








Biotechnology Bulletin ›› 2026, Vol. 42 ›› Issue (4): 161-169.doi: 10.13560/j.cnki.biotech.bull.1985.2025-0692
Previous Articles Next Articles
XU Yu-jiao1( ), SUN Yu-shuai1, LIU Dao-qi1, ZHANG Li2, ZHANG Zhi-chang2, YAO Yu-xin1(
), SUN Yu-shuai1, LIU Dao-qi1, ZHANG Li2, ZHANG Zhi-chang2, YAO Yu-xin1( )
)
Received:2025-06-30
Online:2026-04-26
Published:2026-04-30
Contact:
YAO Yu-xin
E-mail:15966391881@163.com;yaoyx@sdau.edu.cn
XU Yu-jiao, SUN Yu-shuai, LIU Dao-qi, ZHANG Li, ZHANG Zhi-chang, YAO Yu-xin. Functional Analysis of VvHSP18.2 Overexpression in Regulating Salt-alkali Resistance in Grapevines[J]. Biotechnology Bulletin, 2026, 42(4): 161-169.
引物 Primer | 正向引物 Forward primer (5′‒3′) | 反向引物 Reverse primer (5′‒3′) |
|---|---|---|
| VvHSP18.2 | GCAGAGGTCAAGAAACCCGA | CCTCAAAGCATGCTCTTCCT |
| β-Actin | GAGATTCCGTTGTCCAGAAGTC | CAATGTTGCCATAGAGGTCCTT |
Table 1 Primer sequences for RT-qPCR
引物 Primer | 正向引物 Forward primer (5′‒3′) | 反向引物 Reverse primer (5′‒3′) |
|---|---|---|
| VvHSP18.2 | GCAGAGGTCAAGAAACCCGA | CCTCAAAGCATGCTCTTCCT |
| β-Actin | GAGATTCCGTTGTCCAGAAGTC | CAATGTTGCCATAGAGGTCCTT |
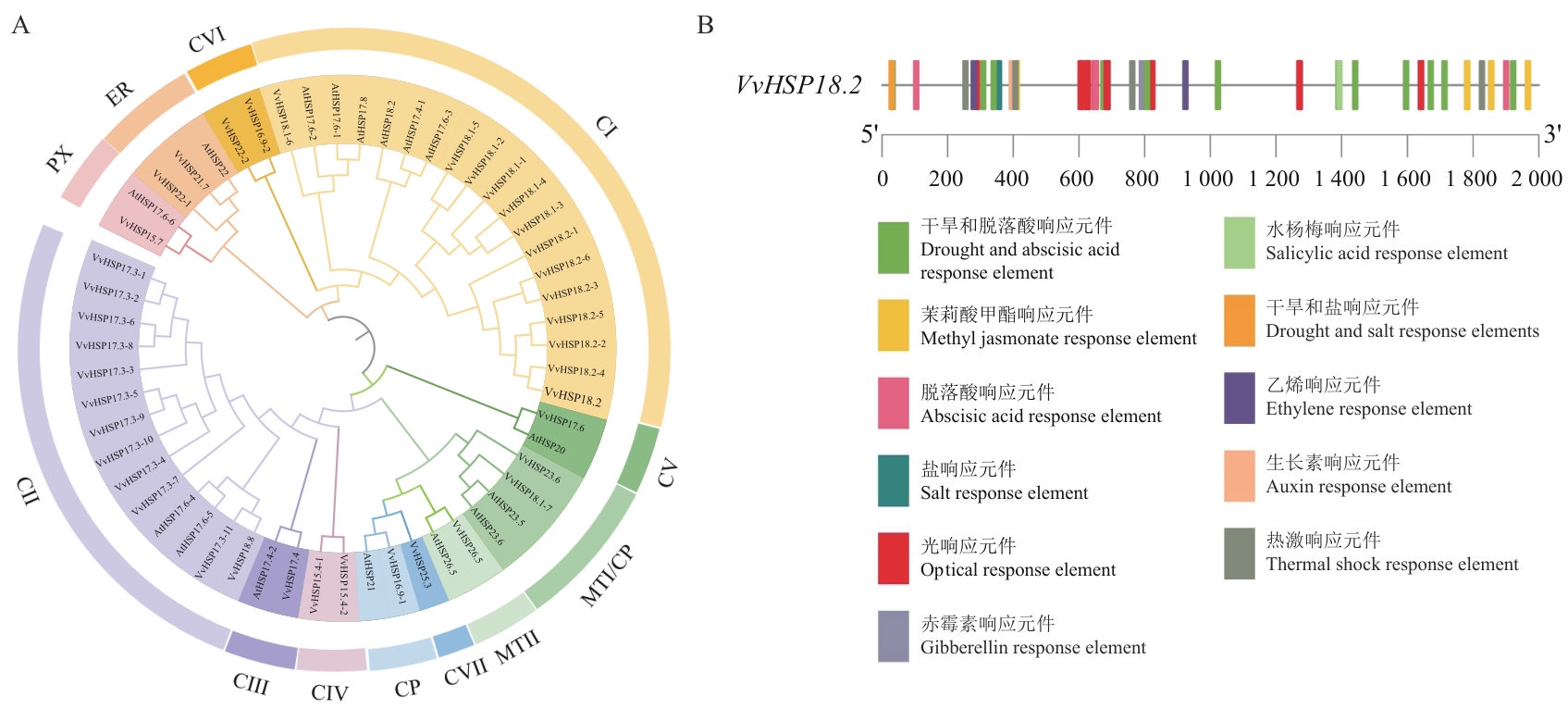
Fig. 1 Phylogenetic evolution and expression analysis of VvHSP18.2A: Phylogenetic tree of HSP proteins in Arabidopsis thaliana (At) and Vitis vinifera (Vv). B: Analysis of the 2 000 bp promoter before VvHSP18.2
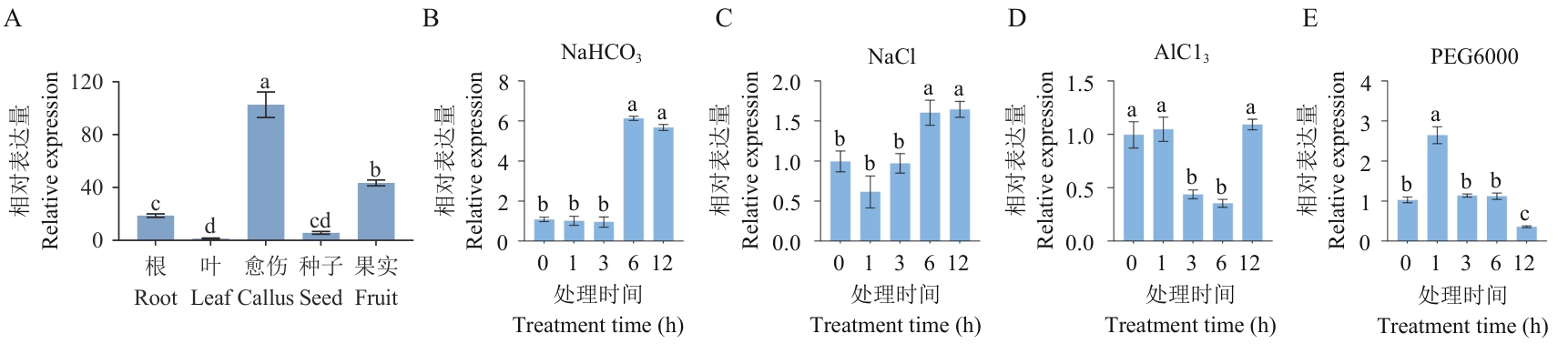
Fig. 2 VvHSP18.2 tissue-specific analysis and abiotic stress response analysisA: VvHSP18.2 tissue-specific analysis. B: NaHCO3 treatment. C: NaCl treatment. D: AlCl3 treatment. E: PEG6000treatment. Different lowercase letters indicate significant differences (P<0.05). The same below
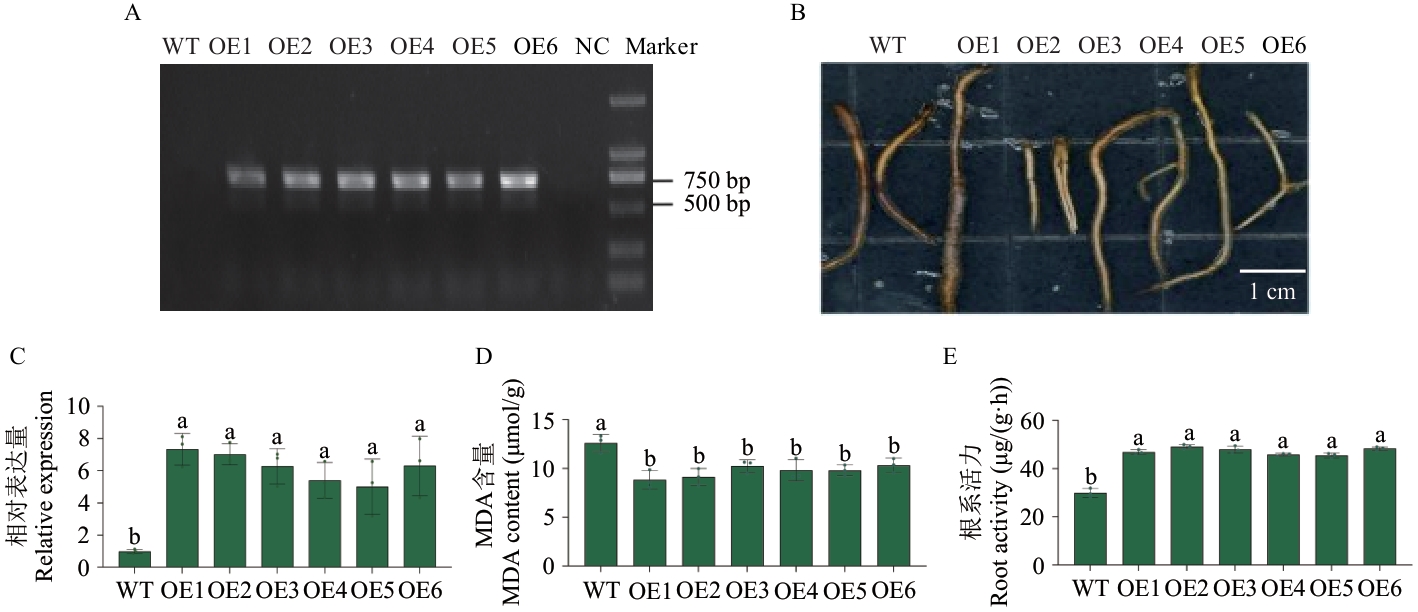
