








• 研究报告 • 下一篇
陈义焰1,2, 张冬儿1,2, 张涛1,2, 刘育灏1,2, 唐杰2, 盛夏冰2,3, 胡远艺2,3, 艾治勇2,3, 李应将2( ), 刘小林2,3(
), 刘小林2,3( )
)
收稿日期:2025-10-02
出版日期:2026-03-02
通讯作者:
刘小林liuxiaolin@hhrrc.ac.cn基金资助:
CHEN Yi-yan1,2, ZHANG Dong-er1,2, ZHANG Tao1,2, LIU Yu-hao1,2, TANG Jie2, SHENG Xia-bing2,3, HU Yuan-yi2,3, AI Zhi-yong2,3, LI Ying-jiang2( ), LIU Xiao-lin2,3(
), LIU Xiao-lin2,3( )
)
Received:2025-10-02
Published:2026-03-02
摘要:
目的 探究OsSULTR2;2基因功能及其调控水稻苗期耐盐性的机制。 方法 以籼稻品种晶4155S为材料,从盐处理前后根部转录组数据中筛选出响应基因OsSULTR2;2;通过RT-qPCR研究该基因表达模式;构建启动子GUS报告载体和CRISPR/Cas9敲除载体;使用RiceXPro和RGAP在线数据库预测基因及启动子表达模式,通过GUS化学染色分析基因启动子活性;在140 mmol/L NaCl条件下,统计敲除株系与野生型的存活率和株高;并通过NBT和DAB染色比较盐胁迫下敲除株系与野生型叶片超氧阴离子(O2-)和过氧化氢(H2O2)的积累情况。 结果 OsSULTR2;2在盐处理后转录水平发生显著上调,并在水稻的各个组织中都能观察到GUS信号;盐胁迫下,OsSULTR2;2敲除株系的存活率和株高均显著高于野生型,且其活性氧(ROS)积累量显著低于野生型。 结论 在水稻中敲除OsSULTR2;2基因可增强水稻的活性氧清除能力,提高转基因水稻的耐盐性,为创制耐盐水稻新种质提供参考。
陈义焰, 张冬儿, 张涛, 刘育灏, 唐杰, 盛夏冰, 胡远艺, 艾治勇, 李应将, 刘小林. 水稻OsSULTR2;2基因功能及调控苗期耐盐性分析[J]. 生物技术通报, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1144.
CHEN Yi-yan, ZHANG Dong-er, ZHANG Tao, LIU Yu-hao, TANG Jie, SHENG Xia-bing, HU Yuan-yi, AI Zhi-yong, LI Ying-jiang, LIU Xiao-lin. Analysis on Function of Rice OsSULTR2;2 and Regulation of Seedling Tolerance to Salt[J]. Biotechnology Bulletin, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1144.
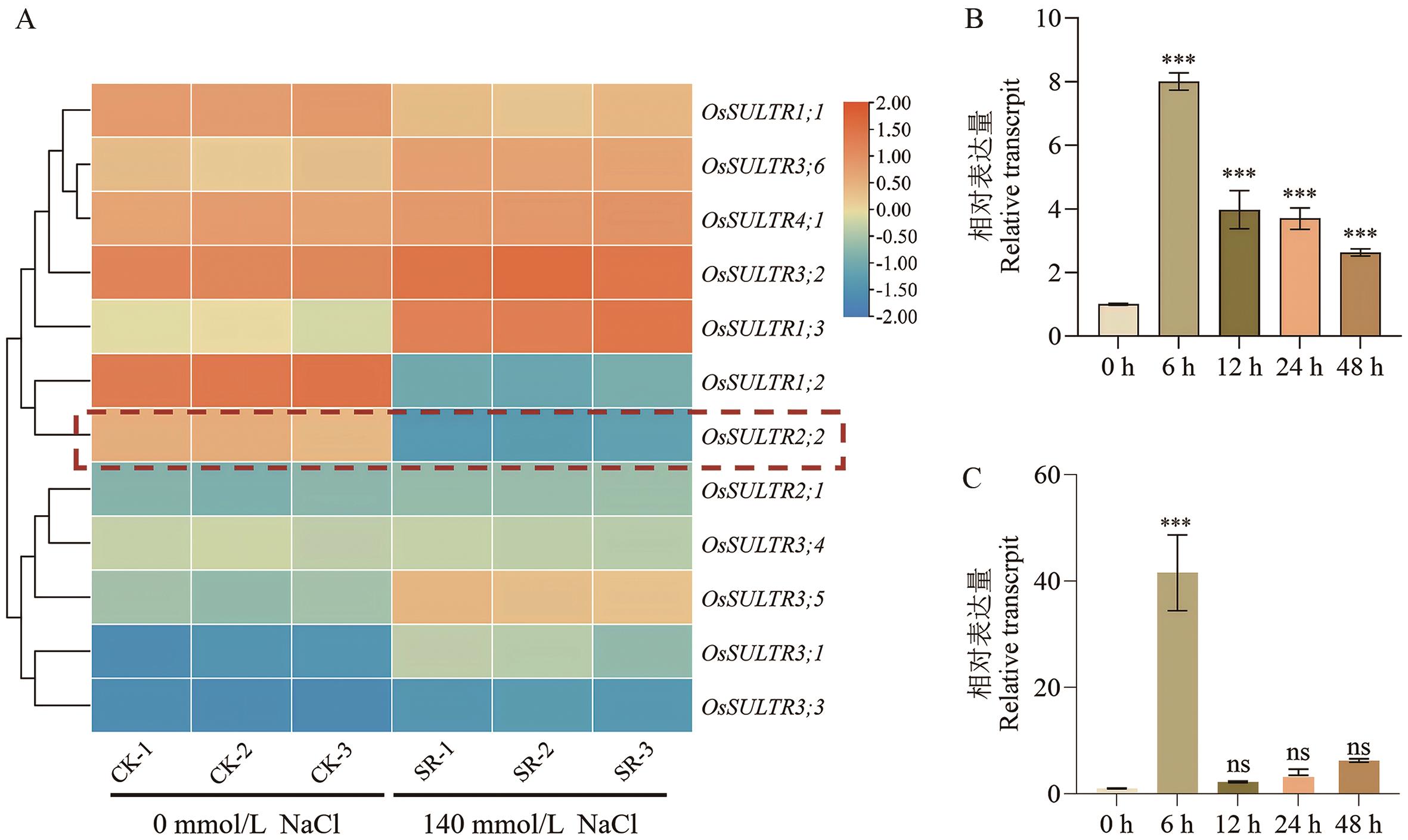
图1 水稻硫酸盐转运蛋白基因家族响应及OsSULTR2;2表达分析A:盐胁迫下晶4155S根部硫酸盐转运蛋白基因家族的转录组分析;B:14 d苗期ZH11经140 mmol/L NaCl处理0-48 h后叶中OsSULTR2;2的表达水平;C:14 d苗期ZH11经140 mmol/L NaCl处理0-48 h后根中OsSULTR2;2的表达水平。***表示处理与对照之间差异极显著(t-test,P<0.001)
