0488
吴双1,2( ), 逯锐琳2, 冯成天2, 袁坤2, 王真辉2, 刘进平1, 刘辉2(
), 逯锐琳2, 冯成天2, 袁坤2, 王真辉2, 刘进平1, 刘辉2( )
)
收稿日期:2024-05-23
出版日期:2024-12-26
发布日期:2025-01-15
通讯作者:
刘辉,男,博士,副研究员,研究方向:植物生理与分子生物学;E-mail: liuhui@catas.cn作者简介:吴双,女,硕士研究生,研究方向:植物分子生物学;E-mail: 605202618@qq.com
基金资助:
WU Shuang1,2( ), LU Rui-lin2, FENG Cheng-tian2, YUAN Kun2, WANG Zhen-hui2, LIU Jin-ping1, LIU Hui2(
), LU Rui-lin2, FENG Cheng-tian2, YUAN Kun2, WANG Zhen-hui2, LIU Jin-ping1, LIU Hui2( )
)
Received:2024-05-23
Published:2024-12-26
Online:2025-01-15
摘要:
【目的】克隆橡胶树硫氧还蛋白基因HbTRXh5,分析其表达特性,探究其在非生物胁迫中的功能,为橡胶树抗逆性遗传改良提供基因资源。【方法】采用RT-PCR方法克隆橡胶树HbTRXh5基因,利用生物信息学方法分析其序列特性和系统进化关系。采用实时荧光定量PCR分析HbTRXh5基因在橡胶树各组织以及非生物胁迫下的表达。构建HbTRXh5基因酵母表达载体并转入酵母,比较转基因酵母和对照酵母在低温、盐和氧化胁迫处理后的存活差异。【结果】HbTRXh5基因编码区长354 bp,编码117个氨基酸。HbTRXh5含有硫氧还蛋白保守结构域和CGPC活性位点,属于h型硫氧还蛋白第I亚组。HbTRXh5在橡胶树各组织中均有表达,以胶乳中的表达量最高。低温、盐以及H2O2和甲基紫精诱导的氧化胁迫处理均能诱导HbTRXh5的表达。成功将HbTRXh5基因转入酿酒酵母INVSc1菌株中并诱导表达。同转pYES2空载体对照酵母相比,转HbTRXh5基因酵母在H2O2处理后具有更高的存活率。相反,在低温和盐胁迫处理后,转HbTRXh5基因酵母的存活率较对照酵母明显降低。【结论】橡胶树HbTRXh5的表达受低温、盐和氧化胁迫调控,酵母中表达HbTRXh5提高了重组酵母对氧化胁迫的抗性,但降低了对低温和盐胁迫的抗性。
吴双, 逯锐琳, 冯成天, 袁坤, 王真辉, 刘进平, 刘辉. 橡胶树HbTRXh5基因在酵母中的表达及抗逆性分析[J]. 生物技术通报, 2024, 40(12): 136-144.
WU Shuang, LU Rui-lin, FENG Cheng-tian, YUAN Kun, WANG Zhen-hui, LIU Jin-ping, LIU Hui. Expression of HbTRXh5 Gene of Hevea brasiliensis in Yeast and Analysis on Its Resistance to Stress[J]. Biotechnology Bulletin, 2024, 40(12): 136-144.

