








生物技术通报 ›› 2026, Vol. 42 ›› Issue (3): 203-212.doi: 10.13560/j.cnki.biotech.bull.1985.2025-1298
胡秋玲1( ), 陈灵1, 黄嘉怡1, 赵梓乔1, 潘璐怡2, 刘慧丽1(
), 陈灵1, 黄嘉怡1, 赵梓乔1, 潘璐怡2, 刘慧丽1( ), 刘太波1(
), 刘太波1( )
)
收稿日期:2025-11-28
出版日期:2026-03-26
发布日期:2026-04-23
通讯作者:
刘太波,男,博士,副教授,研究方向 :多胺调控植物发育与抗逆;E-mail: tbliu@scau.edu.cn作者简介:胡秋玲,女,研究方向 :多胺调控植物生长发育;E-mail: 13202365164@163.com基金资助:
HU Qiu-ling1( ), CHEN Ling1, HUANG Jia-yi1, ZHAO Zi-qiao1, PAN Lu-yi2, LIU Hui-li1(
), CHEN Ling1, HUANG Jia-yi1, ZHAO Zi-qiao1, PAN Lu-yi2, LIU Hui-li1( ), LIU Tai-bo1(
), LIU Tai-bo1( )
)
Received:2025-11-28
Published:2026-03-26
Online:2026-04-23
摘要:
多胺(polyamines, PAs)是一类普遍存在于植物体内的高生物活性的小分子脂肪族含氮碱,作为重要的内源生理活性物质,其在果实发育全过程中起着重要调控作用。研究表明,多胺不仅直接参与植物花芽分化、坐果、果实发育等过程,还能通过与乙烯等激素相互作用,共同调控果实的成熟与衰老。本文系统梳理了多胺在果实发育不同阶段中的调控作用,包括多胺浓度动态变化对花芽分化、花粉萌发、花粉管生长、坐果及果实发育早期阶段的影响。同时,总结了多胺对果实细胞分裂与膨大的调控,并探讨了多胺对果实成熟与衰老进程的调控作用。此外,本文还进一步探讨了多胺在农业生产中的应用潜力,包括多胺在提升果实产量、品质及延长货架期中的应用价值,并展望了利用AI辅助基因编辑技术对植物内源多胺合成、代谢及转运途径的精准调控以优化多胺代谢网络,实现对果实品质改良的应用前景,为新兴生物技术改良果实品质及育种等研究方向提出了新策略。总之,本文系统总结了多胺在果实发育各阶段的重要作用,深化了对多胺调控网络的认识。同时,通过探讨多胺在农业上的应用及其与新兴技术的融合,为果实品质改良与产业可持续发展提供重要理论支撑和新思路。
胡秋玲, 陈灵, 黄嘉怡, 赵梓乔, 潘璐怡, 刘慧丽, 刘太波. 多胺调控果实发育的研究进展[J]. 生物技术通报, 2026, 42(3): 203-212.
HU Qiu-ling, CHEN Ling, HUANG Jia-yi, ZHAO Zi-qiao, PAN Lu-yi, LIU Hui-li, LIU Tai-bo. Advances in the Regulation of Fruit Development by Polyamines[J]. Biotechnology Bulletin, 2026, 42(3): 203-212.
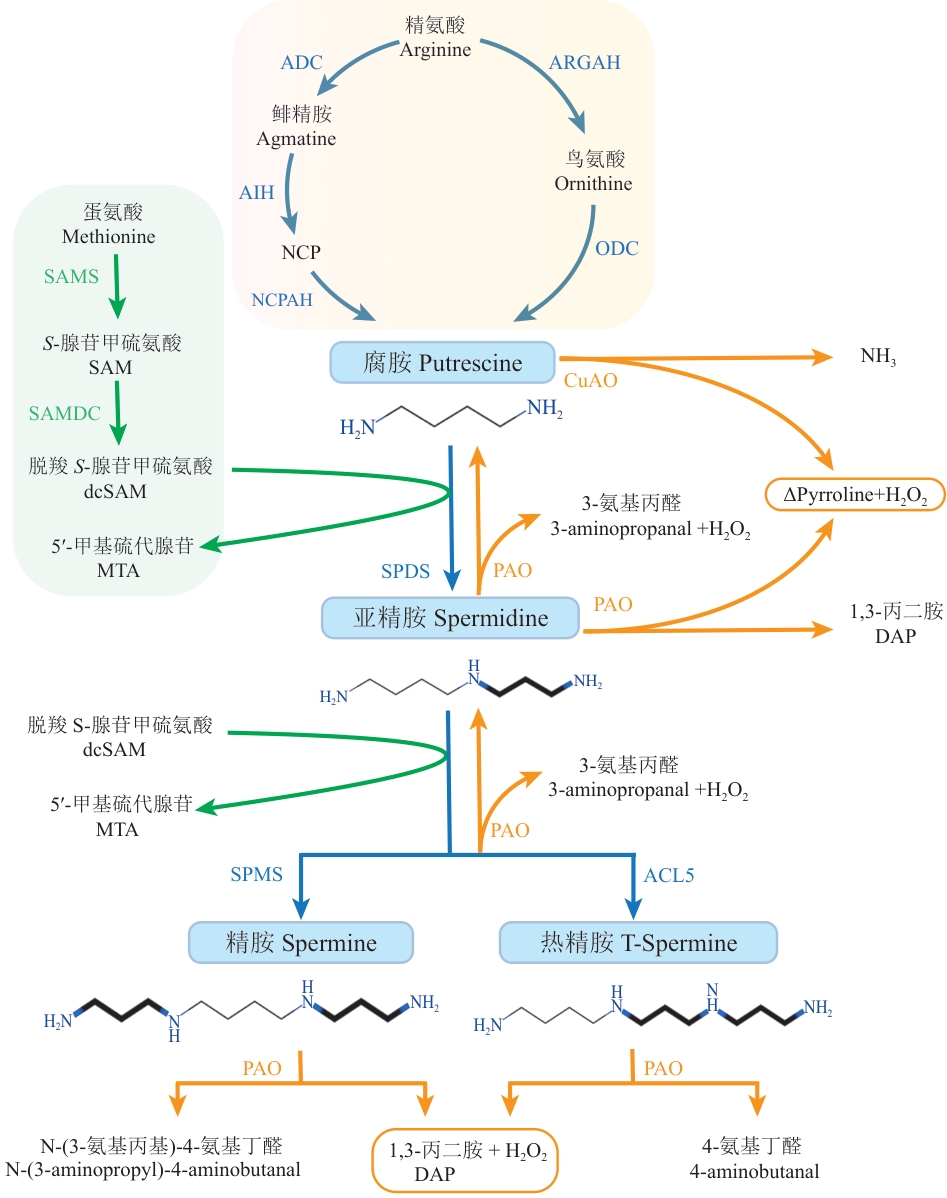
图1 多胺稳态调控网络腐胺(Put)是多胺合成的中心,Put的合成途径包括精氨酸(Arg)途径和鸟氨酸(Orn)途径。Put与dcSAM的氨丙基结合生成三胺Spd,并进一步生成四胺Spm和T-Spm。多胺的分解主要依赖于二胺氧化酶(DAO)和多胺氧化酶(PAO)。ADC:精氨酸脱羧酶;AIH:鲱精胺亚胺水解酶;NCP:N-氨甲酰腐胺;NCPAH:N-氨甲酰腐胺酰胺水解酶;ARGAH:精氨酸酶;ODC:鸟氨酸脱羧酶;SAMS:S-腺苷甲硫氨酸合成酶;SAMDC:S-腺苷甲硫氨酸脱羧酶;SPDS:亚精胺合成酶;SPMS:精胺合成酶;ACL5:热精胺合成酶;PAO:多胺氧化酶;CuAO:铜胺氧化酶;ΔPyrroline:吡咯啉
Fig. 1 Polyamine homeostasis regulatory networkPutrescine (Put) serves as the core of polyamine synthesis, with its synthetic pathways including the arginine (Arg) pathway and the ornithine (Orn) pathway. Put combines with the aminopropyl group of decarboxylated S-adenosylmethionine (dcSAM) to form the triamine spermidine (Spd), which is further converted into the tetraamines spermine (Spm) and thermospermine (T-Spm). The catabolism of polyamines mainly relies on diamine oxidase (DAO) and polyamine oxidase (PAO). ADC: Arginine decarboxylase. AIH: Agmatine ureohydrolase. NCP: N-carbamoylputrescine. NCPAH: N-carbamoylputrescine amidase. ARGAH: Arginase. ODC: Ornithine decarboxylase. SAMS: S-adenosylmethionine synthetase. SAMDC: S-adenosylmethionine decarboxylase. SPDS: Spermidine synthase. SPMS: Spermine synthase. ACL5: Thermospermine synthase. PAO: Polyamine oxidase. CuAO: Copper ammonia oxidase. ΔPyrroline: Delta-pyrroline