Fig. 1 Response of rice (Oryza sativa L.) sulfate transporter gene family and expression analysis of OsSULTR2;2A: Transcriptome analysis of the sulfate transporter gene family in the roots of ‘Jing 4155S’ under salt stress. B: Expression of OsSULTR2;2 in the leaves of 14-day-old ZH11 after treatment with 140 mmol/L NaCl for 0-48 h. C: Expression of OsSULTR2;2 in the roots of 14-day-old ZH11 after treatment with 140 mmol/L NaCl for 0-48 h. *** indicates statistical significance between the treatment and the mock (t-test, P<0.001)
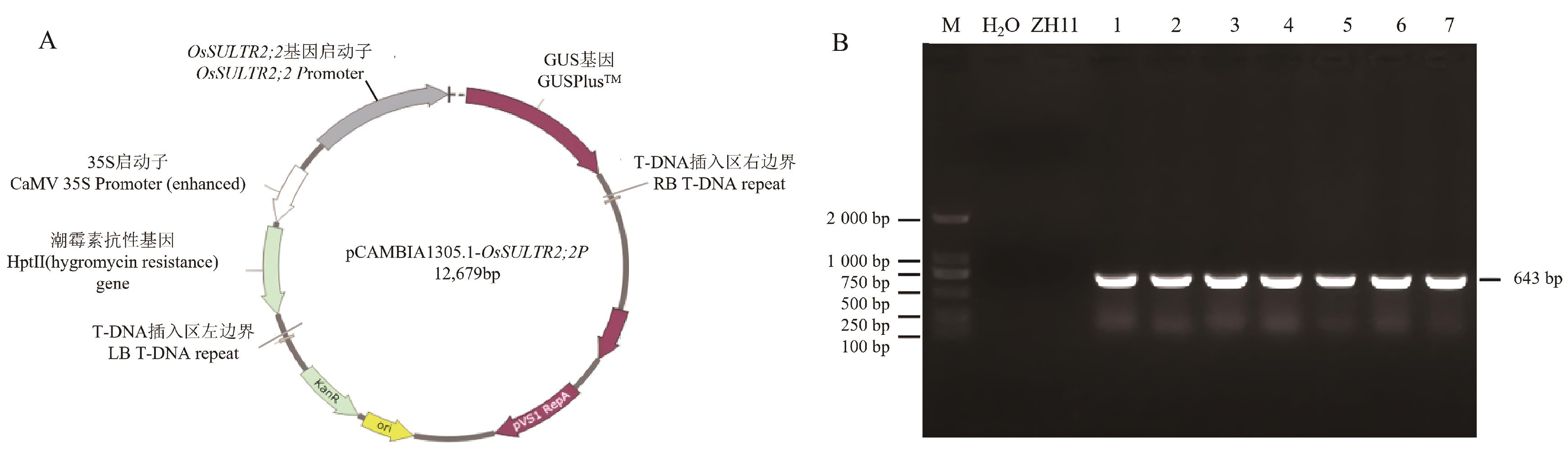
图3 OsSULTR2;2Promoter::GUS载体构建示意图及转基因植株的分子检测A:OsSULTR2;2Promoter::GUS载体构建示意图;B:转OsSULTR2;2Promoter::GUS T0代转基因水稻PCR鉴定;M:DL2000 DNA Marker;H2O:超纯水;ZH11:阴性对照;1:阳性对照;2-7:OsSULTR2;2Promoter::GUS转基因阳性植株
Fig. 3 Schematic diagram of OsSULTR2;2Promoter::GUS vector construction and molecular detection of transgenic plantsA: Schematic diagram of OsSULTR2;2Promoter::GUS vector construction. B:PCR identification of T0 generation transgenic rice harboring OsSULTR2;2Promoter::GUS. M: DL2000 DNA Marker; H₂O: ultrapure water; ZH11: negative control; 1: positive control; 2-7: OsSULTR2;2Promoter::GUS positive transgenic plants
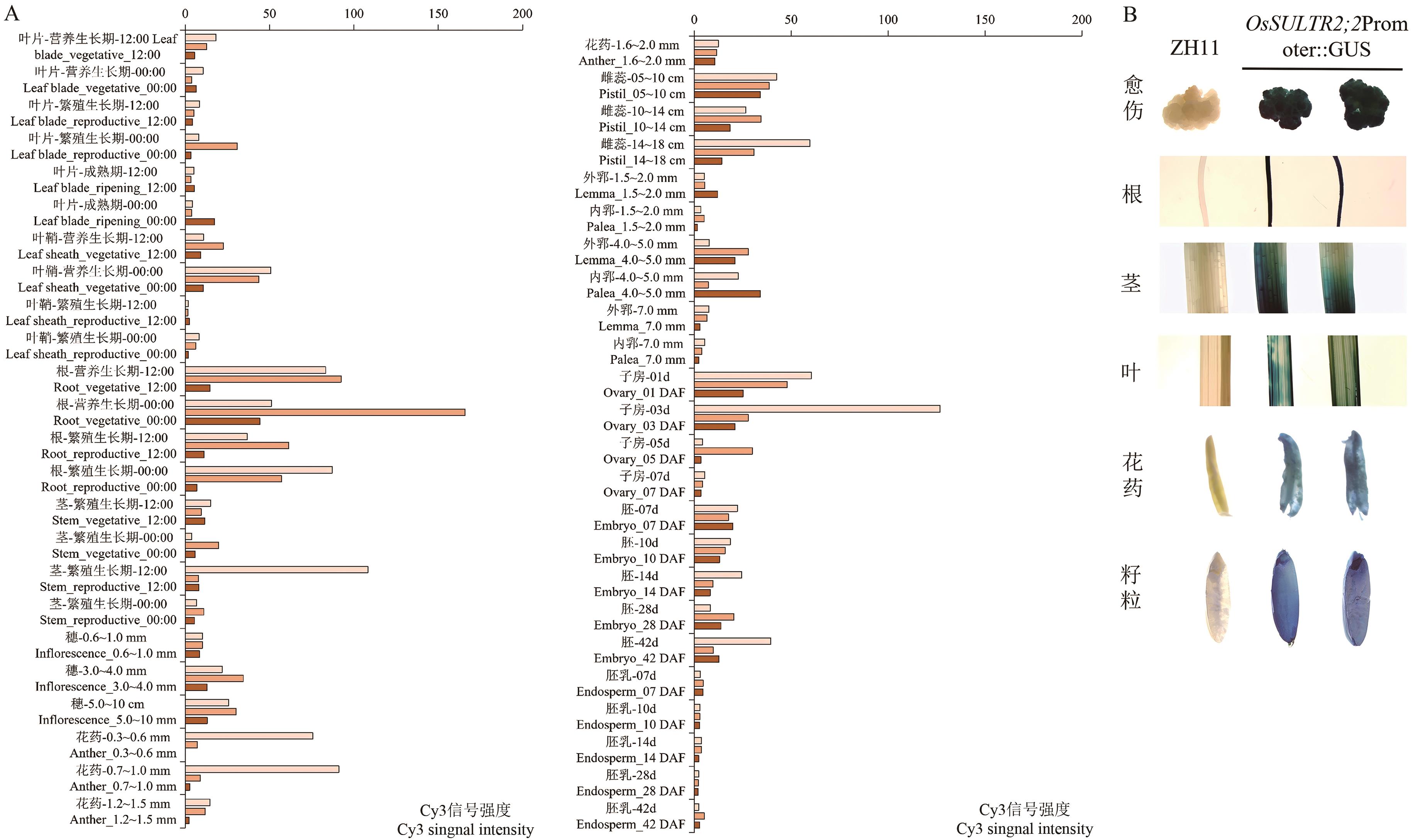
图4 OsSULTR2;2器官水平表达谱及转基因水稻OsSULTR2;2Promoter::GUS不同器官与组织GUS染色A:OsSULTR2;2器官水平表达谱;B:转基因水稻OsSULTR2;2Promoter::GUS不同器官与组织的GUS染色;ZH11:阴性对照。图中标尺为1 mm
Fig. 4 Organ-level expression profile of OsSULTR2;2 and GUS staining of different organs and tissues in transgenic rice carrying OsSULTR2;2Promoter::GUSA: Organ-level expression profile of OsSULTR2;2. B: GUS staining of different organs and tissues in transgenic rice with OsSULTR2;2Promoter::GUS. ZH11: Negative control. Bars are 1 mm
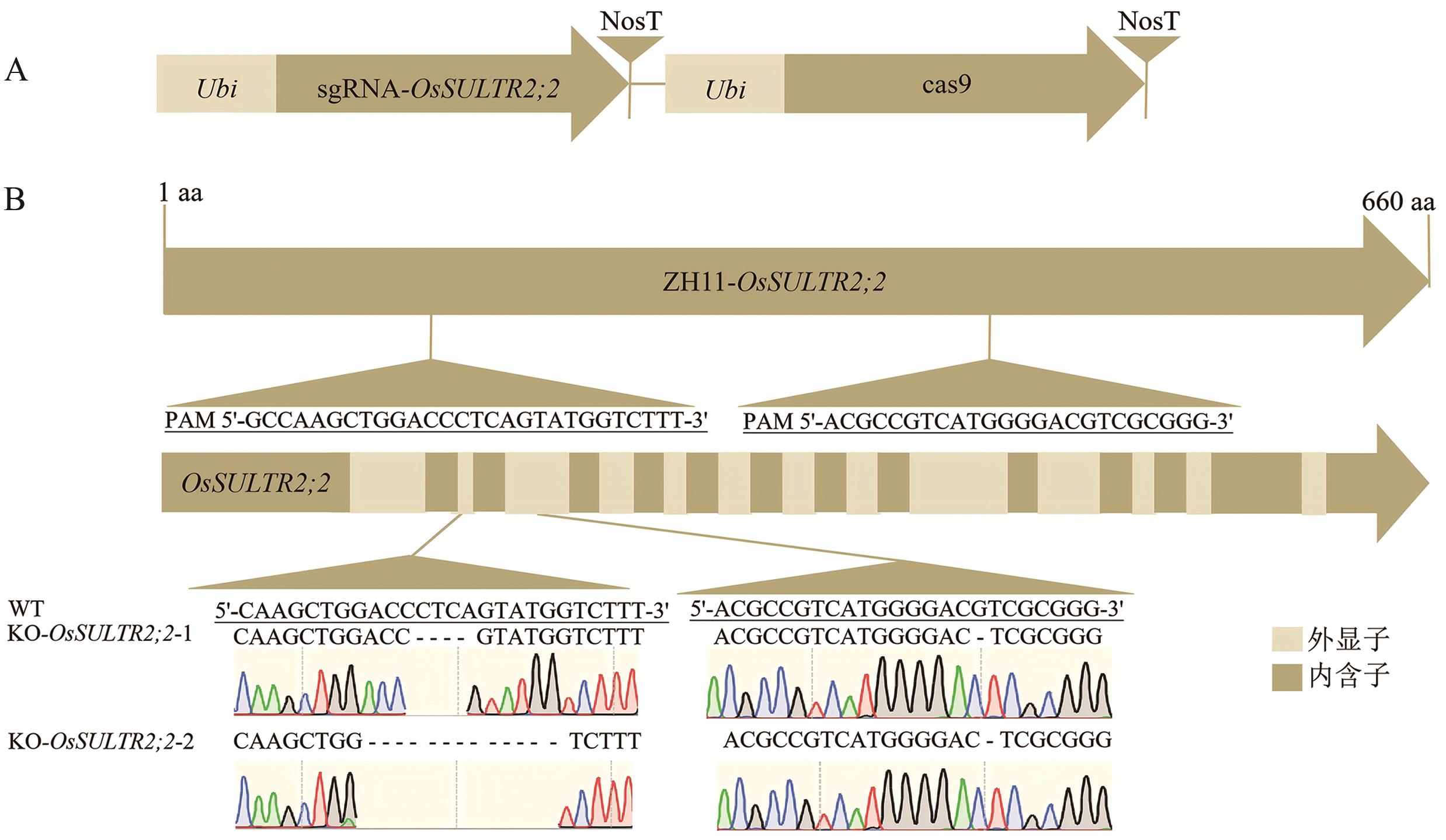
