








• 综述与专论 • 下一篇
王红阳( ), 邱艳红, 王德欣, 夏阳, 孟淑春, 徐秀兰(
), 邱艳红, 王德欣, 夏阳, 孟淑春, 徐秀兰( ), 张海军(
), 张海军( )
)
收稿日期:2025-09-27
出版日期:2026-03-09
通讯作者:
张海军,男,博士,副研究员,研究方向 :蔬菜种子萌发和活力调控机制;E-mail: zhanghaijun@nercv.org作者简介:王红阳,女,博士,助理研究员,研究方向 :蔬菜种子萌发和活力调控机制;E-mail: wanghongyang@nercv.org
基金资助:
WANG Hong-yang( ), QIU Yan-hong, WANG De-xin, XIA Yang, MENG Shu-chun, XU Xiu-lan(
), QIU Yan-hong, WANG De-xin, XIA Yang, MENG Shu-chun, XU Xiu-lan( ), ZHANG Hai-jun(
), ZHANG Hai-jun( )
)
Received:2025-09-27
Published:2026-03-09
摘要:
种子是农业的“芯片”,其萌发作为作物生命周期的起点,直接决定作物出苗质量及产量品质。一氧化氮(nitric oxide, NO)作为一种关键的气体信号分子,在调控种子休眠与萌发中的核心作用已成为植物生物学的研究前沿。本文系统综述了该领域的研究进展,阐释了植物中NO的主要合成与代谢途径及其稳态调控,重点剖析了其信号转导机制,尤其是其通过与激素、活性氧等信号网络的交叉对话以及蛋白质翻译后修饰,协同调控种子休眠与萌发关键节点的分子基础。尽管相关研究已取得显著进展,该领域仍存在若干重要科学问题亟待深入探索。未来在基础研究层面,需进一步解析NO合成、代谢与信号在种子不同组织和萌发阶段的时空动态调控网络;利用多组学技术系统鉴定其下游修饰靶点并阐明功能;揭示NO与光、温等环境信号整合的分子机制。在应用层面,可基于NO调控原理开发新型绿色种子处理技术,提升种子的逆境萌发能力与幼苗抗逆性;通过遗传或生物技术手段调控种子内源NO合成代谢通路,改良作物种子萌发特性,为培育出苗整齐、抗逆性强的新品种提供新策略。本综述通过系统梳理NO在植物种子休眠与萌发中的调控机制,旨在为深入理解种子生命起始的分子网络提供理论依据,并为作物种子品质提升与抗逆栽培技术创新提供新思路。
王红阳, 邱艳红, 王德欣, 夏阳, 孟淑春, 徐秀兰, 张海军. NO调控植物种子休眠和萌发的研究进展[J]. 生物技术通报, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1040.
WANG Hong-yang, QIU Yan-hong, WANG De-xin, XIA Yang, MENG Shu-chun, XU Xiu-lan, ZHANG Hai-jun. Research Progress in NO Regulating Seed Dormancy and Germination[J]. Biotechnology Bulletin, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1040.
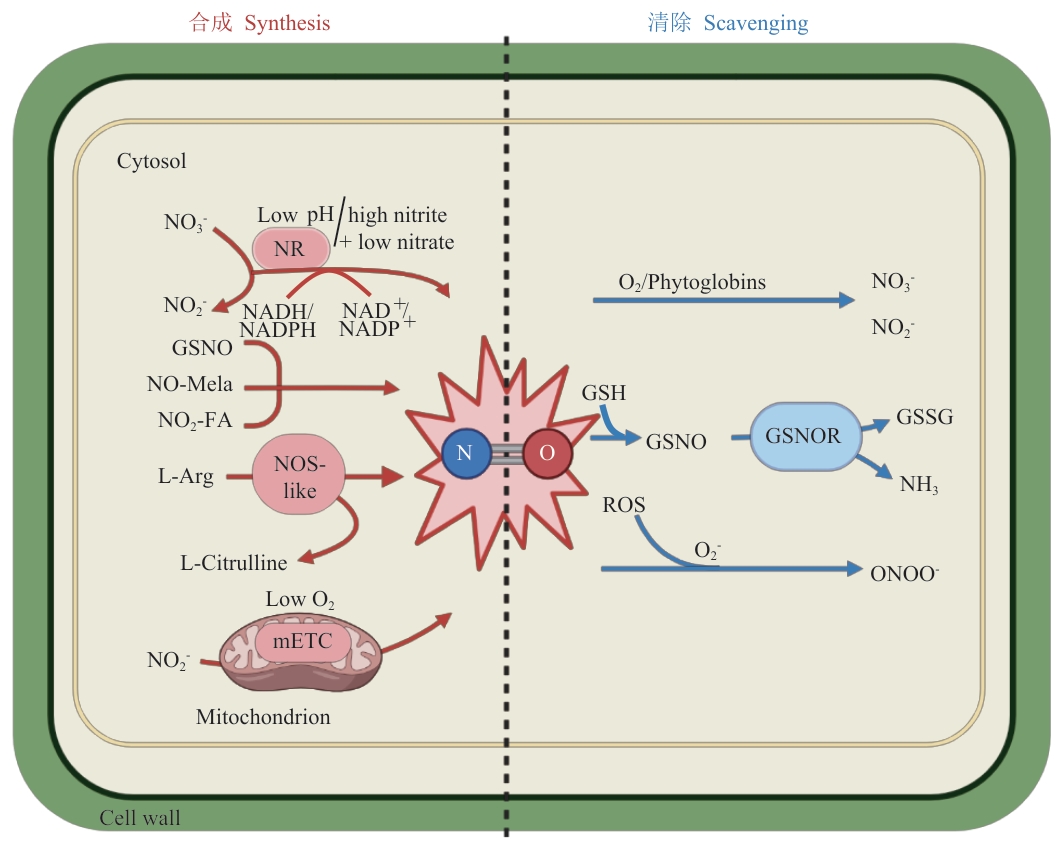
图1 NO的合成与清除虚线左侧为NO的合成过程,虚线右侧为NO的清除过程。NR:硝酸还原酶(例如拟南芥AtNIA1/2);mETC:线粒体电子传递链;L-Citrulline:L-瓜氨酸;GSNO:S-亚硝基谷胱甘肽;NO2-FAs:硝基脂肪酸;NO-Mela:亚硝基褪黑素;Phytoglobins:植物血红蛋白;GSSG:谷胱甘二硫化物;ONOO-:过氧亚硝酸根离子
Fig. 1 Biosynthesis and scavenging of NOThe left side of the dashed line shows the synthesis process of NO, while the right side illustrates the scavenging process of NO. NR: Nitrate reductase (e.g., AtNIA1/2); mETC: mitochondrial electron transport chain; GSNO: S-nitrosoglutathione; NO2-FAs: nitro fatty acids; NO-Mela: N-nitrosomelatonin; GSSG: glutathione disulfide; ONOO-: peroxynitrite
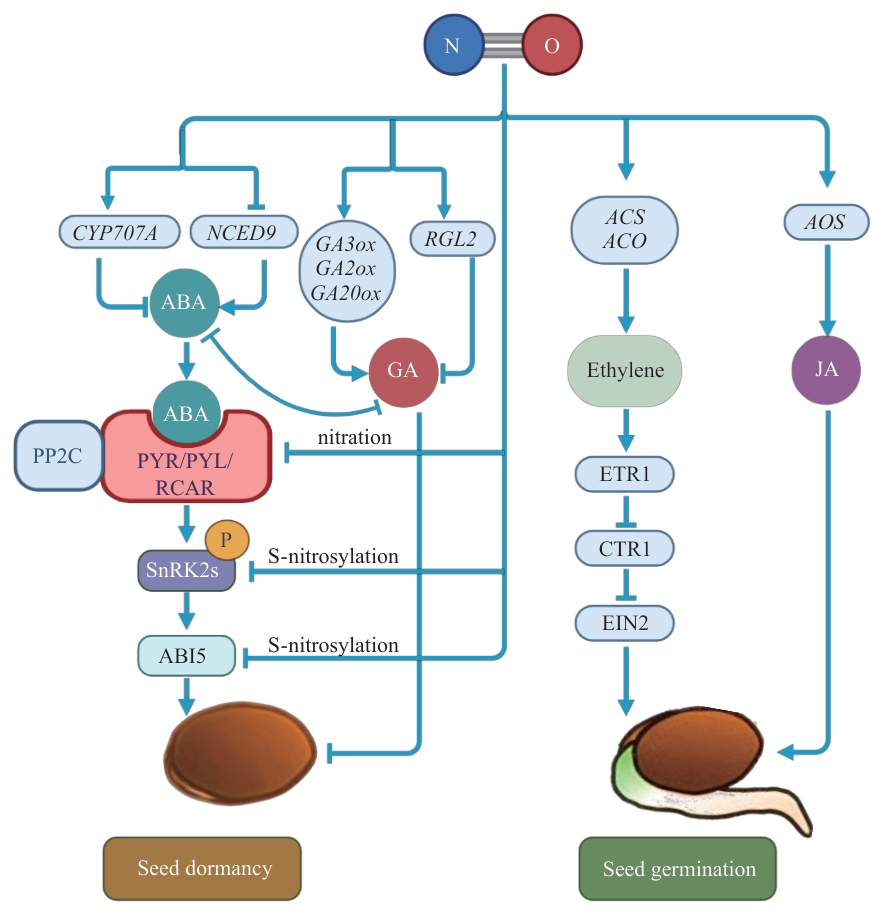
图2 NO与植物激素互作协同调控种子休眠及萌发NO通过抑制ABA合成及信号传递,促进GA、JA和乙烯的合成和信号传递,解除种子休眠并促进种子萌发。Nitration:硝化;S-nitrosylation:S-亚硝基化;CYP707A:细胞色素P450,707家族A亚家族;NCED:9-顺式-环氧类胡萝卜素双加氧酶;PYR/PYL/RCAR:ABA受体;PP2C:2C型蛋白磷酸酶;SnRKs:SNF1相关蛋白激酶;ABI5:脱落酸不敏感蛋白5;GAox:赤霉素氧化酶;RGL2:赤霉素类似物抑制因子2;ACS:S-腺苷-L-甲硫氨酸甲基硫腺苷裂解酶;ACO:ACC氧化酶;ETR:乙烯抗性蛋白1;CTR1:组成型三重反应蛋白1;EIN2:乙烯不敏感蛋白2;AOS:丙二烯氧化物合成酶。本图中的种子模型为示意图,所展示的调控机制在单子叶植物(如水稻)和双子叶植物(如拟南芥)中均存在