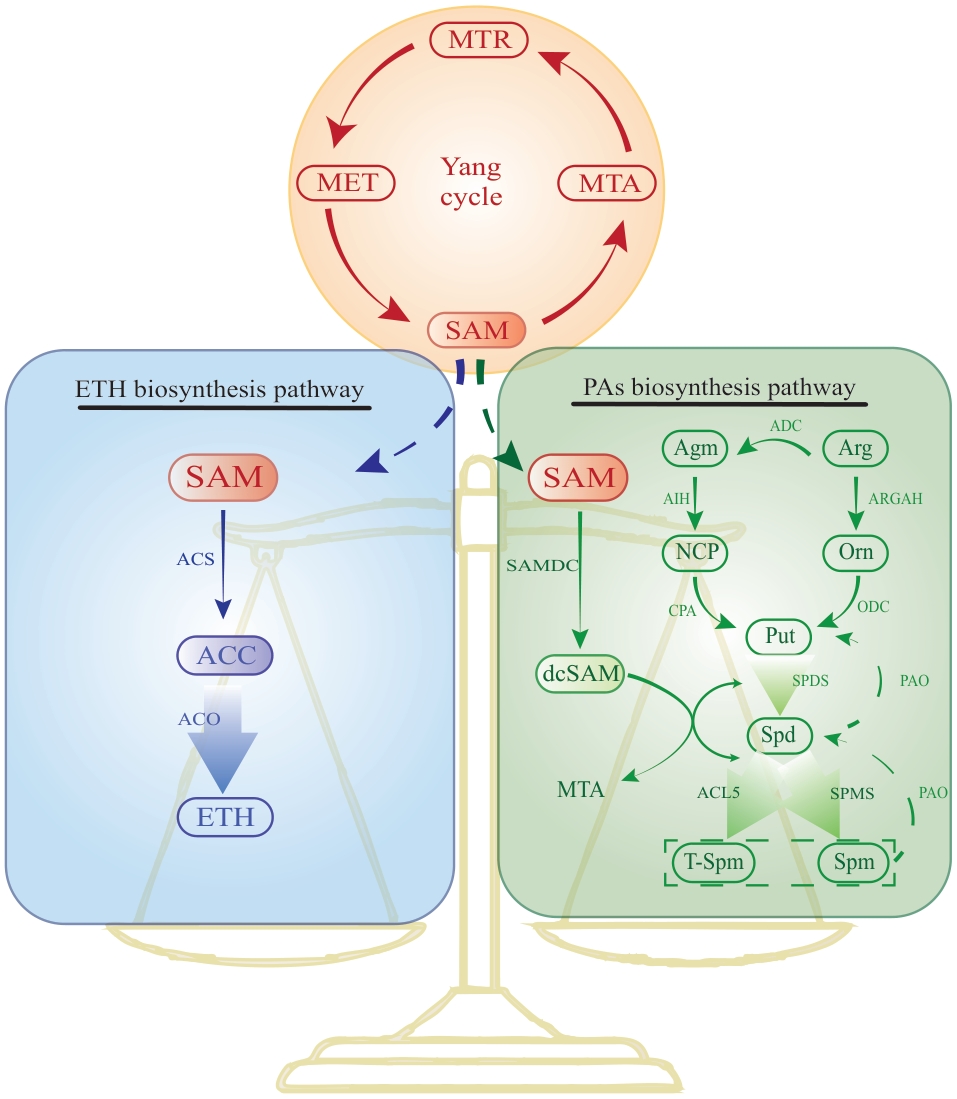
图2 果实中多胺和乙烯含量的稳态调控多胺合成途径和乙烯合成途径通过竞争共同的底物SAM来调控植物体内多胺和乙烯含量的动态变化,维持该动态平衡对果实的发育和成熟进程至关重要。MET:甲硫氨酸;SAM:S-腺苷甲硫氨酸;MTA:5′-甲硫基腺苷;MTR:5′-甲硫基核糖;ACS:ACC 合成酶;ACC:1-氨基环丙烷-1-羧酸;ACO:ACC氧化酶;ETH:乙烯;ADC:精氨酸脱羧酶;AIH:鲱精胺亚胺水解酶;NCP:N-氨甲酰腐胺;CPA:N-氨甲酰腐胺酰胺水解酶;ARGAH:精氨酸酶;ODC:鸟氨酸脱羧酶;Arg:精氨酸;Orn:鸟氨酸;Agm:鲱氨酸;SAMDC:S-腺苷甲硫氨酸脱羧酶;SPDS:亚精胺合成酶;SPMS:精胺合成酶;ACL5:热精胺合成酶;PAO:多胺氧化酶;SPDS:亚精胺合成酶;SPMS:精胺合成酶;Put:腐胺;Spd:亚精胺;Spm:精胺
Fig. 2 Homeostatic regulation of polyamine and ethylene contents in fruitsThe polyamine and ethylene synthesis pathways regulate the dynamic changes of polyamine and ethylene contents in plants by competing for the common substrate S-adenosylmethionine (SAM), and maintaining this dynamic balance is crucial for the development and ripening processes of fruits. MET: Methionine. SAM: S-adenosylmethionine. MTA: 5′-methylthioadenosine. MTR: 5′-methylthioribose. ACS: ACC synthase. ACC: 1-aminocyclopropane-1-carboxylic acid. ACO: ACC oxidase. ETH: Ethylene. ADC: Arginine decarboxylase. AIH: Agmatine iminohydrolase. NCP: N-carbamoylputrescine. ARGAH: Arginase. ODC: Ornithine decarboxylase. Arg: Arginine. Orn: Ornithine. Agm: Agmatine. SAMDC: S-adenosylmethionine decarboxylase. SPDS: Spermidine synthase. SPMS: Spermine synthase. ACL5: Thermospermine synthase. PAO: Polyamine oxidase. SPDS: Spermidine synthase. SPMS: Spermine synthase. Put: Putrescine. Spd: Spermidine. Spm: Spermine
| [1] | Blázquez MA. Polyamines: their role in plant development and stress [J]. Annu Rev Plant Biol, 2024, 75: 95-117. |
| [2] | Liu TB, Qu J, Fang YY, et al. Polyamines: The valuable bio-stimulants and endogenous signaling molecules for plant development and stress response [J]. J Integr Plant Biol, 2025, 67(3): 582-595. |
| [3] | Yu Z, Jia DY, Liu TB. Polyamine oxidases play various roles in plant development and abiotic stress tolerance [J]. Plants, 2019, 8(6): 184. |
| [4] | Chen T, Qin GZ, Tian SP. Regulatory network of fruit ripening: current understanding and future challenges [J]. New Phytol, 2020, 228(4): 1219-1226. |
| [5] | Benkő P, Jee S, Kaszler N, et al. Polyamines treatment during pollen germination and pollen tube elongation in tobacco modulate reactive oxygen species and nitric oxide homeostasis [J]. J Plant Physiol, 2020, 244: 153085. |
