








生物技术通报 ›› 2026, Vol. 42 ›› Issue (4): 17-25.doi: 10.13560/j.cnki.biotech.bull.1985.2025-0590
收稿日期:2025-06-07
出版日期:2026-04-26
发布日期:2026-04-30
通讯作者:
荆艳萍,女,博士,教授,博士生导师,研究方向 :植物分子细胞生物学;E-mail: ypjing@bjfu.edu.cn作者简介:闫琪琪,女,硕士研究生,研究方向 :植物分子细胞生物学;E-mail: 18514235629@163.com
基金资助:
YAN Qi-qi( ), BU Yu-fen, ZHANG Xiao-xin, MA Xiao-cen, JING Yan-ping(
), BU Yu-fen, ZHANG Xiao-xin, MA Xiao-cen, JING Yan-ping( )
)
Received:2025-06-07
Published:2026-04-26
Online:2026-04-30
摘要:
C2结构域蛋白(C2 domain proteins, C2DPs)是一类广泛存在于真核生物中的蛋白质,其典型特征是含有约130个氨基酸残基组成的C2结构域。植物C2结构域脱落酸相关蛋白(C2-domain abscisic acid (ABA)-related protein, CAR)属于C2DPs亚家族I,由一个C2结构域和一个植物特有的“sig”插入域组成。其中,C2结构域以Ca2+依赖的方式结合磷脂而介导蛋白膜定位;而“sig结构域”则使CAR蛋白可以结合不同种类的信号蛋白复合物,进而参与调控植物生长发育和逆境胁迫响应过程。本文系统综述了CAR蛋白的结构特征、组织表达模式和亚细胞定位规律,重点阐述了其在ABA信号转导、向光向重性生长、铁营养吸收、生物及非生物胁迫响应中的功能与作用机制,并对未来研究方向作出展望,以期为深入解析植物CAR蛋白的调控网络及其在作物抗逆遗传改良中的应用提供理论参考。
闫琪琪, 卜芋芬, 张小欣, 马晓岑, 荆艳萍. 植物C2结构域脱落酸相关蛋白的研究进展[J]. 生物技术通报, 2026, 42(4): 17-25.
YAN Qi-qi, BU Yu-fen, ZHANG Xiao-xin, MA Xiao-cen, JING Yan-ping. Advances in the Studies of Plant C2 Domain Abscisic Acid-related Protein[J]. Biotechnology Bulletin, 2026, 42(4): 17-25.
| 蛋白名称 Protein name | 物种 Species | 主要功能 Major functions | 参考文献 Reference |
|---|---|---|---|
| AtCAR1 | 拟南芥 | 参与ABA信号,增强ABA敏感性;负调控碱胁迫响应 | [ |
| AtCAR4 | 拟南芥 | 参与ABA信号;正调控盐胁迫与生物胁迫抗性 | [ |
| AtCAR5 | 拟南芥 | 参与ABA信号 | [ |
| AtCAR6/EHB1 | 拟南芥 | 负调控向光性与向重性;负调控铁吸收;负调控碱胁迫响应 | [ |
| AtCAR9 | 拟南芥 | 参与ABA信号;正调控干旱胁迫耐受性 | [ |
| AtCAR10 | 拟南芥 | 负调控碱胁迫响应 | [ |
| IbCAR1 | 甘薯 | 增强盐胁迫下细胞完整性,激活ROS清除系统 | [ |
| OsGAP1/CAR4 | 水稻 | 增强盐胁迫与生物胁迫抗性 | [ |
表1 不同植物CAR蛋白的主要功能
Table 1 Main functions of CAR proteins in different plants
| 蛋白名称 Protein name | 物种 Species | 主要功能 Major functions | 参考文献 Reference |
|---|---|---|---|
| AtCAR1 | 拟南芥 | 参与ABA信号,增强ABA敏感性;负调控碱胁迫响应 | [ |
| AtCAR4 | 拟南芥 | 参与ABA信号;正调控盐胁迫与生物胁迫抗性 | [ |
| AtCAR5 | 拟南芥 | 参与ABA信号 | [ |
| AtCAR6/EHB1 | 拟南芥 | 负调控向光性与向重性;负调控铁吸收;负调控碱胁迫响应 | [ |
| AtCAR9 | 拟南芥 | 参与ABA信号;正调控干旱胁迫耐受性 | [ |
| AtCAR10 | 拟南芥 | 负调控碱胁迫响应 | [ |
| IbCAR1 | 甘薯 | 增强盐胁迫下细胞完整性,激活ROS清除系统 | [ |
| OsGAP1/CAR4 | 水稻 | 增强盐胁迫与生物胁迫抗性 | [ |
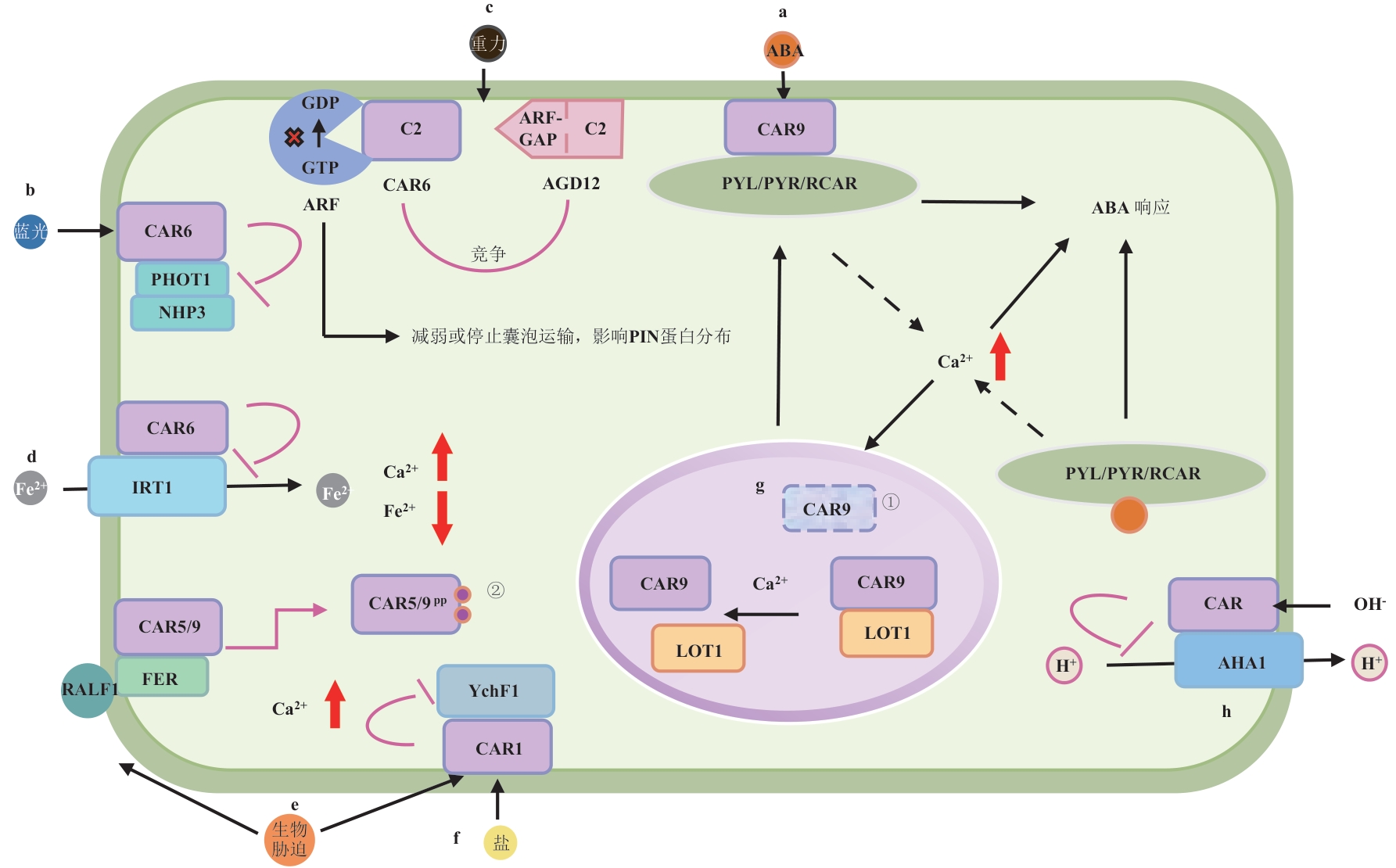