Fig. 2 Role of NO and phytohormones interaction in synergistically regulating seed dormancy and germinationNO promotes seed germination and breaks dormancy by antagonizing ABA biosynthesis and signaling, while concurrently activating the biosynthesis and signaling pathways of GA, JA, and ethylene. CYP707A: Cytochrome P450, Family 707, Subfamily A; NCED: nine-cis-epoxycarotenoid dioxygenase; PYR/PYL/RCAR: PYRABACTIN RESISTANCE1 (PYR1)/PYR1-LIKE (PYL)/REGULATORY COMPONENTS OF ABA RECEPTORS (RCAR) ; PP2C: type 2C protein phosphatases; SnRKs: SNF1-related protein kinase; ABI5: ABSCISIC ACID INSENSITIVE 5; GAox: GA oxidase; RGL2: REPRESSOR OF GA-LIKE2; ACS: S-adenosyl-L-methionine methylthioadenosine-lyase, ACO: ACC oxidase; ETR: ethylene resistant 1; CTR1: constitutive triple response 1; EIN2: ETHYLENE INSENSITIVE2; AOS: allene oxide synthase. The seed schematic represents a general model, and the illustrated NO-mediated mechanisms are operative in both monocot (e.g., rice) and dicot (e.g., Arabidopsis) species
PTM类型 Category | 修饰蛋白 Protein | 修饰位点 Modification sites | 功能描述 Function description | 参考文献 Reference |
|---|---|---|---|---|
S-亚硝基化 S-nitrosylation | APX, GR, DHAR | — | 激活抗坏血酸-谷胱甘肽循环中的抗氧化酶活性,提高顽拗型种子的脱水耐受性 | [ |
| ABI5 | Cys153 | S-亚硝基化后,E3连接酶介导其降解,从而促进种子萌发 | [ | |
| SnRK2.2/ SnRK2.3 | Cys137 | 抑制激酶活性,阻断ABA信号传递,促进种子萌发 | [ | |
| GSNOR1 | — | 高温胁迫诱导GSNOR1发生S-亚硝基化修饰并促使其降解,稳定ABI5蛋白导致种子热休眠 | [ | |
| GADPH | Cys154 | 诱导蛋白寡聚化,加速细胞死亡和种子老化 | [ | |
| HFR | Cys164 | 高温诱导HFR1 S-亚硝基化促使其降解,激活PIF1靶向的SOM基因表达,改变GA与ABA代谢平衡,抑制种子萌发 | [ | |
| MYB30 | Cys49 | NO介导MYB30 S-亚硝基化增强MYB30的转录活性,促进CYP707A2表达,降低ABA含量。打破种子休眠、促进萌发 | [ | |
酪氨酸硝化 Tyrosine nitration | 储藏蛋白 | — | 促进储藏蛋白降解,为胚轴伸长提供必需的氨基酸与维生素,为种子萌发提供能量 | [ |
| PYR/PYL/RCAR | — | 硝化使ABA受体失活,限制ABA信号传导 | [ |
表1 NO直接介导的蛋白翻译后修饰(PTMs)类型
Table 1 Types of protein post-translational modifications (PTMs) directly mediated by NO
PTM类型 Category | 修饰蛋白 Protein | 修饰位点 Modification sites | 功能描述 Function description | 参考文献 Reference |
|---|---|---|---|---|
S-亚硝基化 S-nitrosylation | APX, GR, DHAR | — | 激活抗坏血酸-谷胱甘肽循环中的抗氧化酶活性,提高顽拗型种子的脱水耐受性 | [ |
| ABI5 | Cys153 | S-亚硝基化后,E3连接酶介导其降解,从而促进种子萌发 | [ | |
| SnRK2.2/ SnRK2.3 | Cys137 | 抑制激酶活性,阻断ABA信号传递,促进种子萌发 | [ | |
| GSNOR1 | — | 高温胁迫诱导GSNOR1发生S-亚硝基化修饰并促使其降解,稳定ABI5蛋白导致种子热休眠 | [ | |
| GADPH | Cys154 | 诱导蛋白寡聚化,加速细胞死亡和种子老化 | [ | |
| HFR | Cys164 | 高温诱导HFR1 S-亚硝基化促使其降解,激活PIF1靶向的SOM基因表达,改变GA与ABA代谢平衡,抑制种子萌发 | [ | |
| MYB30 | Cys49 | NO介导MYB30 S-亚硝基化增强MYB30的转录活性,促进CYP707A2表达,降低ABA含量。打破种子休眠、促进萌发 | [ | |
酪氨酸硝化 Tyrosine nitration | 储藏蛋白 | — | 促进储藏蛋白降解,为胚轴伸长提供必需的氨基酸与维生素,为种子萌发提供能量 | [ |
| PYR/PYL/RCAR | — | 硝化使ABA受体失活,限制ABA信号传导 | [ |
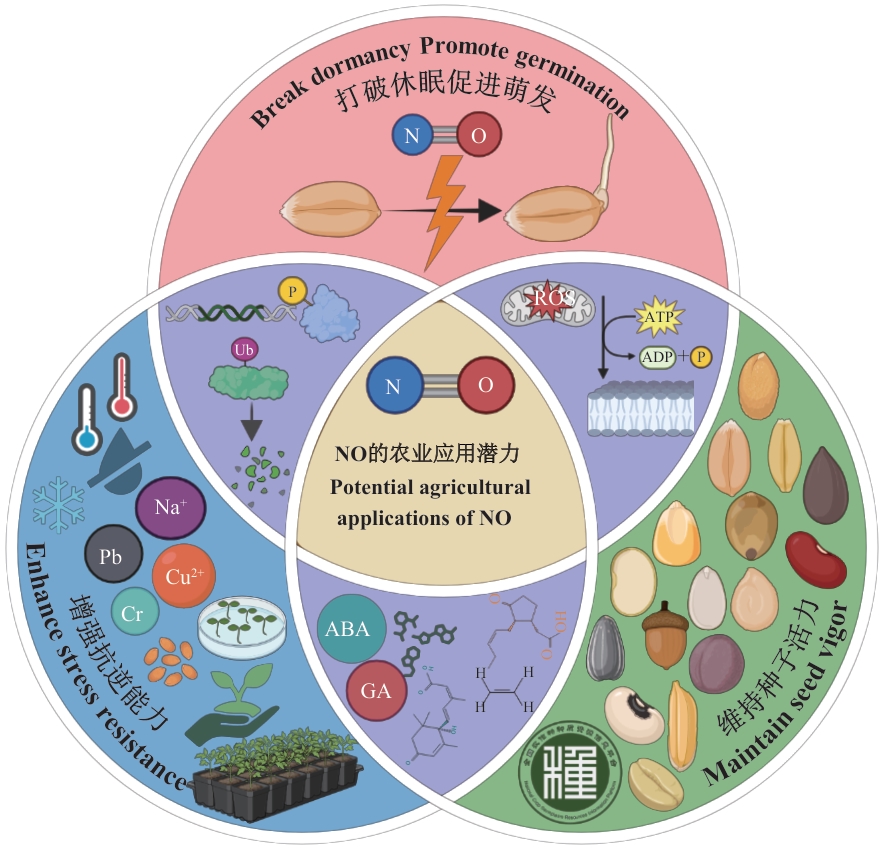
图3 NO的农业应用潜力在农业生产中,NO通过调控基因表达与蛋白质翻译后修饰,介导包含激素平衡重塑、氧化损伤缓解、细胞膜稳定及能量代谢调控在内的多重过程,最终实现打破种子休眠、促进萌发,显著提升种子的抗逆性,并维持其长期活力