图1 HbTRXh5基因编码区核苷酸序列及其编码的氨基酸序列 蓝色背景部分为硫氧还蛋白(TRX)结构域,下划线部分为CGPC活性中心
Fig. 1 Nucleotide sequence of HbTRXh5 gene coding region and its encoded amino acid sequence The blue background indicates the thioredoxin(TRX)domain, and the underlined one shows the highly conserved CGPC active site
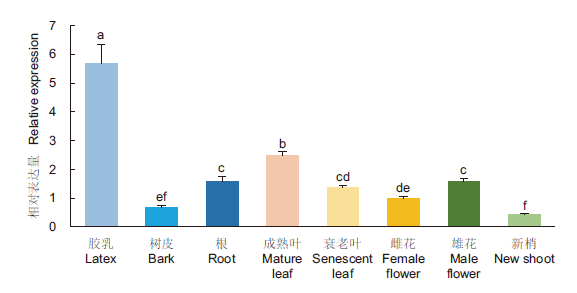
图3 HbTRXh5基因在橡胶树各组织中的表达 不同小写字母表示差异显著(P<0.05)。下同
Fig. 3 Expression of HbTRXh5 gene in various tissues of rubber tree(Hevea brasiliensis) Different lowercase letters indicate a significant difference(P<0.05). The same below
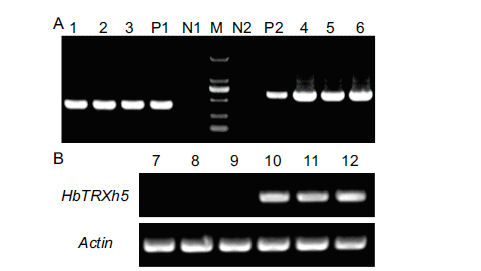
图5 pYES2或pYES2-HbTRXh5转化酵母的分子检测 A:pYES2或pYES2-HbTRXh5转化酵母的PCR检测;B:半定量RT-PCR检测HbTRXh5基因在转化酵母中的表达;M:DL2000 DNA marker(TaKaRa);N1和N2:未加模板的阴性对照;P1:pYES2质粒阳性对照;P2:pYES2-HbTRXh5质粒阳性对照;1-3和7-9:pYES2转化酵母单克隆;4-6和10-12:pYES2-HbTRXh5转化酵母单克隆
Fig. 5 Molecular detection of yeast transformed with pYES2 or pYES2-HbTRXh5 A: PCR detection of yeast transformed with pYES2 or pYES2-HbTRXh5. B: Semi-quantitative RT-PCR detection of HbTRXh5 gene expression in transformed yeast.M: DL2000 DNA marker(TaKaRa); N1 and N2: negative control without template; P1: pYES2 plasmid positive control; P2: pYES2-HbTRXh5 plasmid positive control; 1-3 and 7-9: pYES2 transformed yeast monoclonal; 4-6 and 10-12: pYES2-HbTRXh5 transformed yeast monoclonal
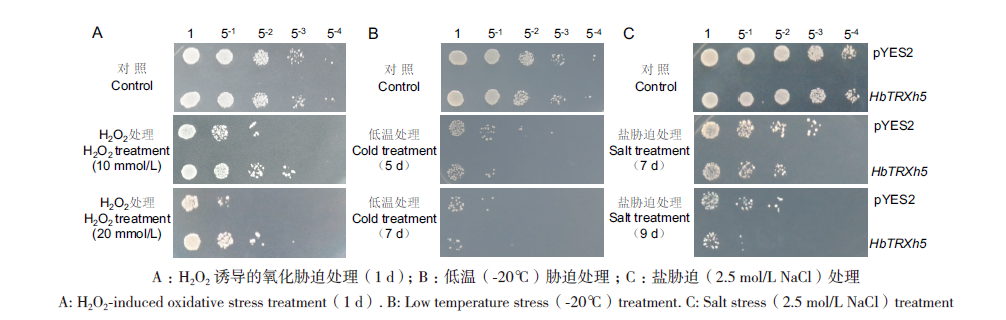
图6 非生物胁迫处理下转HbTRXh5基因酵母和转pYES2空载体对照酵母的存活差异 A:H2O2诱导的氧化胁迫处理(1 d);B:低温(-20℃)胁迫处理;C:盐胁迫(2.5 mol/L NaCl)处理
Fig. 6 Survival differences between HbTRXh5 transgenic yeast and control yeast transformed with pYES2 empty vector after abiotic stress treatment A: H2O2-induced oxidative stress treatment(1 d). B: Low temperature stress(-20℃)treatment. C: Salt stress(2.5 mol/L NaCl)treatment
| [1] | Sevilla F, Martí MC, De Brasi-Velasco S, et al. Redox regulation, thioredoxins, and glutaredoxins in retrograde signalling and gene transcription[J]. J Exp Bot, 2023, 74(19): 5955-5969. |
| [2] |
Geigenberger P, Thormählen I, Daloso DM, et al. The unprecedented versatility of the plant thioredoxin system[J]. Trends Plant Sci, 2017, 22(3): 249-262.
doi: S1360-1385(16)30221-7 pmid: 28139457 |
| [3] | Jiménez A, López-Martínez R, Martí MC, et al. The integration of TRX/GRX systems and phytohormonal signalling pathways in plant stress and development[J]. Plant Physiol Biochem, 2024, 207: 108298. |
| [4] |
Delorme-Hinoux V, Bangash SAK, Meyer AJ, et al. Nuclear thiol redox systems in plants[J]. Plant Sci, 2016, 243: 84-95.