| [6] | Chen JY, Li WW, Li CX, et al. JsSAMDC promotes polyamine synthesis and flowering genes to synergistically regulate female flower bud differentiation [J]. Plant Cell Rep, 2025, 44(9): 204. |
| [7] | Guo JX, Wang SF, Yu XY, et al. Polyamines regulate strawberry fruit ripening by abscisic acid, auxin, and ethylene [J]. Plant Physiol, 2018, 177(1): 339-351. |
| [8] | Hu WL, Wang R, Hao XH, et al. OsLCD3 interacts with OsSAMS1 to regulate grain size via ethylene/polyamine homeostasis control [J]. Plant J, 2024, 119(2): 705-719. |
| [9] | Zhong M, Cui QS, Yang Y, et al. Tomato TGase positively regulates thermotolerance by inducing polyamine to activate autophagy [J]. J Integr Plant Biol, 2025, 67(9): 2350-2365. |
| [10] | Yang HS, Fang YY, Liang ZM, et al. Polyamines: pleiotropic molecules regulating plant development and enhancing crop yield and quality [J]. Plant Biotechnol J, 2024, 22(11): 3194-3201. |
| [11] | 刘颖, 王莹, 龙萃, 等. 植物多胺代谢途径研究进展 [J]. 生物工程学报, 2011, 27(2): 147-155. |
| Liu Y, Wang Y, Long C, et al. Metabolic pathway of polyamines in plants: a review [J]. Chin J Biotechnol, 2011, 27(2): 147-155. | |
| [12] | Mai HF, Qin T, Wei H, et al. Overexpression of OsACL5 triggers environmentally-dependent leaf rolling and reduces grain size in rice [J]. Plant Biotechnol J, 2024, 22(4): 833-847. |
| [13] | Serafini-Fracassini D, Del Duca S. Polyamines: small polycations essential as cell pleiotropic modulators throughout plant life [J]. Plant Biosyst Int J Deal Aspects Plant Biol, 2025, 159(6): 1649-1666. |
| [14] | Huang Y, Gao JH, Ji GM, et al. COP9 SIGNALOSOME SUBUNIT 5A facilitates POLYAMINE OXIDASE 5 degradation to regulate strawberry plant growth and fruit ripening [J]. Plant Cell, 2025, 37(2): koaf022. |
| [15] | Nambeesan S, Datsenka T, Ferruzzi MG, et al. Overexpression of yeast spermidine synthase impacts ripening, senescence and decay symptoms in tomato [J]. Plant J, 2010, 63(5): 836-847. |
| [16] | Li LJ, Gu WR, Li J, et al. Exogenously applied spermidine alleviates photosynthetic inhibition under drought stress in maize (Zea mays L.) seedlings associated with changes in endogenous polyamines and phytohormones [J]. Plant Physiol Biochem, 2018, 129: 35-55. |
| [17] | Majumdar R, Minocha R, Lebar MD, et al. Contribution of maize polyamine and amino acid metabolism toward resistance against Aspergillus flavus infection and aflatoxin production [J]. Front Plant Sci, 2019, 10: 692. |