Fig. 3 Agricultural application potential of NOIn agricultural practice, NO modulates gene expression and post-translational modifications, thereby reshaping hormonal balance, scavenging oxidative damage, maintaining membrane integrity, and reprogramming energy metabolism. These coordinated actions collectively break seed dormancy, promote germination, and ultimately enhance seed resistance to stress and maintain long-term vigor
| [1] | Sajeev N, Koornneef M, Bentsink L. A commitment for life: Decades of unraveling the molecular mechanisms behind seed dormancy and germination [J]. Plant Cell, 2024, 36(5): 1358-1376. |
| [2] | Hubert B, Leprince O, Buitink J. Sleeping but not defenceless: seed dormancy and protection [J]. J Exp Bot, 2024, 75(19): 6110-6124. |
| [3] | Matilla AJ. Current insights into weak seed dormancy and pre-harvest sprouting in crop species [J]. Plants, 2024, 13(18): 2559. |
| [4] | Koshland DE. The molecule of the year [J]. 1992, 258(5090): 1861. |
| [5] | Gupta KJ, Yadav N, Kumari A, et al. New insights into nitric oxide biosynthesis underpin lateral root development [J]. Mol Plant, 2024, 17(5): 691-693. |
| [6] | Ciacka K, Staszek P, Sobczynska K, et al. Nitric oxide in seed biology [J]. Int J Mol Sci, 2022, 23(23): 14951. |
| [7] | León J, Costa-Broseta Á. Present knowledge and controversies, deficiencies, and misconceptions on nitric oxide synthesis, sensing, and signaling in plants [J]. Plant Cell Environ, 2020, 43(1): 1-15. |
| [8] | Jack Q Wilkinson NMC. Identification of the Arabidopsis CHL3 gene as the nitrate reductase structural gene NIA2 [J]. Plant Cell, 1991, 3(5): 461-471. |
| [9] | Desikan R, Griffiths R, Hancock J, et al. A new role for an old enzyme: nitrate reductase-mediated nitric oxide generation is required for abscisic acid-induced stomatal closure in Arabidopsis thaliana [J]. Proc Natl Acad Sci USA, 2002, 99(25): 16314-16318. |
| [10] | Lozano-Juste J, León J. Enhanced abscisic acid-mediated responses in nia1nia2noa1-2 triple mutant impaired in NIA/NR- and AtNOA1-dependent nitric oxide biosynthesis in Arabidopsis [J]. Plant Physiol, 2010, 152(2): 891-903. |
| [11] | Gupta KJ, Kaladhar VC, Fitzpatrick TB, et al. Nitric oxide regulation of plant metabolism [J]. Mol Plant, 2022, 15(2): 228-242. |
| [12] | Astier J, Gross I, Durner J. Nitric oxide production in plants: an update [J]. J Exp Bot, 2018, 69(14): 3401-3411. |
| [13] | Krasuska U, Ciacka K, Orzechowski S, et al. Modification of the endogenous NO level influences apple embryos dormancy by alterations of nitrated and biotinylated protein patterns [J]. Planta, 2016, 244(4): 877-891. |