doi: 10.1016/j.plantsci.2015.12.002 pmid: 26795153 |
| [5] | Meyer Y, Belin C, Delorme-Hinoux V, et al. Thioredoxin and glutaredoxin systems in plants: molecular mechanisms, crosstalks, and functional significance[J]. Antioxid Redox Signal, 2012, 17(8): 1124-1160. |
| [6] | Gelhaye E, Rouhier N, Jacquot JP. The thioredoxin h system of higher plants[J]. Plant Physiol Biochem, 2004, 42(4): 265-271. |
| [7] | Zuo XR, Yang C, Yan YN, et al. Systematic analysis of the thioredoxin gene family in Citrus sinensis: identification, phylogenetic analysis, and gene expression patterns[J]. Plant Signal Behav, 2023, 18(1): 2294426. |
| [8] | Chibani K, Wingsle G, Jacquot JP, et al. Comparative genomic study of the thioredoxin family in photosynthetic organisms with emphasis on Populus trichocarpa[J]. Mol Plant, 2009, 2(2): 308-322. |
| [9] | Boubakri H, Chihaoui SA, Najjar E, et al. Genome-wide analysis and expression profiling of H-type Trx family in Phaseolus vulgaris revealed distinctive isoforms associated with symbiotic N2-fixing performance and abiotic stress response[J]. J Plant Physiol, 2021, 260: 153410. |
| [10] | Ji MG, Park HJ, Cha JY, et al. Expression of Arabidopsis thaliana Thioredoxin-h2 in Brassica napus enhances antioxidant defenses and improves salt tolerance[J]. Plant Physiol Biochem, 2020, 147: 313-321. |
| [11] | Park JH, Lee ES, Chae HB, et al. Disulfide reductase activity of thioredoxin-h2 imparts cold tolerance in Arabidopsis[J]. Biochem Biophys Res Commun, 2021, 568: 124-130. |
| [12] |
闫宝琴, 陈燕桂, 史贤成, 等. 拟南芥AtTrx5增强植物菌核病的抗性研究[J]. 中国油料作物学报, 2023, 45(6): 1109-1118.
doi: 10.19802/j.issn.1007-9084.2023028 |
| Yan BQ, Chen YG, Shi XC, et al. Arabidopsis thioredoxin gene(AtTrx5)enhances plant resistance to Sclerotinia[J]. Chin J Oil Crop Sci, 2023, 45(6): 1109-1118. | |
| [13] |
Tian YC, Fan M, Qin ZX, et al. Hydrogen peroxide positively regulates brassinosteroid signaling through oxidation of the BRASSINAZOLE-RESISTANT1 transcription factor[J]. Nat Commun, 2018, 9: 1063.
doi: 10.1038/s41467-018-03463-x pmid: 29540799 |
| [14] | Zhai JL, Qi Q, Wang MQ, et al. Overexpression of tomato thioredoxin h(SlTrxh)enhances excess nitrate stress tolerance in transgenic tobacco interacting with SlPrx protein[J]. Plant Sci, 2022, 315: 111137. |
| [15] | 莫业勇, 杨琳. 2022年国内外天然橡胶产业发展情况和2023年形势分析[J]. 中国热带农业, 2023(4): 19-25, 70. |
| Mo YY, Yang L. Analysis of the development of domestic and foreign natural rubber industry in 2022 and the situation in 2023[J]. China Trop Agric, 2023(4): 19-25, 70. | |
| [16] | 刘琰琰, 韩冬, 杨菲, 等. 气象灾害对橡胶树的影响及风险评估综述[J]. 福建林业科技, 2016, 43(3): 244-252. |
| Liu YY, Han D, Yang F, et al. Studies for impact of meteorological disasters on Hevea brasiliensis and risk assessment[J]. J Fujian For Sci Technol, 2016, 43(3): 244-252. | |
| [17] |
王帅, 袁坤, 何其光, 等. 橡胶树硫氧还蛋白基因HbCXXS1的克隆及表达分析[J]. 生物技术通报, 2022, 38(12): 214-222.