| [18] | Wang X, Shi GX, Xu QS, et al. Exogenous polyamines enhance copper tolerance of Nymphoides peltatum [J]. J Plant Physiol, 2007, 164(8): 1062-1070. |
| [19] | 曲波, 张微, 陈旭辉, 等. 植物花芽分化研究进展 [J]. 中国农学通报, 2010, 26(24): 109-114. |
| Qu B, Zhang W, Chen XH, et al. Research progress of flower bud differentiation mechanism of plant [J]. Chin Agric Sci Bull, 2010, 26(24): 109-114. | |
| [20] | 赖瑞云, 林建忠, 谢志南, 等. 番木瓜花芽分化期内源多胺的变化 [J]. 闽西职业技术学院学报, 2008, 10(1): 110-112. |
| Lai RY, Lin JZ, Xie ZN, et al. Changes in endogenous polyamine during flower bud differentiation in papaya(Carica papaya) [J]. J Minxi Vocat Tech Coll, 2008, 10(1): 110-112. | |
| [21] | 李璇. 两个砂梨品种花芽分化期芽内内源多胺含量的动态变化 [J]. 中国南方果树, 2013, 42(2): 26-29. |
| Li X. Dynamic changes of endogenous polyamines in the buds of ‘shinsui’ and ‘Housui’ pear (Pyrus pyrifolia) during the differentiation of floral bud [J]. South China Fruits, 2013, 42(2): 26-29. | |
| [22] | 刘少华, 司守霞, 杨途熙, 等. 内源多胺含量对不同杏品种花芽分化及开花期的影响 [J]. 北方园艺, 2019(2): 55-59. |
| Liu SH, Si SX, Yang TX, et al. Effcet of contents of endogenous polymines in flower bud differentiation and anthesis of different apricot (Prunus armeniaca) varieties [J]. North Hortic, 2019(2): 55-59. | |
| [23] | 齐红岩, 黄鹤, 张多娇. 外源多胺对薄皮甜瓜花芽分化及花发育的影响 [J]. 中国蔬菜, 2012(10): 42-47. |
| Qi HY, Huang H, Zhang DJ. Effects of exogenous polyamine application on flower bud differentiation and flower development in muskmelon [J]. China Veg, 2012(10): 42-47. | |
| [24] | Cao XJ, Wen Z, Shang CQ, et al. Polyamine oxidase induces flower formation by promoting spermidine and ABA accumulation in cherry (Cerasus pseudocerasus Lindl.) [J]. Sci Hortic, 2024, 337: 113531. |
| [25] | 齐国辉, 徐继忠, 邵建柱, 等. 多胺、MGBG、水杨酸对鸭梨和雪花梨花粉萌发及花粉管生长的影响 [J]. 河北农业大学学报, 2008, 31(3): 21-27. |
| Qi GH, Xu JZ, Shao JZ, et al. Effects of polyamines, MGBG and salicylic acid on pollen germination and pollen tube growth of Yali pear and Xuehuali pear (Pyrus bretschneideri Rehd.) [J]. J Agric Univ Hebei, 2008, 31(3): 21-27. | |
| [26] | Sorkheh K, Shiran B, Rouhi V, et al. Response of in vitro pollen germination and pollen tube growth of almond (Prunus dulcis Mill.) to temperature, polyamines and polyamine synthesis inhibitor [J]. Biochem Syst Ecol, 2011, 39(4/5/6): 749-757. |
| [27] | Wolukau JN, Zhang SL, Xu GH, et al. The effect of temperature, polyamines and polyamine synthesis inhibitor on in vitro pollen germination and pollen tube growth of Prunus mume [J]. Sci Hortic, 2004, 99(3/4): 289-299. |