Fig. 3 Overexpression of VvHSP18.2 alleviates alkaline salt stress injury in grapevine rootsA: Root system identificationof VvHSP18.2 transgenic grape (NC: Negative control). B: Phenotype of damage to the root system of genetically modified grapes under alkaline salt stress. C: The expressions of VvHSP18.2 in the root system of genetically modified grapes. D: MDA content. E: Root activity
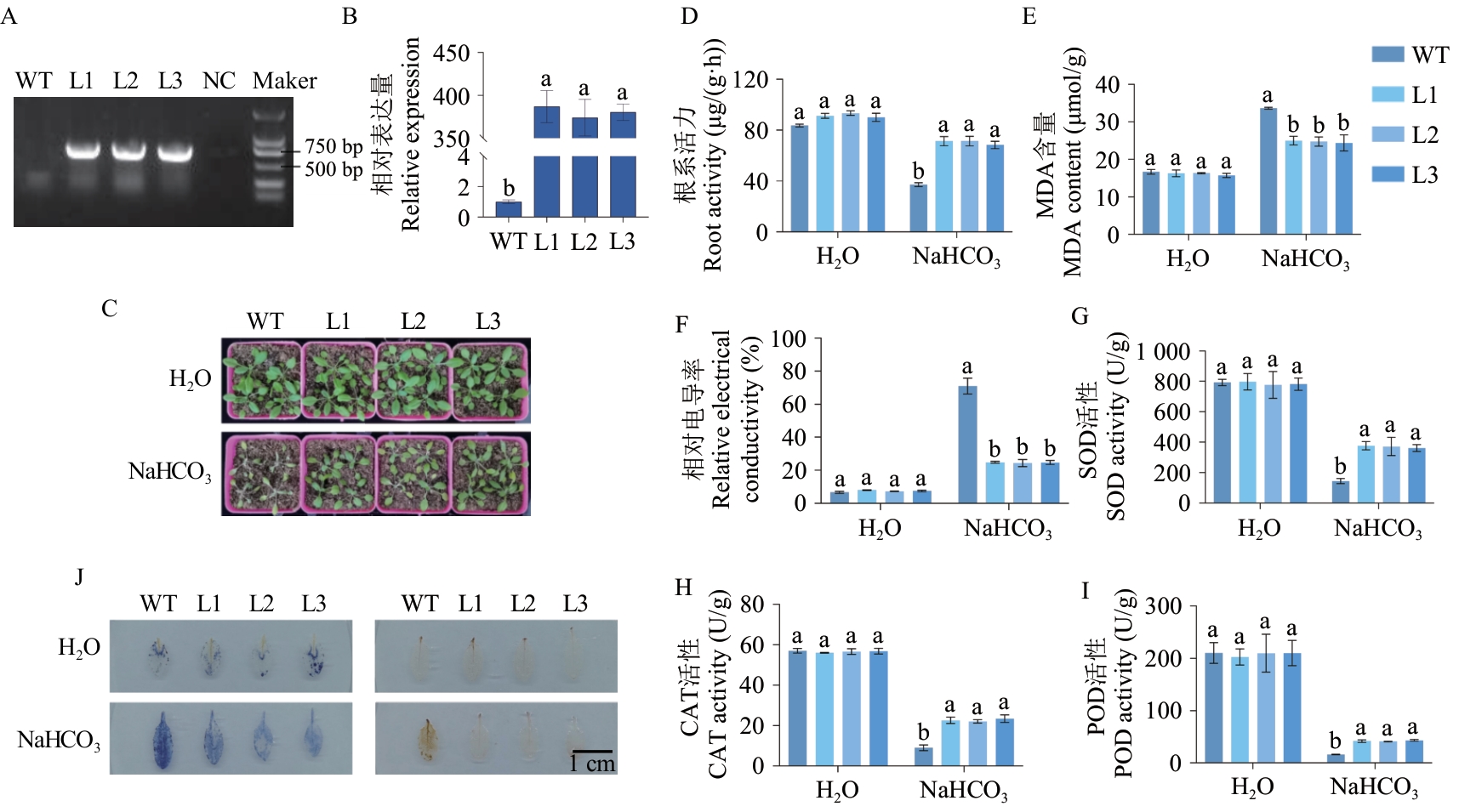
Fig. 4 Identification and overexpression of VvHSP18.2 transgenic Arabidopsis thaliana to enhance Arabidopsis NaHCO3 toleranceA: Identification of VvHSP18.2 transgenic Arabidopsis. B: Expressions of VvHSP18.2 in transgenic Arabidopsis phenotype chart. C: Phenotype chart. D: Root activity content. E: MDA content. F: Relative electrical conductivity. G: SOD activity. H: CAT activity. I: POD activity. J: NBT staining (left) and DAB staining (right). WT: Wild-type Arabidopsisthaliana background. L1, L2, and L3: Three transgenic lines harboring the VvHSP18.2 overexpression construct
Fig. 5 Effect of overexpression of VvHSP18.2 on NaCl resistance in Arabidopsis thalianaA: Phenotypes. B: Fresh weight. C: Root length. D: Number of lateral roots
| [1] | 徐莉, 谢祥恩, 陈毅群. 葡萄抗逆相关转录因子的研究进展[J]. 生物过程, 2024, 14(4): 235-241. |
| Xu L, Xie XE, Chen YQ. Research advances of transcription factors in grape stress resistance [J]. Bioprocess, 2024, 14(4): 235-241. | |
| [2] | 赵秀梅. 葡萄组培苗耐盐筛选技术研究[D]. 杨凌: 西北农林科技大学, 2004. |
| Zhao XM. Study on salt tolerance screening technology of grape tissue culture seedlings [D]. Yangling: Northwest A & F University, 2004. | |
| [3] | 朱自果, 韩燕, 李颖芳, 等. 葡萄耐盐碱性研究进展[J]. 落叶果树, 2024, 56(2): 66-69. |
| Zhu ZG, Han Y, Li YF, et al. Research progress on saline-alkalisalt-alkaline tolerance of grape [J]. Deciduous Fruits, 2024, 56(2): 66-69. | |
| [4] | Wang WX, Vinocur B, Shoseyov O, et al. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response [J]. Trends Plant Sci, 2004, 9(5): 244-252. |
| [5] | Vierling E. The roles of heat shock proteins in plants [J]. Annu Rev Plant Physiol Plant Mol Biol, 1991, 42: 579-620. |