图5 OsSULTR2;2基因的转基因材料分析A:CRISPR/Cas9敲除载体sgRNA-OsSULTR2;2-cas9示意图;B:CRISPR/Cas9敲除材料KO-OsSULTR2;2-1测序结果显示在第一靶点位置缺失CTCA碱基,第二靶点位置缺失G碱基。KO-OsSULTR2;2-2测序结果显示在第一靶点位置缺失ACCCTCAGTATGG碱基,第二靶点位置缺失G碱基。导致OsSULTR2;2在突变体中ORF提前终止。其中米色方块表示外显子,卡其色方块表示内含子
Fig. 5 Analysis of transgenic materials for gene OsSULTR2;2A: Schematic diagram of the CRISPR/Cas9 knockout vector sgRNA-OsSULTR2;2-cas9. B: Sequencing results of the CRISPR/Cas9 knockout material KO-OsSULTR2;2-1 showed a deletion of the CTCA base at the first target site and a deletion of the G base at the second target site; sequencing results of KO-OsSULTR2;2-2 showed a deletion of the ACCCTCAGTATGG base at the first target site and a deletion of the G base at the second target site. These deletions led to the premature termination of the open reading frame (ORF) of OsSULTR2;2 in the mutants. Among them, the light brown squares indicate exons, and the light khaki squares indicate introns
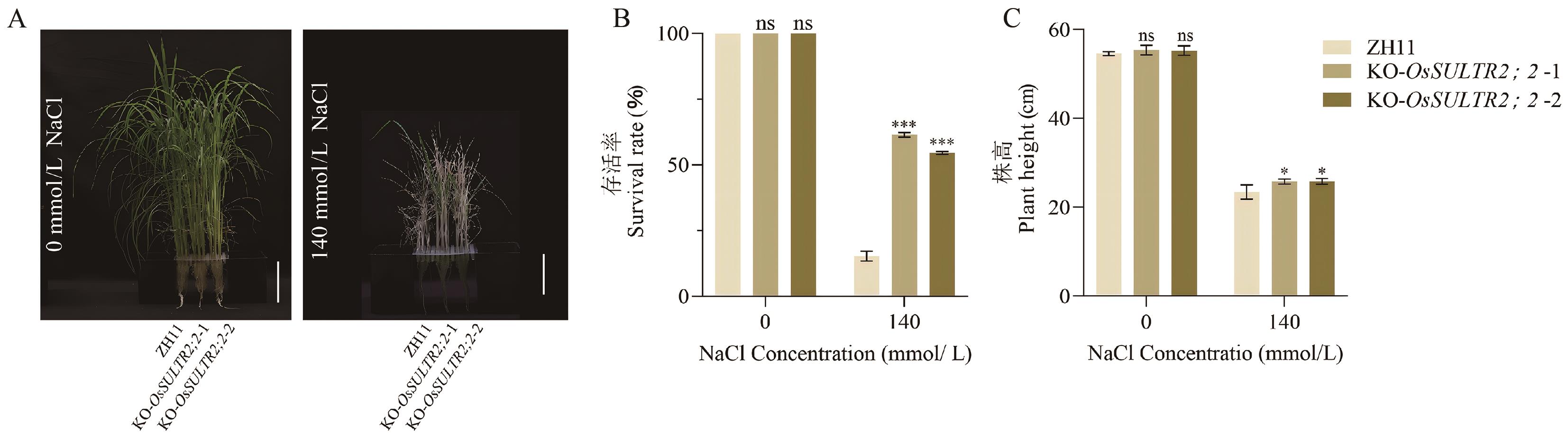
图6 OsSULTR2;2敲除纯合突变体水稻苗期耐盐性分析A:各株系在NaCl处理前后的生长状况;B:水稻幼苗的存活率;C:水稻幼苗的株高;差异显著性分析通过t检验完成,*P < 0.05,***P < 0.001, n = 24。图中标尺为10 cm
Fig. 6 Analysis of salt tolerance at the seedling stage of OsSULTR2;2 knockout homozygous mutant riceA: Growth status of each line before and after NaCl treatment. B: Survival rate of rice seedlings. C: Plant height of rice seedlings. Significant difference analysis was performed by t-test, with *: P < 0.05 and ***: P < 0.001, n = 24. Bars are 10 cm

图7 OsSULTR2;2敲除株系盐胁迫下ROS积累检测A:NBT染色结果;B:DAB染色结果。标尺长度为1 mm
Fig. 7 Detection of ROS accumulation in OsSULTR2;2 knockout lines under salt stressA: NBT staining results. B: DAB staining results. The scale bar is 1 mm
| [1] | Munns R, Tester M. Mechanisms of salinity tolerance [J]. Annu Rev Plant Biol, 2008, 59: 651-681. |