| [14] | Modolo LV, Augusto O, Almeida IMG, et al. Nitrite as the major source of nitric oxide production by Arabidopsis thaliana in response to Pseudomonas syringae [J]. FEBS Lett, 2005, 579(17): 3814-3820. |
| [15] | Bethke PC, Badger MR, Jones RL. Apoplastic synthesis of nitric oxide by plant tissues [J]. Plant Cell, 2004, 16(2): 332-341. |
| [16] | Corpas FJ, González-Gordo S, Palma JM. NO source in higher plants: present and future of an unresolved question [J]. Trends Plant Sci, 2022, 27(2): 116-119. |
| [17] | Karpinska B, Foyer CH. Superoxide signalling and antioxidant processing in the plant nucleus [J]. J Exp Bot, 2024, 75(15): 4599-4610. |
| [18] | Ciacka K, Krasuska U, Otulak-Kozieł K, et al. Dormancy removal by cold stratification increases glutathione and S-nitrosoglutathione content in apple seeds [J]. Plant Physiol Biochem, 2019, 138: 112-120. |
| [19] | Berger A, Boscari A, Puppo A, et al. Nitrate reductases and hemoglobins control nitrogen-fixing symbiosis by regulating nitric oxide accumulation [J]. J Exp Bot, 2021, 72(3): 873-884. |
| [20] | Liu YY, Liu ZY, Wu XT, et al. Role of protein S-nitrosylation in plant growth and development [J]. Plant Cell Rep, 2024, 43(8): 204. |
| [21] | Fuentes-Terrón A, Latter R, Madden S, et al. Destined for destruction: The role of methionine aminopeptidases and plant cysteine oxidases in N-degron formation [J]. Plant Physiol, 2024, 197(1): kiae667. |
| [22] | Gibbs DJ, Md Isa N, Movahedi M, et al. Nitric oxide sensing in plants is mediated by proteolytic control of group VII ERF transcription factors [J]. Mol Cell, 2014, 53(3): 369-379. |
| [23] | Sedlářová M, Jedelská T, Lebeda A, et al. Progress in plant nitric oxide studies: implications for phytopathology and plant protection [J]. Int J Mol Sci, 2025, 26(5): 2087. |
| [24] | Naaz S, Pande A, Laxmi A. Nitric oxide-mediated thermomemory: a new perspective on plant heat stress resilience [J]. Front Plant Sci, 2025, 16: 1525336. |
| [25] | Kumar D, Ohri P. Say “NO” to plant stresses: Unravelling the role of nitric oxide under abiotic and biotic stress [J]. Nitric Oxide, 2023, 130: 36-57. |
| [26] | Wong A, Tian XC, Yang YX, et al. Identification of potential nitric oxide-sensing proteins using the H-NOX motif [J]. Mol Plant, 2021, 14(2): 195-197. |
| [27] | Zhao X, Cao HN, Liu Y, et al. A new plant guanosine cyclase ZjGC found from jujube regulates growth and development via endogenous hormones [J]. Front Plant Sci, 2025, 16: 1633496. |
| [28] | Fejes G, Bodor T, Szőllősi R, et al. Nitric oxide as an integral element in priming-induced tolerance and plant stress memory [J]. J Exp Bot, 2025, 76(13): 3669-3685. |