图2 CAR蛋白参与的调控网络及其生物学功能a和g:LOT1蛋白与CAR蛋白在细胞核中互作,抑制CAR蛋白泛素化修饰而维持其稳定性。受到干旱胁迫或者存在ABA信号时,ABA被细胞质或细胞核(未画出)中的PYR/PYL/RCAR受体(简称PYLs)识别,触发细胞质或细胞核中Ca²⁺浓度升高,降低了CAR蛋白与LOT1蛋白之间的亲和性,促使CAR蛋白转移至细胞膜,介导ABA受体PYLs到细胞膜上,响应ABA信号,增强植物的抗旱性[19-20];b:CAR与NPH3的BTB/POZ结构域结合,干扰了CUL3-NPH3复合体的组装及其介导的PHOT1泛素化降解,负向调控植物向光性[26];c:CAR蛋白与ARF-GAP家族成员AGD12竞争性结合囊泡运输相关小G蛋白ARF,阻碍AGD12对ARF的GTP酶激活作用,抑制ARF正常的GTP/GDP循环,影响囊泡运输,破坏PIN蛋白的极性定位,进而影响生长素分布而对向重性产生负面影响[27];d:CAR蛋白负调控植物铁转运蛋白IRT1,影响植物铁吸收[23];e:CAR蛋白负调控病原体感染负调节因子YchF1,解除其抑制作用而增强植物生物胁迫抗性[29],CAR蛋白可以增强脂筏结构稳定性,富集免疫相关蛋白,增强免疫信号,此外,与FER互作的CARs发生磷酸化,从膜上解离,维持膜上蛋白含量的动态平衡,避免免疫信号过度激活[25];f:CAR蛋白负调控盐胁迫负调节因子YchF1,增强植物耐盐性[32];h:CAR蛋白负调控质膜H+-ATPase酶AHA1活性,负调控植物耐碱性[28];红色箭头表示离子浓度上升(↑)或者下降(↓),图中①表示CAR蛋白的泛素化,②表示CAR蛋白的磷酸化
Fig. 2 Regulatory network and biological functions associated with CAR proteina & g: The LOT1 protein interacts with the CAR protein in the nucleus, inhibiting the ubiquitination modification of the CAR protein and thereby maintaining its stability. When subjected to drought stress or in the presence of ABA signaling, ABA is recognized by cytosolic or nuclear (not shown) PYR/PYL/RCAR receptors (abbreviated as PYLs). This recognition triggers an increase in cytosolic or nuclear Ca²⁺ concentration, which reduces the affinity between the CAR protein and the LOT1 protein. This promotes the relocation of the CAR protein to the plasma membrane, where it mediates the membrane recruitment of ABA receptor PYLs. This process enables the cell to respond to ABA signaling and enhances the plant’s drought resistance[19-20]; b: CAR binds to the BTB/POZ domain of NPH3, thereby impairing CUL3-NPH3 assembly, inhibiting ubiquitin-dependent degradation of PHOT1, and consequently attenuating plant phototropism[26]; c: CAR competitively inhibits ARF-GAP family members (e.g., AGD12) by binding to ADP-ribosylation factor (ARF) GTPases. This disrupts ARF GTP/GDP cycling, impairs PIN protein polar localization via vesicle trafficking defects, and compromises root gravitropism[27]; d: CAR protein negatively regulates the plant iron transporter IRT1, thereby affecting plant iron uptake[23]; e: CAR proteins negatively regulate YchF1-a negative regulator of pathogen infection-thereby relieving its inhibitory effect and enhancing plant resistance to biotic stresses[29]. Additionally, CAR proteins stabilize lipid raft structures, promoting the enrichment of immune-related proteins and strengthening immune signaling. Furthermore, CARs that interact with FER undergo phosphorylation, dissociate from the membrane, maintain the dynamic balance of membrane protein content, and