| [6] | Waters ER, Lee GJ, Vierling E. Evolution, structure and function of the small heat shock proteins in plants [J]. J Exp Bot, 1996, 47(3): 325-338. |
| [7] | 张琼, 王锦霞, 孟诗琪, 等. 甜菜热激蛋白基因BvHSP18.2的克隆和生物信息学分析 [J]. 中国农学通报, 2022, 38(27): 111-118. |
| Zhang Q, Wang JX, Meng SQ, et al. Sugar beet heat-shock protein gene BvHSP18.2: cloning and bioinformatics analysis [J]. Chin Agric Sci Bull, 2022, 38(27): 111-118. | |
| [8] | Kim DH, Xu ZY, Hwang I. AtHSP17.8 overexpression in transgenic lettuce gives rise to dehydration and salt stress resistance phenotypes through modulation of ABA-mediated signaling [J]. Plant Cell Rep, 2013, 32(12): 1953-1963. |
| [9] | Wang X, Zhang H, Shao LY, et al. Expression and function analysis of a rice OsHSP40 gene under salt stress [J]. Genes Genomics, 2019, 41(2): 175-182. |
| [10] | Sato Y, Yokoya S. Enhanced tolerance to drought stress in transgenic rice plants overexpressing a small heat-shock protein, sHSP17.7 [J]. Plant Cell Rep, 2008, 27(2): 329-334. |
| [11] | Wang J, Gao X, Dong J, et al. Over-expression of the heat-responsive wheat gene TaHSP23.9 in transgenic Arabidopsis conferred tolerance to heat and salt stress [J]. Front Plant Sci, 2020, 11: 243. |
| [12] | Yang T, Zhang L, Hao HY, et al. Nuclear-localized AtHSPR links abscisic acid-dependent salt tolerance and antioxidant defense in Arabidopsis [J]. Plant J, 2015, 84(6): 1274-1294. |
| [13] | Ma W, Li J, Liu FJ, et al. GhHSP24.7 mediates mitochondrial protein acetylation to regulate stomatal conductance in response to abiotic stress in cotton [J]. Crop J, 2023, 11(4): 1128-1139. |
| [14] | 易小渠. 狗牙根干旱高温胁迫相关基因的克隆和功能解析 [D]. 武汉: 华中农业大学, 2020. |
| Yi XQ. Cloning and functional elucidation of genes related to drought and high temperature stress in canine root [D]. Wuhan: Huazhong Agricultural University, 2020. | |