| [29] | Zhang Y, Wang RR, Wang XD, et al. Nitric oxide regulates seed germination by integrating multiple signalling pathways [J]. Int J Mol Sci, 2023, 24(10): 9052. |
| [30] | Sekita MC, dos Santos Dias DCF, Pinheiro DT, et al. Nitric oxide in physiological potential and biochemical mechanisms of pea seeds under water deficit [J]. J Seed Sci, 2022, 44: e202244016. |
| [31] | 杨小环, 杨婧怡, 王子然, 等. 六价铬对红芸豆种子萌发和幼苗生长的毒害作用及外源NO的缓解效应 [J]. 中国生态农业学报: 中英文, 2024, 32(8): 1366-1376. |
| Yang XH, Yang JY, Wang ZR, et al. Toxic effect of hexavalent chromium on seed germination and seedling growth of red kidney bean and the alleviation effect of exogenous NO [J]. Chin J Eco Agric, 2024, 32(8): 1366-1376. | |
| [32] | 王志科, 王金成. 外源NO对NaCl胁迫下玉米种子萌发和幼苗生理特性的影响 [J]. 山东农业科学, 2025, 57(2): 78-83. |
| Wang ZK, Wang JC. Effects of exogenous nitric oxide on seed germination and seedling physiological characteristics of maize under NaCl stress [J]. Shandong Agric Sci, 2025, 57(2): 78-83. | |
| [33] | 吴建飞, 黄茵, 温天旺, 等. 硝普钠浸种对铜胁迫下棉花种子萌发和幼苗抗氧化系统及铜离子吸收分配的影响 [J]. 农业环境科学学报, 2025, 44(1): 31-40. |
| Wu JF, Huang Y, Wen TW, et al. Effects of seed presoaking with sodium nitroprussiate on cotton seed germination, antioxidant system, and copper ion uptake and distribution under copper stress [J]. J Agro Environ Sci, 2025, 44(1): 31-40. | |
| [34] | Liu FF, Qiao XH, Yang T, et al. Nitric oxide promoted the seed germination of Cynanchum auriculatum under cadmium stress [J]. Agronomy, 2024, 14(1): 86. |
| [35] | 尹美强, 王栋, 王金荣, 等. 外源一氧化氮对盐胁迫下高粱种子萌发及淀粉转化的影响 [J]. 中国农业科学, 2019, 52(22): 4119-4128. |
| Yin MQ, Wang D, Wang JR, et al. Effects of exogenous nitric oxide on seed germination and starch transformation of sorghum seeds under salt stress [J]. Sci Agric Sin, 2019, 52(22): 4119-4128. | |
| [36] | Shu K, Liu XD, Xie Q, et al. Two faces of one seed: hormonal regulation of dormancy and germination [J]. Mol Plant, 2016, 9(1): 34-45. |
| [37] | Sano N, Marion-Poll A. ABA metabolism and homeostasis in seed dormancy and germination [J]. Int J Mol Sci, 2021, 22(10): 5069. |
| [38] | Xu JR, Lu XF, Liu YZ, et al. Interaction between ABA and NO in plants under abiotic stresses and its regulatory mechanisms [J]. Front Plant Sci, 2024, 15: 1330948. |
| [39] | Liu YG, Shi L, Ye NH, et al. Nitric oxide-induced rapid decrease of abscisic acid concentration is required in breaking seed dormancy in Arabidopsis [J]. New Phytol, 2009, 183(4): 1030-1042. |
| [40] | Andryka-Dudek P, Ciacka K, Wiśniewska A, et al. Nitric oxide-induced dormancy removal of apple embryos is linked to alterations in expression of genes encoding ABA and JA biosynthetic or transduction pathways and RNA nitration [J]. Int J Mol Sci, 2019, 20(5): 1007. |