| [2] | Navada S, Vadstein O, Gaumet F, et al. Biofilms remember: Osmotic stress priming as a microbial management strategy for improving salinity acclimation in nitrifying biofilms [J]. Water Res, 2020, 176: 115732. |
| [3] | 王甜甜, 郝怀庆, 冯雪, 等. 植物HKT蛋白耐盐机制研究进展 [J]. 植物学报, 2018, 53(5): 710-725. |
| Wang TT, Hao HQ, Feng X, et al. Research advances in the function of the high-affinity K+ transporter (HKT) proteins and plant salt tolerance [J]. Chin Bull Bot, 2018, 53(5): 710-725. | |
| [4] | Almeida DM, Oliveira MM, Saibo NJM. Regulation of Na+ and K+ homeostasis in plants: towards improved salt stress tolerance in crop plants [J]. Genet Mol Biol, 2017, 40(1 ): 326-345. |
| [5] | Pan T, Liu MM, Kreslavski VD, et al. Non-stomatal limitation of photosynthesis by soil salinity [J]. Crit Rev Environ Sci Technol, 2021, 51(8): 791-825. |
| [6] | Ye SH, Huang ZB, Zhao GB, et al. Differential physiological responses to salt stress between salt-sensitive and salt-tolerant Japonica rice cultivars at the post-germination and seedling stages [J]. Plants, 2021, 10(11): 2433. |
| [7] | Takahashi H. Sulfate transport systems in plants: functional diversity and molecular mechanisms underlying regulatory coordination [J]. J Exp Bot, 2019, 70(16): 4075-4087. |
| [8] | Akbudak MA, Filiz E, Kontbay K. Genome-wide identification and cadmium induced expression profiling of sulfate transporter (SULTR) genes in sorghum (Sorghum bicolor L.) [J]. BioMetals, 2018, 31(1): 91-105. |
| [9] | Vatansever R, Koc I, Ozyigit II, et al. Genome-wide identification and expression analysis of sulfate transporter (SULTR) genes in potato (Solanum tuberosum L.) [J]. Planta, 2016, 244(6): 1167-1183. |