doi: 10.13560/j.cnki.biotech.bull.1985.2022-0290 |
| Wang S, Yuan K, He QG, et al. Cloning and expression analysis of HbCXXS1, a thioredoxin gene in Hevea brasiliensis[J]. Biotechnol Bull, 2022, 38(12): 214-222. | |
| [18] |
李双江, 冯成天, 胡义钰, 等. 橡胶树HbDHAR2基因克隆及表达分析[J]. 华北农学报, 2021, 36(3): 25-32.
doi: 10.7668/hbnxb.20191875 |
| Li SJ, Feng CT, Hu YY, et al. Cloning and expression analysis of HbDHAR2 gene from Hevea brasiliensis[J]. Acta Agric Boreali Sin, 2021, 36(3): 25-32. | |
| [19] |
Li HP, Qin YX, Xiao XH, et al. Screening of valid reference genes for real-time RT-PCR data normalization in Hevea brasiliensis and expression validation of a sucrose transporter gene HbSUT3[J]. Plant Sci, 2011, 181(2): 132-139.
doi: 10.1016/j.plantsci.2011.04.014 pmid: 21683878 |
| [20] |
Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method[J]. Nat Protoc, 2008, 3: 1101-1108.
doi: 10.1038/nprot.2008.73 pmid: 18546601 |
| [21] | Nuruzzaman M, Sharoni AM, Satoh K, et al. The thioredoxin gene family in rice: genome-wide identification and expression profiling under different biotic and abiotic treatments[J]. Biochem Biophys Res Commun, 2012, 423(2): 417-423. |
| [22] | Zhou JF, Song TQ, Zhou HW, et al. Genome-wide identification, characterization, evolution, and expression pattern analyses of the typical thioredoxin gene family in wheat(Triticum aestivum L.)[J]. Front Plant Sci, 2022, 13: 1020584. |
| [23] | Zhang JR, Zhao T, Yan FD, et al. Genome-wide identification and expression analysis of thioredoxin(Trx)genes in seed development of Vitis vinifera[J]. J Plant Growth Regul, 2022, 41(7): 3030-3045. |
| [24] |
Zhang Y, Leclercq J, Montoro P. Reactive oxygen species in Hevea brasiliensis latex and relevance to tapping panel dryness[J]. Tree Physiol, 2017, 37(2): 261-269.
doi: 10.1093/treephys/tpw106 pmid: 27903918 |
| [25] | 李梦园, 樊亚栋, 张新宁, 等. 小麦TaTrxh9基因的序列特征及其对渗透胁迫的响应[J]. 西北植物学报, 2019, 39(5): 896-903. |
| Li MY, Fan YD, Zhang XN, et al. Sequence characteristics of TaTrxh9 gene and its response to osmotic stresses in wheat[J]. Acta Bot Boreali Occidentalia Sin, 2019, 39(5): 896-903. | |
| [26] | Xiang J, Li M, Li YY, et al. Overexpression of grapevine VyTRXy improves drought tolerance by maintaining photosynthesis and enhancing the antioxidant and osmolyte capacity of plants[J]. Int J Mol Sci, 2023, 24(22): 16388. |
| [27] |
刘辉, 邓治, 杨洪, 等. 橡胶树HbMC2在酵母中的表达和抗逆性分析[J]. 生物技术通报, 2018, 34(9): 202-208.
doi: 10.13560/j.cnki.biotech.bull.1985.2018-0298 |
| Liu H, Deng Z, Yang H, et al. Expression and stress tolerance analysis of HbMC2 gene from Hevea brasliensis in yeast[J]. Biotechnol Bull, 2018, 34(9): 202-208. | |
| [28] |
李博静, 郑腊梅, 吴乌云, 等. 西蒙得木HSP20基因家族的进化、表达和功能分析[J]. 生物技术通报, 2024, 40(6): 190-202.
doi: 10.13560/j.cnki.biotech.bull.1985.2024-0012 |
| Li BJ, Zheng LM, Wu WY, et al. expression, and functional analysis of the HSP20 gene family from Simmondisa chinensis[J]. Biotechnol Bull, 2024, 40(6):190-202. | |
| [29] |
胡尚钦, 汪军成, 姚立蓉, 等. 盐生草根系基因HgAKR6C的克隆与初步功能分析[J]. 草业学报, 2024, 33(1): 61-74.