prevent excessive activation of immune signaling[25]; f: CAR inhibits the salt stress negative regulator YchF1, conferring enhanced salinity tolerance[32]; h: CAR downregulates the plasma membrane H⁺-ATPase AHA1, thereby compromising plant tolerance to alkaline stress[28]. Red arrows indicate ion concentration increase (↑) or decrease (↓). Label ① marks ubiquitination of CAR, and ② indicates phosphorylation of CAR
| [1] | Ono Y, Kurokawa T, Kawahara K, et al. Cloning of rat brain protein kinase C complementary DNA [J]. FEBS Lett, 1986, 203(2): 111-115. |
| [2] | Parker PJ, Coussens L, Totty N, et al. The complete primary structure of protein kinase C-the major phorbol ester receptor [J]. Science, 1986, 233(4766): 853-859. |
| [3] | Ono Y, Kurokawa T, Fujii T, et al. Two types of complementary DNAs of rat brain protein kinase C Heterogeneity determined by alternative splicing [J]. FEBS Lett, 1986, 206(2): 347-352. |
| [4] | Geribaldi-Doldán N, Gómez-Oliva R, Domínguez-García S, et al. Protein kinase C: targets to regenerate brain injuries [J]. Front Cell Dev Biol, 2019, 7: 39. |
| [5] | Nishizuka Y. The molecular heterogeneity of protein kinase C and its implications for cellular regulation [J]. Nature, 1988, 334(6184): 661-665. |
| [6] | Bazzi MD, Nelsestuen GL. Protein kinase C interaction with calcium: a phospholipid-dependent process [J]. Biochemistry, 1990, 29(33): 7624-7630. |
| [7] | Bazzi MD, Nelsestuen GL. Association of protein kinase C with phospholipid vesicles [J]. Biochemistry, 1987, 26(1): 115-122. |
| [8] | Zhang DP, Aravind L. Identification of novel families and classification of the C2 domain superfamily elucidate the origin and evolution of membrane targeting activities in eukaryotes [J]. Gene, 2010, 469(1/2): 18-30. |
| [9] | 陈锦华. 拟南芥C2结构域蛋白的进化分析及其功能研究 [D]. 长沙: 湖南农业大学, 2019. |
| Chen JH. Analysis of evolution and function of C2 domain proteins in Arabidopsis [D]. Changsha: Hunan Agricultural University, 2019. | |
| [10] | Zhang HJ, Zeng YT, Seo J, et al. Global identification and characterization of C2 domain-containing proteins associated with abiotic stress response in rice (Oryza sativa L.) [J]. Int J Mol Sci, 2022, 23(4): 2221. |
| [11] | Zhao LY, Wang JZ, Zhou YT, et al. Genome-wide identification of plant C2 domain-containing protein family and the role of OsNTMC2T2.2 under chilling stress in rice [J]. Environ Exp Bot, 2025, 237: 106202. |