| [10] | Honsel A, Kojima M, Haas R, et al. Sulphur limitation and early sulphur deficiency responses in poplar: significance of gene expression, metabolites, and plant hormones [J]. J Exp Bot, 2012, 63(5): 1873-1893. |
| [11] | Heidari P, Hasanzadeh S, Faraji S, et al. Genome-wide characterization of the sulfate transporter gene family in oilseed crops: Camelina sativa and Brassica napus [J]. Plants, 2023, 12(3): 628. |
| [12] | Yoshimoto N, Takahashi H, Smith FW, et al. Two distinct high-affinity sulfate transporters with different inducibilities mediate uptake of sulfate in Arabidopsis roots [J]. Plant J, 2002, 29(4): 465-473. |
| [13] | Rouached H, Wirtz M, Alary R, et al. Differential regulation of the expression of two high-affinity sulfate transporters, SULTR1.1 and SULTR1.2, in Arabidopsis [J]. Plant Physiol, 2008, 147(2): 897-911. |
| [14] | Yoshimoto N, Inoue E, Saito K, et al. Phloem-localizing sulfate transporter, Sultr1;3, mediates re-distribution of sulfur from source to sink organs in Arabidopsis [J]. Plant Physiol, 2003, 131(4): 1511-1517. |
| [15] | Liu X, Wu FH, Li JX, et al. Glutathione homeostasis and Cd tolerance in the Arabidopsis sultr1;1-sultr1;2 double mutant with limiting sulfate supply [J]. Plant Cell Rep, 2016, 35(2): 397-413. |
| [16] | Takahashi H, Watanabe-Takahashi A, Smith FW, et al. The roles of three functional sulphate transporters involved in uptake and translocation of sulphate in Arabidopsis thaliana [J]. Plant J, 2000, 23(2): 171-182. |
| [17] | Gallardo K, Courty PE, Le Signor C, et al. Sulfate transporters in the plant’s response to drought and salinity: regulation and possible functions [J]. Front Plant Sci, 2014, 5: 580. |