| [28] | Malik AU, Singh Z. Abscission of mango fruitlets as influenced by biosynthesis of polyamines [J]. J Hortic Sci Biotechnol, 2003, 78(5): 721-727. |
| [29] | 欧良喜, 袁沛元, 邱燕萍, 等. 荔枝花穗发育及坐果初期内源多胺含量的变化分析 [J]. 农业科技通讯, 2009(9): 76-78. |
| Ou LX, Yuan PY, Qiu YP, et al. Analysis on the changes of endogenous polyamines content in the development of litchi panicle and the initial stage of fruit setting [J]. Bull Agric Sci Technol, 2009(9): 76-78. | |
| [30] | Gunale AV, Nainwad RV, Sg P, et al. Standardization of various concentration of polyamines on growth and yield of mango (Mangifera indica L.) cv. Kesar [J]. Int J Adv Biochem Res, 2024, 8(12): 357-361. |
| [31] | Wu GY, Li QY, Tan Y, et al. Advances in understanding the mechanisms of organ abscission in vivo and in vitro plants [J]. Plant Growth Regul, 2024, 103(2): 293-306. |
| [32] | Kitashiba H, Hao YJ, Honda C, et al. Two types of spermine synthase gene: MdACL5 and MdSPMS are differentially involved in apple fruit development and cell growth [J]. Gene, 2005, 361: 101-111. |
| [33] | Pál M, Szalai G, Gondor OK, et al. Unfinished story of polyamines: Role of conjugation, transport and light-related regulation in the polyamine metabolism in plants [J]. Plant Sci, 2021, 308: 110923. |
| [34] | 李璇, 周京一, 王秀敏, 等. 内源多胺含量在砂梨果实发育过程中的变化研究 [J]. 上海农业学报, 2016, 32(2): 85-88. |
| Li X, Zhou JY, Wang XM, et al. Changes of endogenous polyamine metabolism in different fruit development of sand pear (Pyrus serotina Nakai) [J]. Acta Agric Shanghai, 2016, 32(2): 85-88. | |
| [35] | 李建国, 刘顺枝, 王泽槐. 荔枝果实发育过程中内源多胺含量的变化 [J]. 植物生理学通讯, 2004(2): 153-156. |
| Li JG, Liu SZ, Wang ZH. Changes in endogenous polyamine contents during fruit development of litchi (Litchi chinensis) [J]. Plant Physiol Commun, 2004(2): 153-156. | |
| [36] | Shiozaki S, Ogata T, Horiuchi S. Endogenous polyamines in the pericarp and seed of the grape berry during development and ripening [J]. Sci Hortic, 2000, 83(1): 33-41. |
| [37] | 刘顺枝, 李建国, 王泽槐. 荔枝果实发育过程中多胺合成酶活性和多胺含量的变化 [C]//中国植物生理学会第九次全国会议论文集. 贵阳, 2004: 68. |
| Liu SZ, Li JG, Wang ZH. Changes in polyamine synthase activity and polyamine content during litchi fruit development [C]//Proceedings of the Ninth National Conference of the Chinese Society for Plant Physiology. Guiyang, 2004: 68. | |