| [12] | Wang NN, Shi YY, Jiang Q, et al. A 14-3-3 protein positively regulates rice salt tolerance by stabilizing phospholipase C1 [J]. Plant Cell Environ, 2023, 46(4): 1232-1248. |
| [13] | Han SC, Wang YL, Li YX, et al. The OsNAC41-RoLe1-OsAGAP module promotes root development and drought resistance in upland rice [J]. Mol Plant, 2024, 17(10): 1573-1593. |
| [14] | Schneider R, Tang L, Lampugnani ER, et al. Two complementary mechanisms underpin cell wall patterning during xylem vessel development [J]. Plant Cell, 2017, 29(10): 2433-2449. |
| [15] | Sun Y, Zhao JY, Li YT, et al. Genome-wide analysis of the C2 domain family in soybean and identification of a putative abiotic stress response gene GmC2-148 [J]. Front Plant Sci, 2021, 12: 620544. |
| [16] | Niu JS, Li ZJ, Zhu JR, et al. Genome-wide identification and characterization of the C2 domain family in Sorghum bicolor (L.) and expression profiles in response to saline-alkali stress [J]. Physiol Mol Biol Plants, 2022, 28(9): 1695-1711. |
| [17] | 张兰军, 姬飞腾, 王丽丽, 等. 复苏植物旋蒴苣苔C2结构域小蛋白BhC2DP1参与植物对ABA的反应 [J]. 植物学报, 2012, 47(1): 11-27. |
| Zhang LJ, Ji FT, Wang LL, et al. A small C2-domain protein from the resurrection plant Boea hygrometrica promotes plant responses to abscisic acid [J]. Chin Bull Bot, 2012, 47(1): 11-27. | |
| [18] | Guo AY, Wu WQ, Bai D, et al. Recruitment of HAB1 and SnRK2.2 by C2-domain protein CAR1 in plasma membrane ABA signaling [J]. Plant J, 2024, 119(1): 237-251. |
| [19] | Rodriguez L, Gonzalez-Guzman M, Diaz M, et al. C2-domain abscisic acid-related proteins mediate the interaction of PYR/PYL/RCAR abscisic acid receptors with the plasma membrane and regulate abscisic acid sensitivity in Arabidopsis [J]. Plant Cell, 2014, 26(12): 4802-4820. |
| [20] | Qin T, Tian QZ, Wang GF, et al. LOWER TEMPERATURE 1 enhances ABA responses and plant drought tolerance by modulating the stability and localization of C2-domain ABA-related proteins in Arabidopsis [J]. Mol Plant, 2019, 12(9): 1243-1258. |
| [21] | Diaz M, Sanchez-Barrena MJ, Gonzalez-Rubio JM, et al. Calcium-dependent oligomerization of CAR proteins at cell membrane modulates ABA signaling [J]. Proc Natl Acad Sci USA, 2016, 113(3): E396-E405. |
| [22] | Rizo J, Südhof TC. C2-domains, structure and function of a universal Ca2+-binding domain [J]. J Biol Chem, 1998, 273(26): 15879-15882. |