| [15] | Zhang FJ, Li ZY, Zhang DE, et al. Identification of Hsp20 gene family in Malus domestica and functional characterization of Hsp20 class I gene MdHsp18.2b [J]. Physiol Plant, 2024, 176(2): e14288. |
| [16] | 曹晓蕾. 葡萄MYC2提高碱性盐耐性的分子机制[D]. 泰安: 山东农业大学, 2022. |
| Cao XL. Molecular mechanism of improving alkaline salt tolerance in grape MYC2 [D]. Tai’an: Shandong Agricultural University, 2022. | |
| [17] | Jin ZX, Lv XN, Sun YS, et al. Comprehensive discovery of salt-responsive alternative splicing events based on Iso-Seq and RNA-seq in grapevine roots [J]. Environ Exp Bot, 2021, 192: 104645. |
| [18] | 贾海红. GhMKK9介导的MAPK级联信号途径调控棉花抗枯萎病的分子机理研究 [D]. 泰安: 山东农业大学, 2018. |
| Jia HH. Molecular mechanism of GhMKK9 mediated MAPK cascade signaling pathway in regulating resistance to cotton Fusarium wilt [D]. Tai’an: Shandong Agricultural University, 2018. | |
| [19] | 相广庆. VvERF1B促进草酸分泌提高葡萄根系碱性盐耐性的分子机制 [D]. 泰安: 山东农业大学, 2024. |
| Xiang GQ. Molecular mechanisms underlying VvERF1B promoting oxalate acid secretion and improving alkaline salt tolerance in grapevine roots [D]. Tai’an: Shandong Agricultural University, 2024. | |
| [20] | Velikova V, Yordanov I, Edreva A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants protective role of exogenous polyamines [J]. Plant Sci, 2000, 151(1): 59-66. |
| [21] | 逯明辉, 宋慧, 李晓明, 等. 冷害过程中黄瓜叶片SOD、CAT和POD活性的变化 [J]. 西北植物学报, 2005, 25(8): 1570-1573. |
| Lu MH, Song H, Li XM, et al. Changes of SOD, CAT and POD activities in cucumber leaves during cold damage [J]. Acta Bot Boreali Occidentalia Sin, 2005, 25(8): 1570-1573. | |
| [22] | Zhang M, Shen ZW, Meng GQ, et al. Genome-wide analysis of the Brachypodium distachyon (L.) P. Beauv. Hsp90 gene family reveals molecular evolution and expression profiling under drought and salt stresses [J]. PLoS One, 2017, 12(12): e0189187. |
| [23] | 于洪涛. 斧翅沙芥叶片响应干旱胁迫的转录组学和蛋白质组学分析 [D]. 呼和浩特: 内蒙古农业大学, 2024. |
| Yu HT. Transcriptomic and proteomic analysis of Pugionium dolabratum maxim. leaves responsed to drought stress [D]. Hohhot: Inner Mongolia Agricultural University, 2024. | |