| [18] | Kataoka T, Hayashi N, Yamaya T, et al. Root-to-shoot transport of sulfate in Arabidopsis. evidence for the role of SULTR3;5 as a component of low-affinity sulfate transport system in the root vasculature [J]. Plant Physiol, 2004, 136(4): 4198-4204. |
| [19] | Kumar S, Asif MH, Chakrabarty D, et al. Comprehensive analysis of regulatory elements of the promoters of rice sulfate transporter gene family and functional characterization of OsSul1;1promoter under different metal stress [J]. Plant Signal Behav, 2015, 10(4): e990843. |
| [20] | Hideki T. Sulfate transport systems in plants: functional diversity and molecular mechanisms underlying regulatory coordination [J]. J Exp Bot, 2019, 70(16): 4075-4087. |
| [21] | Chen Z, Zhao PX, Miao ZQ, et al. SULTR3s function in chloroplast sulfate uptake and affect ABA biosynthesis and the stress response [J]. Plant Physiol, 2019, 180(1): 593-604. |
| [22] | Xu XM, Xu ZY, Li ZY, et al. Increasing resistance to bacterial leaf streak in rice by editing the promoter of susceptibility gene OsSULRT3;6 [J]. Plant Biotechnol J, 2021, 19(6): 1101-1103. |
| [23] | 梁嘉燕, 胡晓丹, 孔雷蕾, 等. 水稻SULTR基因家族功能研究进展及其育种应用展望 [J]. 广东农业科学, 2025, 52(8): 13-24. |
| Liang JY, Hu XD, Kong LL, et al. Advances in functional research and breeding application prospects of the SULTR gene family in rice [J]. Guangdong Agricultural Sciences, 2025, 52(8): 13-24. | |
| [24] | Zhao W, Zhou Q, Tian ZZ, et al. Apply biochar to ameliorate soda saline-alkali land, improve soil function and increase corn nutrient availability in the Songnen Plain [J]. Sci Total Environ, 2020, 722: 137428. |
| [25] | 刘淼, 梁正伟. 低氮高密增微肥对苏打盐碱地水稻产量和氮肥利用率的影响 [J]. 土壤与作物, 2021, 10(3): 245-255. |