| [23] | Khan I, Gratz R, Denezhkin P, et al. Calcium-promoted interaction between the C2-domain protein EHB1 and metal transporter IRT1 inhibits Arabidopsis iron acquisition [J]. Plant Physiol, 2019, 180(3): 1564-1581. |
| [24] | Cui MM, Gupta SK, Bauer P. Role of the plant-specific calcium-binding C2-domain abscisic acid-related (car) protein family in environmental signaling [J]. Eur J Cell Biol, 2023, 102(2): 151322. |
| [25] | Chen WJ, Zhou HN, Xu F, et al. CAR modulates plasma membrane nano-organization and immune signaling downstream of RALF1-FERONIA signaling pathway [J]. New Phytol, 2023, 237(6): 2148-2162. |
| [26] | Knauer T, Dümmer M, Landgraf F, et al. A negative effector of blue light-induced and gravitropic bending in Arabidopsis [J]. Plant Physiol, 2011, 156(1): 439-447. |
| [27] | Dümmer M, Michalski C, Essen LO, et al. EHB1 and AGD12, two calcium-dependent proteins affect gravitropism antagonistically in Arabidopsis thaliana [J]. J Plant Physiol, 2016, 206: 114-124. |
| [28] | Guo AY, Wu WQ, Liu WC, et al. C2-domain abscisic acid-related proteins regulate the dynamics of a plasma membrane H+-ATPase in response to alkali stress [J]. Plant Physiol, 2024, 196(4): 2784-2794. |
| [29] | Cheng SS, Ku YS, Cheung MY, et al. AtGAP1 promotes the resistance to Pseudomonas syringae pv. tomato DC3000 by regulating cell-wall thickness and stomatal aperture in Arabidopsis [J]. Int J Mol Sci, 2022, 23(14): 7540. |
| [30] | Cheung MY, Li MW, Yung YL, et al. The unconventional P-loop NTPase OsYchF1 and its regulator OsGAP1 play opposite roles in salinity stress tolerance [J]. Plant Cell Environ, 2013, 36(11): 2008-2020. |
| [31] | Cheung MY, Zeng NY, Tong SW, et al. Constitutive expression of a rice GTPase-activating protein induces defense responses [J]. New Phytol, 2008, 179(2): 530-545. |
| [32] | You C, Li C, Ma M, et al. A C2-domain abscisic acid-related gene, IbCAR1, positively enhances salt tolerance in sweet potato (Ipomoea batatas (L.) lam.) [J]. Int J Mol Sci, 2022, 23(17): 9680. |
| [33] | Cheung MY, Ngo JC, Chen ZZ, et al. A structure model explaining the binding between a ubiquitous unconventional G-protein (OsYchF1) and a plant-specific C2-domain protein (OsGAP1) from rice [J]. Biochem J, 2020, 477(20): 3935-3949. |