| [24] | Zhao P, Wang D, Wang R, et al. Genome-wide analysis of the potato Hsp20 gene family: identification, genomic organization and expression profiles in response to heat stress [J]. BMC Genomics, 2018, 19(1): 61. |
| [25] | Zhang BS, Liu Z, Zhou RN, et al. Genome-wide analysis of soybean DnaJA-family genes and functional characterization of GmDnaJA6 responses to saline and alkaline stress [J]. Crop J, 2023, 11(4): 1230-1241. |
| [26] | 杨永青, 郭岩. 热激蛋白Hsp17.6CII在调控植物耐盐碱中的应用: CN201611224135.1 [P]. 2020-02-14. |
| Yang YQ, Guo Y. Use of heat shock protein Hsp17.6CII in regulating plant salt-alkali tolerance: CN201611224135.1 [P]. 2020-02-14. | |
| [27] | Zhai MZ, Sun YD, Jia CX, et al. Over-expression of JrsHSP17.3 gene from Juglans regia confer the tolerance to abnormal temperature and NaCl stresses [J]. J Plant Biol, 2016, 59(5): 549-558. |
| [28] | Ni ZY, Liu N, Yu YH, et al. The cotton 70-kDa heat shock protein GhHSP70-26 plays a positive role in the drought stress response [J]. Environ Exp Bot, 2021,191: 104628. |
| [29] | 仲维婷, 张琼, 兴旺, 刘大丽.异源表达甜菜BvHSP18.2基因增强拟南芥对镉胁迫的耐受性研究 [J].中国农学通报, 2024, 40(29): 14-20 |
| Zhong WT, Zhang Q, Xing W, Liu DL. Heterologous expression of sugar beet BvHSP18.2 gene enhances tolerance to cadmium stress in Arabidopsis thaliana [J]. Chinese Agricultural Science Bulletin, 2024, 40(29): 14-20. | |
| [30] | Chang HW, Wu TT, Shalmani A, et al. Heat shock protein HvHSP16.9 from wild barley enhances tolerance to salt stress [J]. Physiol Mol Biol Plants, 2024, 30(5): 687-704. |
| [31] | Lee KW, Cha JY, Kim KH, et al. Overexpression of alfalfa mitochondrial HSP23 in prokaryotic and eukaryotic model systems confers enhanced tolerance to salinity and arsenic stress [J]. Biotechnology Letters, 2012, 34: 167-174. |
