








• 研究报告 • 下一篇
张珊1,2( ), 孙志鹏1,2, 赵德刚1,2,3, 赵懿琛1,2,4(
), 孙志鹏1,2, 赵德刚1,2,3, 赵懿琛1,2,4( )
)
收稿日期:2025-09-22
出版日期:2026-03-02
通讯作者:
赵懿琛,女,博士,教授,研究方向 :植物次级信号转导、植物逆境胁迫下的生理生化及植物次生代谢产物生物合成通路;E-mail: yczhao@gzu.edu.cn作者简介:张珊,女,硕士研究生,研究方向 :植物基因工程;E-mail: szsy923@163.com
基金资助:
ZHANG Shan1,2( ), SUN Zhi-peng1,2, ZHAO De-gang1,2,3, ZHAO Yi-chen1,2,4(
), SUN Zhi-peng1,2, ZHAO De-gang1,2,3, ZHAO Yi-chen1,2,4( )
)
Received:2025-09-22
Published:2026-03-02
摘要:
目的 β-1,4-葡聚糖酶(β-1,4-Glucanase, BGL)是一种可水解细胞壁纤维素的酶,能够通过促进砧木与接穗的愈合有效提高嫁接成活率。解析杜仲(Eucommia ulmoides)BGL基因功能,对建立高效嫁接技术体系及深入开展相关基因功能研究具有重要意义。 方法 分别构建由创伤诱导型启动子FAR6和生长素诱导型启动子GH3驱动的BGL基因植物表达载体,并通过遗传转化杜仲获得转基因植株。利用微嫁接技术比较不同接穗和砧木组合的成活率,采用RT-qPCR分析伤口愈合相关基因的表达,并通过解剖学观察评估嫁接部位愈伤组织形成和愈合情况。 结果 RT-qPCR结果表明,转FAR6::BGL植株中BGL基因受创伤诱导表达,而转GH3::BGL植株中BGL基因受生长素诱导表达。微嫁接实验显示,转FAR6::BGL接穗的成活率(63.33%)显著高于转GH3::BGL接穗(53.33%)和野生型接穗(33.33%)。以转FAR6::BGL植株为砧木时,与野生型接穗的嫁接成活率达80%,显著高于野生型砧木(30%)。此外,转FAR6::BGL砧木可促进伤口愈合相关基因(EuERF115-like、EuNAC1和EuWOX13-2)的表达。解剖学观察表明,其嫁接部位愈伤组织形成更快,伤口愈合时间更短。 结论 转FAR6::BGL杜仲砧木能显著提高杜仲微嫁接成活率,促进伤口愈合相关基因的表达,加速嫁接部位愈合过程,为克服转基因植株生根困难及移栽成活率低等问题提供新策略。
张珊, 孙志鹏, 赵德刚, 赵懿琛. 杜仲BGL基因创制高效嫁接砧木[J]. 生物技术通报, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1002.
ZHANG Shan, SUN Zhi-peng, ZHAO De-gang, ZHAO Yi-chen. Eucommia ulmoides BGL Gene for Developing Highly Efficient Grafting Rootstocks[J]. Biotechnology Bulletin, doi: 10.13560/j.cnki.biotech.bull.1985.2025-1002.
| 引物名称 Primer name | 引物序列 Primer sequence (5′-3′) |
|---|---|
| GUS-F | GGTGATTGATGAAACTGCTG |
| GUS-R | GAACATTACATTGACGCAGG |
| β-1,4--F | ATGGGTTCTCCACTTACACAGTC |
| β-1,4--R | TTAAGGTGCTAAATGGATATTCTTG |
| EuActin-F | GTGTTATGGTTGGGATGGG |
| EuActin-R | TGCTGACTATGCCGTGTTC |
| β-1,4-qRT-F | GGTTTGGAGACCACTACCCG |
| β-1,4-qRT-R | GTGTGTTTGGGTTGGGTTCG |
| EuERF115-like-qRT-F | CATCGTCATCGTCATCATC |