| [41] | Wang PC, Du YY, Hou YJ, et al. Nitric oxide negatively regulates abscisic acid signaling in guard cells by S-nitrosylation of OST1 [J]. Proc Natl Acad Sci USA, 2015, 112(2): 613-618. |
| [42] | Iqbal N, Umar S, Khan NA, et al. Crosstalk between abscisic acid and nitric oxide under heat stress: exploring new vantage points [J]. Plant Cell Rep, 2021, 40(8): 1429-1450. |
| [43] | Kępczyński J, Wójcik A, Dziurka M. NO-mediated dormancy release of Avena fatua caryopses is associated with decrease in abscisic acid sensitivity, content and ABA/GAs ratios [J]. Planta, 2023, 257(6): 101. |
| [44] | Bethke PC, Libourel IGL, Aoyama N, et al. The Arabidopsis aleurone layer responds to nitric oxide, gibberellin, and abscisic acid and is sufficient and necessary for seed dormancy [J]. Plant Physiol, 2007, 143(3): 1173-1188. |
| [45] | Nagel M, Alqudah AM, Bailly M, et al. Novel loci and a role for nitric oxide for seed dormancy and preharvest sprouting in barley [J]. Plant Cell Environ, 2019, 42(4): 1318-1327. |
| [46] | Carrillo-Barral N, Matilla AJ, Iglesias-Fernández R, et al. Nitrate-induced early transcriptional changes during imbibition in non-after-ripened Sisymbrium officinale seeds [J]. Physiol Plant, 2013, 148(4): 560-573. |
| [47] | Zhao CH, Zhang Y, Yang L. Integrating physiology, transcriptome, and metabolomics reveals the potential mechanism of nitric oxide concentration-dependent regulation of embryo germination in Sorbus pohuashanensis [J]. Plants, 2025, 14(3): 344. |
| [48] | Gniazdowska A, Krasuska U, Bogatek R. Dormancy removal in apple embryos by nitric oxide or cyanide involves modifications in ethylene biosynthetic pathway [J]. Planta, 2010, 232(6): 1397-1407. |
| [49] | Gniazdowska A, Dobrzyńska U, Babańczyk T, et al. Breaking the apple embryo dormancy by nitric oxide involves the stimulation of ethylene production [J]. Planta, 2007, 225(4): 1051-1057. |
| [50] | Sami A, Rehman S, Tanvir MA, et al. Assessment of the germination potential of Brassica oleracea seeds treated with karrikin 1 and cyanide, which modify the ethylene biosynthetic pathway [J]. J Plant Growth Regul, 2021, 40(3): 1257-1269. |
| [51] | Jacobsen JV, Barrero JM, Hughes T, et al. Roles for blue light, jasmonate and nitric oxide in the regulation of dormancy and germination in wheat grain (Triticum aestivum L.) [J]. Planta, 2013, 238(1): 121-138. |
| [52] | Bailly C. The signalling role of ROS in the regulation of seed germination and dormancy [J]. Biochem J, 2019, 476(20): 3019-3032. |
| [53] | Ye TT, Ma TX, Chen Y, et al. The role of redox-active small molecules and oxidative protein post-translational modifications in seed aging [J]. Plant Physiol Biochem, 2024, 213: 108810. |