| [32] | Wu J, Gao T, Hu J, et al. Research advances in function and regulation mechanisms of plant small heat shock proteins (sHSPs) under environmental stresses [J]. Sci Total Environ, 2022, 15: 825:154054. |
| [33] | Xu JY, Xue CC, Xue D, et al. Overexpression of GmHsp90s, a heat shock protein 90 (Hsp90) gene family cloning from soybean, decrease damage of abiotic stresses in Arabidopsis thaliana [J]. PLoS One, 2013, 8(7): e69810. |
| [34] | Ono K, Hibino T, Kohinata T, et al. Overexpression of DnaK from a halotolerantcyanobacterium aphanothece halophytica enhances the high-temperatue tolerance of tobacco during germination and early growth [J]. Plant Sci, 2001, 160(3): 455-461. |
| [35] | Sung DY, Guy CL. Physiological and molecular assessment of altered expression of Hsp70-1 in Arabidopsis. Evidence for pleiotropic consequences [J]. Plant Physiol, 2003, 132(2): 979-987. |
| [36] | Alvim FC, Carolino SM, Cascardo JC, et al. Enhanced accumulation of BiP in transgenic plants confers tolerance to water stress [J]. Plant Physiol, 2001, 126(3): 1042-1054. |
| [37] | Sugino M, Hibino T, Tanaka Y, et al. Overexpression of DnaK from a halotolerantcyanobacterium aphanothece halophytica acquires resistance to salt stress in transgenic tobacco plants [J]. Plant Sci, 1999, 146(2): 81-88. |
| [38] | Jiang C, Bi Y, Li M, et al. A small heat shock protein gene (RcHSP17.8) from Chinese rose confers resistance to various abiotic stresses in transgenic tobacco [J]. Plant Cell Tissue Organ Cult, 2020, 141 (2), 407-415. |
| [39] | Fu C, Liu X, Yang W, et al. Enhanced salt tolerance in tomato plants constitutively expressing heat-shock protein in the endoplasmic reticulum [J]. Genet Mol Res, 2016, 15(2): gmr.15028301. |
| [40] | Sun J, Cheng G, Huang L, et al. Modified expression of a heat shock protein gene, CaHSP22.0, results in high sensitivity to heat and salt stress in pepper (Capsicum annuum L.) [J]. Sci. Hortic, 2019, 249: 364-373. |