| [38] | Liu JH, Nada K, Pang XM, et al. Role of polyamines in peach fruit development and storage [J]. Tree Physiol, 2006, 26(6): 791-798. |
| [39] | Handa AK, Mattoo AK. Differential and functional interactions emphasize the multiple roles of polyamines in plants [J]. Plant Physiol Biochem, 2010, 48(7): 540-546. |
| [40] | Mattoo AK, Minocha SC, Minocha R, et al. Polyamines and cellular metabolism in plants: transgenic approaches reveal different responses to diamine putrescine versus higher polyamines spermidine and spermine [J]. Amino Acids, 2010, 38(2): 405-413. |
| [41] | Agudelo-Romero P, Ali K, Choi YH, et al. Perturbation of polyamine catabolism affects grape ripening of Vitis vinifera cv. Trincadeira [J]. Plant Physiol Biochem, 2014, 74: 141-155. |
| [42] | Lester GE. Polyamines and their cellular anti-senescence properties in honey dew muskmelon fruit [J]. Plant Sci, 2000, 160(1): 105-112. |
| [43] | Gao F, Mei XR, Li YZ, et al. Update on the roles of polyamines in fleshy fruit ripening, senescence, and quality [J]. Front Plant Sci, 2021, 12: 610313. |
| [44] | Liu MY, Wang CR, Ji HL, et al. Ethylene biosynthesis and signal transduction during ripening and softening in non-climacteric fruits: an overview [J]. Front Plant Sci, 2024, 15: 1368692. |
| [45] | Xu YJ, Tang SP, Jian CQ, et al. Polyamines and ethylene interact in mediating the effect of nitrogen rates on synthesis of amino acids in rice grains [J]. Food Energy Secur, 2022, 11(4): e408. |
| [46] | Lasanajak Y, Minocha R, Minocha SC, et al. Enhanced flux of substrates into polyamine biosynthesis but not ethylene in tomato fruit engineered with yeast S-adenosylmethionine decarboxylase gene [J]. Amino Acids, 2014, 46(3): 729-742. |
| [47] | Van de Poel B, Bulens I, Oppermann Y, et al. S-adenosyl-l-methionine usage during climacteric ripening of tomato in relation to ethylene and polyamine biosynthesis and transmethylation capacity [J]. Physiol Plant, 2013, 148(2): 176-188. |
| [48] | Gao F, Li JY, Li WJ, et al. Abscisic acid and polyamines coordinately regulate strawberry drought responses [J]. Plant Stress, 2024, 11: 100387. |
| [49] | Koushesh saba M, Arzani K, Barzegar M. Postharvest polyamine application alleviates chilling injury and affects apricot storage ability [J]. J Agric Food Chem, 2012, 60(36): 8947-8953. |
| [50] | Cui ZG, Jiang YB, Chen Y, et al. Overexpressing peach PpePAO1 gene improves the nutrient value of tomato and enhances resistance against Botrytis cinerea [J]. Postharvest Biol Technol, 2024, 211: 112792. |