| [34] | Soon FF, Ng LM, Zhou XE, et al. Molecular mimicry regulates ABA signaling by SnRK2 kinases and PP2C phosphatases [J]. Science, 2012, 335(6064): 85-88. |
| [35] | Ma Y, Szostkiewicz I, Korte A, et al. Regulators of PP2C phosphatase activity function as abscisic acid sensors [J]. Science, 2009, 324(5930): 1064-1068. |
| [36] | Park SY, Fung P, Nishimura N, et al. Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins [J]. Science, 2009, 324(5930): 1068-1071. |
| [37] | Nishimura N, Sarkeshik A, Nito K, et al. PYR/PYL/RCAR family members are major in-vivo ABI1 protein phosphatase 2C-interacting proteins in Arabidopsis [J]. Plant J, 2010, 61(2): 290-299. |
| [38] | Christie JM, Suetsugu N, Sullivan S, et al. Shining light on the function of NPH3/RPT2-like proteins in phototropin signaling [J]. Plant Physiol, 2018, 176(2): 1015-1024. |
| [39] | Christie JM. Phototropin blue-light receptors [J]. Annu Rev Plant Biol, 2007, 58: 21-45. |
| [40] | Briggs WR, Christie JM. Phototropins 1 and 2: versatile plant blue-light receptors [J]. Trends Plant Sci, 2002, 7(5): 204-210. |
| [41] | Pedmale UV, Liscum E. Regulation of phototropic signaling in Arabidopsis via phosphorylation state changes in the phototropin 1-interacting protein NPH3 [J]. J Biol Chem, 2007, 282(27): 19992-20001. |
| [42] | Sakai T, Ken HG. Molecular genetic analysis of phototropism in Arabidopsis [J]. Plant Cell Physiol, 2012, 53(9): 1517-1534. |
| [43] | Vernoud V, Horton AC, Yang ZB, et al. Analysis of the small GTPase gene superfamily of Arabidopsis [J]. Plant Physiol, 2003, 131(3): 1191-1208. |
| [44] | Dümmer M, Forreiter C, Galland P. Gravitropism in Arabidopsis thaliana: Root-specific action of the EHB gene and violation of the resultant law [J]. J Plant Physiol, 2015, 189: 24-33. |
| [45] | Cointry V, Vert G. The bifunctional transporter-receptor IRT1 at the heart of metal sensing and signalling [J]. New Phytol, 2019, 223(3): 1173-1178. |
| [46] | Reyt G, Boudouf S, Boucherez J, et al. Iron- and ferritin-dependent reactive oxygen species distribution: impact on Arabidopsis root system architecture [J]. Mol Plant, 2015, 8(3): 439-453. |
| [47] | Vert G, Grotz N, Dédaldéchamp F, et al. IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth [J]. Plant Cell, 2002, 14(6): 1223-1233. |