| [41] | Sun XB, Sun CY, Li ZG, et al. AsHSP17, a creeping bentgrass small heat shock protein modulates plant photosynthesis and ABA-dependent and independent signalling to attenuate plant response to abiotic stress [J]. Plant Cell Environ, 2016, 39(6): 1320-1337. |
| [1] | WANG Fang, SHAO Hui-ru, LYU Lin-long, ZHAO Dian, HU Zhen, LYU Jian-zhen, JIANG Liang. Establishment of TurboID Proximity Labeling Technology in Plants and Bacteria [J]. Biotechnology Bulletin, 2025, 41(9): 44-53. |
| [2] | LI Ya-qiong, GESANG La-mao, CHEN Qi-di, YANG Yu-huan, HE Hua-zhuan, ZHAO Yao-fei. Heterologous Overexpression of Sorghum SbSnRK2.1 Enhances the Resistance to Salt Stress in Arabidopsis [J]. Biotechnology Bulletin, 2025, 41(8): 115-123. |
| [3] | FENG Bing, YAN Cai-xia, LIU Yi, DONG Kai-yue, ZHAO Nan, ZHAO Rui, CHEN Shao-liang. Populus × canescens AHL17 Negatively Regulates Tolerance to Cadmium in Arabidopsis thaliana [J]. Biotechnology Bulletin, 2025, 41(6): 269-283. |
| [4] | LIU Tong-tong, LI Xiao-hui, YANG Jun-long, CHEN Wang, YU Meng, WANG Chao-fan, WANG Feng-ru, KE Shao-ying. Functional Study on ZmSTART1 Regulation of Maize Vascular Bundle Formation [J]. Biotechnology Bulletin, 2025, 41(4): 115-122. |
| [5] | YANG Tao, LI Lin, MO Xiao-lian, CHEN Xiao-long, WANG Jian, HUANG Yuan, ZHAO Jie-hong, ZOU Jie. Functional Study of DoDELLA2 in Dendrobium officinale Kimura et Migo [J]. Biotechnology Bulletin, 2025, 41(12): 240-253. |
| [6] | YANG Zong-hui, LI Li-bin, MENG Zhao-juan, GAO Tian, ZHU Li-xia, DU Hai-mei, DONG Wei-wei, CAO Qi-wei. Comparative Transcriptomics Reveals Synergistic Control of Cucumber Sex Determination by Ethylene Signaling and Epigenetic Regulation [J]. Biotechnology Bulletin, 2025, 41(12): 139-155. |
| [7] | WU Zhi-jian, LIU Guang-yang, LIN Zhi-hao, SHENG Bin, CHEN Ge, XU Xiao-min, WANG Jun-wei, XU Dong-hui. Research Progress of Nano-regulation of Vegetable Seed Germination and Its Mechanism [J]. Biotechnology Bulletin, 2025, 41(1): 14-24. |