| [51] | Tao YJ, Wang J, Miao J, et al. The spermine synthase OsSPMS1 regulates seed germination, grain size, and yield [J]. Plant Physiol, 2018, 178(4): 1522-1536. |
| [52] | Lv YS, Shao GN, Jiao GA, et al. Targeted mutagenesis of POLYAMINE OXIDASE 5 that negatively regulates mesocotyl elongation enables the generation of direct-seeding rice with improved grain yield [J]. Mol Plant, 2021, 14(2): 344-351. |
| [53] | Mehta RA, Cassol T, Li N, et al. Engineered polyamine accumulation in tomato enhances phytonutrient content, juice quality, and vine life [J]. Nat Biotechnol, 2002, 20(6): 613-618. |
| [54] | Mattoo AK, Chung SH, Goyal RK, et al. Overaccumulation of higher polyamines in ripening transgenic tomato fruit revives metabolic memory, upregulates anabolism-related genes, and positively impacts nutritional quality [J]. J AOAC Int, 2007, 90(5): 1456-1464. |
| [55] | Dibble ARG, Davies PJ, Mutschler MA. Polyamine content of long-keeping alcobaca tomato fruit [J]. Plant Physiol, 1988, 86(2): 338-340. |
| [56] | Çolak AM, Peral Eyduran S, Tas A, et al. The role of spermidine in postharvest fruit physiology: effects on quality characteristics and metabolite content of sweet cherry fruit during cold storage [J]. ACS Omega, 2025, 10(10): 10567-10578. |
| [57] | Orman E. Effect of postharvest spermidine treatments on quality and biochemical properties of nectarine fruits [J]. Turk J Agric For, 2023, 47(2): 186-195. |
| [58] | Li FP, Chen JX, Luo W, et al. DeepPGDB: a novel paradigm for AI-guided interactive plant genomic database [J]. Plant Commun, 2025, 6(12): 101494. |
| [59] | D'Incà R, Mattioli R, Tomasella M, et al. A Solanum lycopersicum polyamine oxidase contributes to the control of plant growth, xylem differentiation, and drought stress tolerance [J]. Plant J, 2024, 119(2): 960-981. |
| [60] | Zhang RX, Chai N, Liu TL, et al. The type V effectors for CRISPR/Cas-mediated genome engineering in plants [J]. Biotechnol Adv, 2024, 74: 108382. |
| [61] | Chuai GH, Ma HH, Yan JF, et al. DeepCRISPR: optimized CRISPR guide RNA design by deep learning [J]. Genome Biol, 2018, 19: 80. |
| [62] | Wessels HH, Méndez-Mancilla A, Guo XY, et al. Massively parallel Cas13 screens reveal principles for guide RNA design [J]. Nat Biotechnol, 2020, 38(6): 722-727. |
| [63] | 张子悦, 周欣智, 吕斌. 人工智能驱动的基因编辑技术及其应用 [J]. 中国生物化学与分子生物学报, 2025, 41(4): 522-532. |