| [54] | EL-MAAROUF-BOUTEAU H, Sajjad Y, Bazin J, et al. Reactive oxygen species, abscisic acid and ethylene interact to regulate sunflower seed germination [J]. Plant Cell Environ, 2015, 38(2): 364-374. |
| [55] | Yiğit İ, Atici Ö. Seed priming with nitric oxide mitigates exogenous methylglyoxal toxicity by restoring glyoxalase and antioxidant systems in germinating maize (Zea mays L.) seeds [J]. Cereal Res Commun, 2022, 50(4): 811-820. |
| [56] | Liu YG, Ye NH, Liu R, et al. H2O2 mediates the regulation of ABA catabolism and GA biosynthesis in Arabidopsis seed dormancy and germination [J]. J Exp Bot, 2010, 61(11): 2979-2990. |
| [57] | Ciacka K, Tyminski M, Gniazdowska A, et al. Nitric oxide as a remedy against oxidative damages in apple seeds undergoing accelerated ageing [J]. Antioxidants, 2021, 11(1): 70. |
| [58] | Lin W, Shang JX, Li XY, et al. Nitric oxide regulates multiple signal pathways in plants via protein S-nitrosylation [J]. Curr Issues Mol Biol, 2025, 47(6): 407. |
| [59] | Bai XG, Yang LM, Tian MH, et al. Nitric oxide enhances desiccation tolerance of recalcitrant Antiaris toxicaria seeds via protein S-nitrosylation and carbonylation [J]. PLoS One, 2011, 6(6): e20714. |
| [60] | Albertos P, Romero-Puertas MC, Tatematsu K, et al. S-nitrosylation triggers ABI5 degradation to promote seed germination and seedling growth [J]. Nat Commun, 2015, 6: 8669. |
| [61] | Wang PC, Zhu JK, Lang ZB. Nitric oxide suppresses the inhibitory effect of abscisic acid on seed germination by S-nitrosylation of SnRK2 proteins [J]. Plant Signal Behav, 2015, 10(6): e1031939. |
| [62] | Zhao HY, Ma L, Shen JL, et al. S-nitrosylation of the transcription factor MYB30 facilitates nitric oxide-promoted seed germination in Arabidopsis [J]. Plant Cell, 2024, 36(2): 367-382. |
| [63] | Zeng MY, He YQ, Gao X, et al. Characteristics and functions of glyceraldehyde 3-phosphate dehydrogenase S-nitrosylation during controlled aging of elm and Arabidopsis seeds [J]. J Exp Bot, 2021, 72(20): 7020-7034. |
| [64] | Ying SB, Yang WJ, Li P, et al. Phytochrome B enhances seed germination tolerance to high temperature by reducing S-nitrosylation of HFR1 [J]. EMBO Rep, 2022, 23(10): e54371. |
| [65] | Wei WJ, Hu YL, Yang WJ, et al. S-nitrosoglutathion reductase activity modulates the thermotolerance of seeds germination by controlling ABI5 stability under high temperature [J]. Phyton Int J Exp Bot, 2021, 90(4): 1075-1087. |
| [66] | León J. Protein tyrosine nitration in plant nitric oxide signaling [J]. Front Plant Sci, 2022, 13: 859374. |