| Liu M, Liang ZW. Effect of nitrogen fertilizer reduction-high cultivation density-micro fertilizer increment on rice yield and nitrogen use efficiency in saline-sodic soil [J]. Soils Crops, 2021, 10(3): 245-255. | |
| [26] | 马国辉, 郑殿峰, 母德伟, 等. 耐盐碱水稻研究进展与展望 [J]. 杂交水稻, 2024, 39(1): 1-10. |
| Ma GH, Zheng DF, Mu DW, et al. Research progress and prospect of saline-alkali tolerant rice [J]. Hybrid Rice, 2024, 39(1): 1-10. | |
| [27] | Soudthedlath K, Nakamura T, Ushiwatari T, et al. SULTR2;1 adjusts the bolting timing by transporting sulfate from rosette leaves to the primary stem [J]. Plant Cell Physiol, 2024, 65(5): 770-780. |
| [28] | Wu G, Ding Y, Li N, et al. Genome-wide identification of the sulfate transporter gene family reveals that BolSULTR2; 1 regulates plant resistance to alternaria brassicicola through the modulation of glutathione biosynthesis in broccoli [J]. Antioxidants, 2025, 14(4): 496. |
| [29] | 黄伦增, 许云泓, 孟凡文, 等. 杨树PtTST3基因启动子克隆及表达活性分析 [J]. 分子植物育种, 2022, 20(17): 5649-5657. |
| Huang LZ, Xu YH, Meng FW, et al. Cloning and expression activity analysis of PtTST3 gene promoter in poplar [J]. Mol Plant Breed, 2022, 20(17): 5649-5657. | |
| [30] | 向警, 黄倩, 鞠春燕, 等. 外源褪黑素对盐胁迫下水稻种子萌发与幼苗生长的影响 [J]. 植物生理学报, 2021, 57(2): 393-401. |
| Xiang J, Huang Q, Ju CY, et al. Effect of exogenous melatonin on seed germination and seedling growth of rice under salt stress [J]. Plant Physiol Commun, 2021, 57(2): 393-401. | |
| [31] | Yang GY, Wang YC, Xia DA, et al. Overexpression of a GST gene (ThGSTZ1) from Tamarix hispida improves drought and salinity tolerance by enhancing the ability to scavenge reactive oxygen species [J]. Plant Cell Tiss Organ Cult, 2014, 117(1): 99-112. |