| Zhang ZY, Zhou XZ, Lu B. Harnessing artificial intelligence for advances in gene editing [J]. Chin J Biochem Mol Biol, 2025, 41(4): 522-532. | |
| [64] | 林春姿, 黄其伟, 侯艳, 等. 水稻多胺氧化酶基因(OsPAO4)CRISPR/Cas9编辑突变体的创制 [J]. 广东农业科学, 2023, 50(3): 1-10. |
| Lin CZ, Huang QW, Hou Y, et al. Construction of rice mutants of the polyamine oxidase gene OsPAO4 based on CRISPR/Cas9 editing [J]. Guangdong Agric Sci, 2023, 50(3): 1-10. | |
| [65] | Lyu YS, Cao LM, Huang WQ, et al. Disruption of three polyamine uptake transporter genes in rice by CRISPR/Cas9 gene editing confers tolerance to herbicide paraquat [J]. aBIOTECH, 2022, 3(2): 140-145. |
| [66] | Hai Y, Shinsky SA, Porter NJ, et al. Histone deacetylase 10 structure and molecular function as a polyamine deacetylase [J]. Nat Commun, 2017, 8: 15368. |
| [67] | Zhai LR, Cheng Y, Wu MX, et al. Metabolic syndrome promotes endometrial cancer by Oleic acid-mediated polyamine accumulation [J]. Nat Commun, 2026, 17: 388. |
| [1] | 尹跃, 秦小雅, 米佳, 安巍, 何军, 张锋锋. 枸杞FBN基因家族鉴定及与类胡萝卜素代谢的相关性分析[J]. 生物技术通报, 2026, 42(3): 338-348. |
| [2] | 李天源, 亓新亮, 刘珊, 张建成, 王鹏飞, 张帅, 贾璐婷, 穆霄鹏. 欧李SPL基因家族的鉴定及在果实发育过程中的表达分析[J]. 生物技术通报, 2026, 42(3): 362-373. |
| [3] | 罗威, 宫奥, 仲阳, 胡迪, 周洪源, 张泓欣, 艾菊, 罗有卫, 高冬丽. 黄瓜SEPALLATA2基因敲除对果实及疣状结构发育的多效性影响[J]. 生物技术通报, 2026, 42(3): 283-293. |
| [4] | 刘淼, 林涛, 贾乐松, 胡丰, 李涛, 李志万, 刘美芳, 郑方燕, 崔龙. 从野生到栽培:番茄果实色泽的演化与调控机制[J]. 生物技术通报, 2026, 42(3): 187-202. |
| [5] | 刘娜, 曾宝珍, 贾兆星, 祝英方. 表观遗传调控番茄果实发育及成熟的研究进展[J]. 生物技术通报, 2026, 42(3): 37-47. |
| [6] | 马世杰, 李铮, 李蔚, 郭仰东, 张娜. 光信号调控园艺作物果实发育的研究进展[J]. 生物技术通报, 2026, 42(3): 5-18. |
| [7] | 张高翔, 吴玉碧, 郭亚静, 纪薇, 杨忠义. 葡萄WD40基因家族鉴定及表达量分析[J]. 生物技术通报, 2026, 42(3): 324-337. |
| [8] | 李成泉, 史庆华, 杨晓玉. 园艺作物果实发育的miRNA调控网络:从分子机制到种质创新[J]. 生物技术通报, 2026, 42(3): 19-36. |
| [9] | 王潇奕, 李金焱, 邢醒, 朱鸿亮. 基于乙烯响应筛选调控番茄成熟且影响呼吸的基因及其功能分析[J]. 生物技术通报, 2026, 42(3): 275-282. |
| [10] | 袁梦博, 赵光伟, 贺玉花, 黄祥, 徐永阳, 张健, 孔维虎, 田小琴, 户克云, 唐伶俐. CmCRC基因影响甜瓜性别分化的功能研究[J]. 生物技术通报, 2026, 42(3): 294-301. |
| [11] | 陈常璐, 杨志芳, 曹嵩晓, 李杨青, 叶靖烽, 吕海艳, 陈姗姗, 陈昊. 脱落酸与乙烯协同调控薄皮甜瓜果实软化的机制[J]. 生物技术通报, 2026, 42(3): 302-311. |
| [12] | 陈玲嫣, 李伟勋, 逄欣欣, 高翔, 焦绪瑶. 拟杆菌遗传操作工具箱:从传统方法到合成生物学前沿[J]. 生物技术通报, 2026, 42(2): 51-64. |
| [13] | 刘佳丽, 宋经荣, 赵文宇, 张馨元, 赵子洋, 曹一博, 张凌云. 蓝莓R2R3-MYB基因鉴定及类黄酮调控基因表达分析[J]. 生物技术通报, 2025, 41(9): 124-138. |
| [14] | 王赛笛, 张高阳, 吕欢欢, 孙忠科, 李成伟. 植物中直链淀粉合成及其含量提高策略的研究进展[J]. 生物技术通报, 2025, 41(8): 22-33. |
| [15] | 刁辰洋, 崔有志, 李炳志. 靶向诱变介导的微生物进化技术研究进展[J]. 生物技术通报, 2025, 41(8): 11-21. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||