| [8] | WEN Jing, LI Qian-qian, ZHANG Ming-da, TAN Ming-yue, JIN Bo-yang, SHEN XIU-li, DU Zhi-qiang. Molecular Mechanism of Duox 2 Regulating Innate Immunity against Bacteria in Procambarus clarkii Intestine [J]. Biotechnology Bulletin, 2025, 41(1): 324-332. |
| [9] | CAI Zhi-cheng, WANG Yuan-yuan, SANG Xiao-han, ZENG Li-xian, DENG Wen-tao, WANG Jia-mei. Research Progress of Cold Plasma Activated Solution in Antibacteria and Removing Biofilm [J]. Biotechnology Bulletin, 2024, 40(6): 95-104. |
| [10] | ZHANG Yi-heng, LIU Jia-zheng, WANG Xue-chen, SUN Zheng-zhe, XUE Ya-jun, WANG Pei, HAN Hua, ZHENG Hong-wei, LI Xiao-juan. Dynamic Changes of Arabidopsis Endoplasmic Reticulum Based on Enhanced Super-resolution Images [J]. Biotechnology Bulletin, 2024, 40(4): 67-76. |
| [11] | ZOU Xiu-wei, YUE Jia-ni, LI Zhi-yu, DAI Liang-ying, LI Wei. Functional Analysis of Rice Heat Shock Transcription Factor HsfA2b Regulating the Resistance to Abiotic Stresses [J]. Biotechnology Bulletin, 2024, 40(2): 90-98. |
| [12] | LI Yu, LI Su-zhen, CHEN Ru-mei, LU Hai-qiang. Advances in the Regulation of Iron Homeostasis by bHLH Transcription Factors in Plant [J]. Biotechnology Bulletin, 2023, 39(7): 26-36. |
| [13] | LI Zhi-qi, YUAN Yue, MIAO Rong-qing, PANG Qiu-ying, ZHANG Ai-qin. Melatonin Contents in Eutrema salsugineum and Arabidopsis thaliana Under Salt Stress, and Expression Pattern Analysis of Synthesis Related Genes [J]. Biotechnology Bulletin, 2023, 39(5): 142-151. |
| [14] | ZHOU Heng, XIE Yan-jie. Recent Progress in Oxidative Stress Signaling and Response in Plants [J]. Biotechnology Bulletin, 2023, 39(11): 36-43. |
| [15] | LIN Rong, ZHENG Yue-ping, XU Xue-zhen, LI Dan-dan, ZHENG Zhi-fu. Functional Analysis of ACOL8 Gene in the Ethylene Synthesis and Response in Arabidopsis thaliana [J]. Biotechnology Bulletin, 2023, 39(1): 157-165. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||