| [32] | Shuaiqi Y, Nana C, Jiaxuan Q, et al. OsUGE2 regulates plant growth through affecting ROS homeostasis and iron level in rice [J]. Rice, 2024, 17(1): 6. |
| [1] | 殷亚龙, 张明洋, 王洁敏, 苗雪雪, 陈劲, 王伟平. 水稻非生物胁迫协同耐受机制研究进展[J]. 生物技术通报, 2026, 42(4): 26-37. |
| [2] | 王玉昆, 原远, 王斌, 朱云娜, 任晓强, 任飞, 叶红. 转录组和脂质代谢组联合分析不同紫苏α-亚麻酸合成调控差异[J]. 生物技术通报, 2026, 42(4): 129-140. |
| [3] | 刘青媛, 吴洪启, 陈秀娥, 陈剑, 姜远泽, 何燕子, 喻奇伟, 刘仁祥. 转录因子NtMYB96a调控烟草耐旱性的功能研究[J]. 生物技术通报, 2026, 42(4): 239-250. |
| [4] | 杨婷, 杨宗桃, 艾静, 王禹童, 李燕烨, 邓军, 刘家勇, 赵勇, 张跃彬. 不同基因型甘蔗表型特征及根部转录组学分析[J]. 生物技术通报, 2026, 42(4): 190-201. |
| [5] | 杨跃琴, 邢英, 仲子荷, 田维军, 杨雪清, 王建旭. 甲基汞胁迫下水稻OsMATE34的表达及功能分析[J]. 生物技术通报, 2026, 42(1): 86-94. |
| [6] | 费思恬, 侯鹰翔, 李兰, 张超. 水稻赤霉素信号负调控因子SLR1的生物学功能及其调控网络[J]. 生物技术通报, 2026, 42(1): 13-30. |
| [7] | 樊荣辉, 罗远华, 陈艺荃, 方能炎, 陈燕, 钟淮钦, 叶秀仙. 文心兰‘金辉’和‘香水文心’花香形成比较分析[J]. 生物技术通报, 2026, 42(1): 105-113. |
| [8] | 王芳, 邵会茹, 吕林龙, 赵点, 胡振, 吕建珍, 姜亮. 植物和细菌TurboID邻近蛋白标记方法的建立[J]. 生物技术通报, 2025, 41(9): 44-53. |
| [9] | 刘建国, 刘格儿, 郭颖欣, 王斌, 王玉昆, 卢金凤, 黄文庭, 朱云娜. 转录组和代谢组联合解析‘桂柚1号’和‘沙田柚’果实品质差异[J]. 生物技术通报, 2025, 41(9): 168-181. |
| [10] | 刘泽洲, 段乃彬, 岳丽昕, 王清华, 姚行浩, 高莉敏, 孔素萍. 大蒜叶片蜡质成分分析及蜡质缺失基因Ggl-1筛选[J]. 生物技术通报, 2025, 41(9): 219-231. |
| [11] | 闫梦阳, 梁晓阳, 戴君昂, 张妍, 关团, 张辉, 刘良波, 孙志华. 阿莫西林降解菌的筛选及降解机制研究[J]. 生物技术通报, 2025, 41(9): 314-325. |
| [12] | 邓美壁, 严浪, 詹志田, 朱敏, 和玉兵. RUBY辅助的水稻高效CRISPR基因编辑[J]. 生物技术通报, 2025, 41(8): 65-73. |
| [13] | 柴军发, 洪波, 贾彦霞. 转录组和代谢组联合分析三株蜡蚧轮枝菌菌株毒力差异[J]. 生物技术通报, 2025, 41(8): 311-321. |
| [14] | 白雨果, 李婉迪, 梁建萍, 石志勇, 卢庚龙, 刘红军, 牛景萍. 哈茨木霉T9131对黄芪幼苗的促生机理[J]. 生物技术通报, 2025, 41(8): 175-185. |
| [15] | 李成花, 豆飞飞, 任毓昭, 刘彩霞, 刘凤楼, 王掌军, 李清峰. 外施水杨酸对白粉菌侵染小麦的影响及白粉菌转录组分析[J]. 生物技术通报, 2025, 41(7): 272-280. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||