| [67] | Castillo MC, Lozano-Juste J, González-Guzmán M, et al. Inactivation of PYR/PYL/RCAR ABA receptors by tyrosine nitration may enable rapid inhibition of ABA signaling by nitric oxide in plants [J]. Sci Signal, 2015, 8(392): ra89. |
| [68] | Vollár M, Feigl G, Oláh D, et al. Nitro-oleic acid in seeds and differently developed seedlings of Brassica napus L [J]. Plants, 2020, 9(3): 406. |
| [69] | Sun CL, Zhang YX, Liu LJ, et al. Molecular functions of nitric oxide and its potential applications in horticultural crops [J]. Hortic Res, 2021, 8(1): 71. |
| [70] | Wang RR, Wang MF, Yang L. Antagonistic effects of nitric oxide and far-red light on Sorbus pohuashanensis embryo germination [J]. J Plant Growth Regul, 2025. . |
| [71] | Liu SJ, Song SH, Wang WQ, et al. De novo assembly and characterization of germinating lettuce seed transcriptome using Illumina paired-end sequencing [J]. Plant Physiol Biochem, 2015, 96: 154-162. |
| [72] | Pande A, Mun BG, Methela NJ, et al. Heavy metal toxicity in plants and the potential NO-releasing novel techniques as the impending mitigation alternatives [J]. Front Plant Sci, 2022, 13: 1019647. |
| [73] | Ciacka K, Krasuska U, Staszek P, et al. Effect of nitrogen reactive compounds on aging in seed [J]. Front Plant Sci, 2020, 11: 101 |
| [1] | 张学琼, 潘素君, 李魏, 戴良英. 植物磷酸盐转运蛋白在胁迫响应中的研究进展[J]. 生物技术通报, 2025, 41(7): 28-36. |
| [2] | 王从欢, 伍国强, 魏明. 植物CBL调控逆境胁迫响应的作用机制[J]. 生物技术通报, 2025, 41(7): 1-16. |
| [3] | 刘晓天, 何毓琦, 刘畅, 薛华. 家榆UpNOA1的克隆及其互作蛋白的筛选[J]. 生物技术通报, 2025, 41(6): 297-306. |
| [4] | 刘源, 赵冉, 卢振芳, 李瑞丽. 植物类胡萝卜素生物代谢途径及其功能研究进展[J]. 生物技术通报, 2025, 41(5): 23-31. |
| [5] | 段若昕, 陈盈盈, 林金星, 李瑞丽. 种子休眠及其调控机制研究进展[J]. 生物技术通报, 2025, 41(12): 16-26. |
| [6] | 韩昱, 袁青云, 章青平, 吴春来, 贺巍, 张芬. 茶树CsNPF家族6个基因的克隆与表达分析及CsNPF7.3功能验证[J]. 生物技术通报, 2025, 41(10): 264-276. |
| [7] | 武志健, 刘广洋, 林志豪, 盛彬, 陈鸽, 许晓敏, 王军伟, 徐东辉. 蔬菜种子萌发的纳米调控及其机制研究进展[J]. 生物技术通报, 2025, 41(1): 14-24. |
| [8] | 马博涛, 伍国强, 魏明. bZIP转录因子在植物逆境胁迫响应和生长发育中的作用[J]. 生物技术通报, 2024, 40(9): 148-160. |
| [9] | 刘文浩, 吴刘记, 徐芳. 小肽调控植物分生组织发育的机制及其在作物改良中的研究进展[J]. 生物技术通报, 2024, 40(7): 1-18. |
| [10] | 王迪, 张晓宇, 宋宇鑫, 郑东然, 田静, 李玉花, 王宇, 吴昊. 细胞全能性转录因子调控植物组培再生的分子机制研究进展[J]. 生物技术通报, 2024, 40(6): 23-33. |
| [11] | 张娜, 刘梦楠, 屈展帆, 崔祎平, 倪嘉瑶, 王华忠. 小麦烯醇化酶基因ENO2的可变翻译分析和原核表达[J]. 生物技术通报, 2024, 40(5): 112-119. |
| [12] | 花子晴, 周静远, 董合忠. 双子叶植物下胚轴和顶端弯钩发育及其对出苗的调控机制[J]. 生物技术通报, 2024, 40(4): 23-32. |
| [13] | 陈应娥, 梁巧兰. 植物脱落酸及其受体基因PYL9的作用研究进展[J]. 生物技术通报, 2024, 40(12): 1-11. |
| [14] | 乔承彬, 宋佳伟, 杨辉, 段凯蓉, 冉杰, 孔维儒, 冯培媛, 罗成科, 李培富, 田蕾. 水稻叶宽调控机制及相关基因研究进展[J]. 生物技术通报, 2024, 40(11): 88-102. |
| [15] | 林鑫焱, 张传忠, 戴兵, 王馨珩, 刘剑锋, 温丽, 徐兴健, 方军. 水稻穗发芽遗传与分子机制的研究进展[J]. 生物技术通报, 2024, 40(1): 24-31. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||