doi: 10.11686/cyxb2023076 |
| Hu SQ, Wang JC, Yao LR, et al. Cloning and preliminary functional analysis of the root gene HgAKR6C of Halogeton glomeratus[J]. Acta Prataculturae Sin, 2024, 33(1): 61-74. | |
| [30] | Zhang CJ, Zhao BC, Ge WN, et al. An apoplastic h-type thioredoxin is involved in the stress response through regulation of the apoplastic reactive oxygen species in rice[J]. Plant Physiol, 2011, 157(4): 1884-1899. |
| [31] | Luan JY, Dong JX, Song X, et al. Overexpression of Tamarix hispida ThTrx5 confers salt tolerance to Arabidopsis by activating stress response signals[J]. Int J Mol Sci, 2020, 21(3): 1165. |
| [1] | 殷缘, 程爽, 刘定豪, 邓晓霞, 李凯月, 王竞红, 蔺吉祥. 外源过氧化氢(H2O2)影响非生物胁迫下植物生长与生理代谢机制的研究进展[J]. 生物技术通报, 2025, 41(1): 1-13. |
| [2] | 武志健, 刘广洋, 林志豪, 盛彬, 陈鸽, 许晓敏, 王军伟, 徐东辉. 蔬菜种子萌发的纳米调控及其机制研究进展[J]. 生物技术通报, 2025, 41(1): 14-24. |
| [3] | 李禹欣, 李苗, 杜晓芬, 韩康妮, 连世超, 王军. 谷子SiSAP基因家族的鉴定与表达分析[J]. 生物技术通报, 2025, 41(1): 143-156. |
| [4] | 申鹏, 高雅彬, 丁红. 马铃薯SAT基因家族的鉴定和表达分析[J]. 生物技术通报, 2024, 40(9): 64-73. |
| [5] | 满全财, 孟姿诺, 李伟, 蔡心汝, 苏润东, 付长青, 高顺娟, 崔江慧. 马铃薯AQP基因家族鉴定及表达分析[J]. 生物技术通报, 2024, 40(9): 51-63. |
| [6] | 李勇慧, 鲍星星, 段一珂, 赵运霞, 于相丽, 陈尧, 张延召. 灵宝杜鹃bZIP家族全基因组鉴定及表达特征分析[J]. 生物技术通报, 2024, 40(8): 186-198. |
| [7] | 崔原瑗, 王昭懿, 白双宇, 任毓昭, 豆飞飞, 刘彩霞, 刘凤楼, 王掌军, 李清峰. 大麦非特异性磷脂酶C基因家族全基因组鉴定及苗期胁迫表达分析[J]. 生物技术通报, 2024, 40(8): 74-82. |
| [8] | 刘丹丹, 王雷刚, 孙明慧, 焦小雨, 吴琼, 王文杰. 茶树海藻糖-6-磷酸合成酶(TPS)基因家族鉴定与表达分析[J]. 生物技术通报, 2024, 40(8): 152-163. |
| [9] | 余纽, 柳帆, 杨锦昌. 油楠SgTPS7的克隆及其在萜类生物合成和非生物胁迫中的功能[J]. 生物技术通报, 2024, 40(8): 164-173. |
| [10] | 吴丁洁, 陈盈盈, 徐静, 刘源, 张航, 李瑞丽. 植物赤霉素氧化酶及其功能研究进展[J]. 生物技术通报, 2024, 40(7): 43-54. |
| [11] | 胡雅丹, 伍国强, 刘晨, 魏明. MYB转录因子在调控植物响应逆境胁迫中的作用[J]. 生物技术通报, 2024, 40(6): 5-22. |
| [12] | 常雪瑞, 王田田, 王静. 辣椒E2基因家族的鉴定及分析[J]. 生物技术通报, 2024, 40(6): 238-250. |
| [13] | 李景艳, 周家婧, 袁媛, 苏晓艺, 乔文慧, 薛岩磊, 李国婧, 王瑞刚. 拟南芥AtiPGAM2基因参与非生物胁迫的响应[J]. 生物技术通报, 2024, 40(5): 215-224. |
| [14] | 杜兵帅, 邹昕蕙, 王子豪, 张馨元, 曹一博, 张凌云. 油茶SWEET基因家族的全基因组鉴定及表达分析[J]. 生物技术通报, 2024, 40(5): 179-190. |
| [15] | 郭慧妍, 董雪, 安梦楠, 夏子豪, 吴元华. 泛素化修饰关键酶在植物抗逆反应中的功能研究进展[J]. 生物技术通报, 2024, 40(4): 1-11. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||