| [48] | Yung YL, Cheung MY, Miao R, et al. Site-directed mutagenesis shows the significance of interactions with phospholipids and the G-protein OsYchF1 for the physiological functions of the rice GTPase-activating protein 1 (OsGAP1) [J]. J Biol Chem, 2015, 290(39): 23984-23996. |
| [49] | 强晓楠, 李鑫, 陈佳, 等. 拟南芥RALF多肽家族的功能多样性初步分析 [J]. 生物技术通报, 2019, 35(1): 2-10. |
| Qiang XN, Li X, Chen J, et al. Preliminary analysis of functional diversity of RALF peptide family in Arabidopsis thaliana [J]. Biotechnol Bull, 2019, 35(1): 2-10. | |
| [50] | 谌伟军. 拟南芥RALF-FER信号调控CAR家族介导膜筏形成及其初步功能研究 [D]. 长沙: 湖南大学, 2022. |
| Chen WJ. The receptor kinase FERONIA-CAR axis regulates proper lipid order formation and its preliminary function to response RALF signaling [D]. Changsha: Hunan University, 2022. | |
| [51] | Fu Q, Li HB, Wang BQ, et al. The RALF1 peptide-FERONIA complex phosphorylates the endosomal sorting protein FREE1 to attenuate abscisic acid signaling [J]. Plant Physiol, 2024, 197(1): kiae625. |
| [1] | 刘林娅, 刘欢艳, 梁鑫钰, 宋姝熠, 何斌, 王绪英, 黄亚成. ‘红阳’猕猴桃BGAL基因家族的全基因组鉴定与表达分析[J]. 生物技术通报, 2026, 42(3): 312-323. |
| [2] | 马莹莹, 游惠婉, 郑积荣, 汪俏梅, 刘丽红. 基于PSY多层级调控的园艺作物品质形成机制研究进展[J]. 生物技术通报, 2026, 42(3): 96-110. |
| [3] | 付涵, 孙书豪, 张思晴, 艾妞, 于杨, 于连伟, 王琼琼, 韩晓玉, 施艳, 韩卫丽, 杨雪. 本氏烟BOI家族基因的全基因组鉴定与表达分析[J]. 生物技术通报, 2026, 42(2): 207-217. |
| [4] | 淦晨露, 游雨婷, 谢菡萏, 曾子贤, 朱博. 植物黄素单加氧酶研究进展[J]. 生物技术通报, 2026, 42(1): 1-12. |
| [5] | 王从欢, 伍国强, 魏明. 植物CBL调控逆境胁迫响应的作用机制[J]. 生物技术通报, 2025, 41(7): 1-16. |
| [6] | 魏雨佳, 李岩, 康语涵, 弓晓楠, 杜敏, 涂岚, 石鹏, 于子涵, 孙彦, 张昆. 白颖苔草CrMYB4基因的克隆和表达分析[J]. 生物技术通报, 2025, 41(7): 248-260. |
| [7] | 昝舒雯, 谢欢欢, 张宇琴, 王文娟, 张鹏飞, 梁晋军, 温鹏飞. VvAGAMOUS通过VvCRABS CLAW调控葡萄心皮发育[J]. 生物技术通报, 2025, 41(5): 208-217. |
| [8] | 樊玥妮, 仙保山, 师艺萍, 任梦圆, 徐佳慧, 魏绍巍, 许晓敬, 罗晓峰, 舒凯. SPINDLY和SECRET AGENT介导的蛋白糖基化调控植物发育与逆境响应[J]. 生物技术通报, 2025, 41(4): 1-8. |
| [9] | 王斌, 林薇, 肖艳辉, 袁晓. 植物富含甘氨酸蛋白家族功能研究进展[J]. 生物技术通报, 2025, 41(2): 1-17. |
| [10] | 李晶晶, 胡进红, 梁旺利, 麻玉荣, 梁文裕, 王玲霞. ‘宁杞1号’响应NaCl胁迫相关基因的差异表达分析[J]. 生物技术通报, 2025, 41(2): 202-209. |
| [11] | 方玉洁, 刘宽, 崔寒冰, 王幼平. 植物SUMO E3连接酶研究进展[J]. 生物技术通报, 2025, 41(12): 1-15. |
| [12] | 吴丁洁, 陈盈盈, 徐静, 刘源, 张航, 李瑞丽. 植物赤霉素氧化酶及其功能研究进展[J]. 生物技术通报, 2024, 40(7): 43-54. |
| [13] | 郝思怡, 张君珂, 王斌, 曲朋燕, 李瑞得, 程春振. 香蕉ELF3的克隆与表达分析[J]. 生物技术通报, 2024, 40(5): 131-140. |
| [14] | 陈盈盈, 吴丁洁, 刘源, 张航, 刘艳娇, 王晶宇, 李瑞丽. 14-3-3蛋白及其在植物中的功能研究进展[J]. 生物技术通报, 2024, 40(4): 12-22. |
| [15] | 付威, 韦素云, 陈赢男. 植物生长发育动态QTL解析研究进展[J]. 生物技术通报, 2024, 40(2): 9-19. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||