| EuERF115-like-qRT-R | ACTAAGGTGGTCCATTGAG |
| EuNAC1-qRT-F | AAGGATCAGTTGGAGCTGCC |
| EuNAC1-qRT-R | AGACATTCCTGGAAGCTGCC |
| EuWOX13-2-qRT-F | GGTCTGAGGGCATGTGTTTT |
| EuWOX13-2-qRT-R | TTGGAGATATGGGTGGTGGT |
表1 引物序列
Table 1 Primer sequences
| 引物名称 Primer name | 引物序列 Primer sequence (5′-3′) |
|---|---|
| GUS-F | GGTGATTGATGAAACTGCTG |
| GUS-R | GAACATTACATTGACGCAGG |
| β-1,4--F | ATGGGTTCTCCACTTACACAGTC |
| β-1,4--R | TTAAGGTGCTAAATGGATATTCTTG |
| EuActin-F | GTGTTATGGTTGGGATGGG |
| EuActin-R | TGCTGACTATGCCGTGTTC |
| β-1,4-qRT-F | GGTTTGGAGACCACTACCCG |
| β-1,4-qRT-R | GTGTGTTTGGGTTGGGTTCG |
| EuERF115-like-qRT-F | CATCGTCATCGTCATCATC |
| EuERF115-like-qRT-R | ACTAAGGTGGTCCATTGAG |
| EuNAC1-qRT-F | AAGGATCAGTTGGAGCTGCC |
| EuNAC1-qRT-R | AGACATTCCTGGAAGCTGCC |
| EuWOX13-2-qRT-F | GGTCTGAGGGCATGTGTTTT |
| EuWOX13-2-qRT-R | TTGGAGATATGGGTGGTGGT |
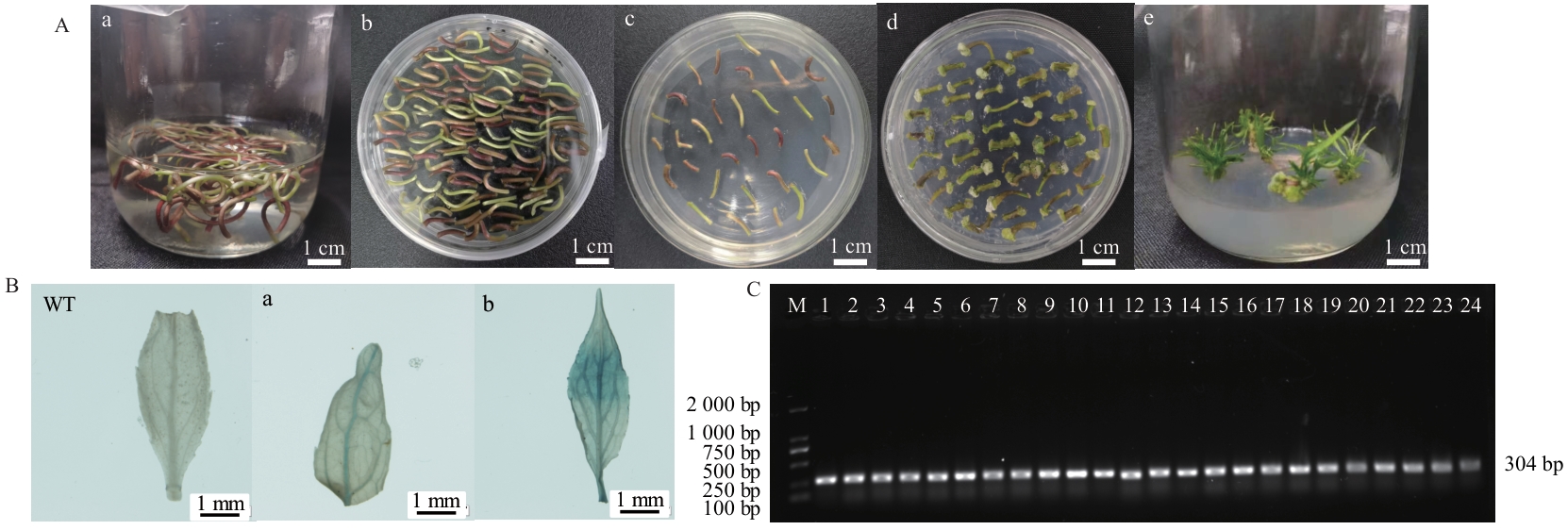
图1 转基因杜仲抗性芽的获得与鉴定A:杜仲遗传转化流程图(a:悬浮培养;b:共培养;c:筛选培养;d:诱导愈伤;e:抗性芽);B:转基因杜仲GUS检测(WT:野生型杜仲;a:转FAR6::BGL杜仲;b:转GH3::BGL杜仲);C:转基因杜仲PCR扩增鉴定(GUS基因:304 bp),M:DNA DL 2 000 marker;1-12:转FAR6::BGL植株;13-24:转GH3::BGL植株
Fig. 1 Acquisition and identification of transgenic E. ulmoides resistant budsA: E. ulmoides genetic transformation flow chart (a: Suspension culture. b: Co-culture. c: Screening culture. d: Induced callus. e: Resistant bud). B: GUS detection of transgenic E. ulmoides (WT: Wild type E. ulmoides. a: FAR6::BGLE. ulmoides. b: GH3::BGLE. ulmoides). C: PCR amplification of transgenic E. ulmoides (GUS gene:304 bp). M: DNA DL2 000 marker. 1-12: Transfer FAR6::BGL plants. 13-24: Transfer GH3::BGL plants
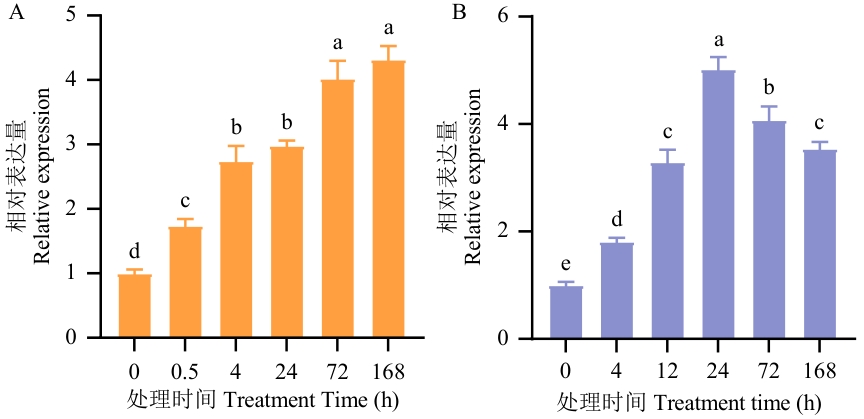
图2 杜仲中BGL基因的相对表达量分析A:创伤处理;B:10 μmol/L IAA处理;不同小写字母表示在P<0.05水平差异显著。下同
Fig. 2 Analysis of the relative expression of BGL gene in E. ulmoidesA: Injury treatment; B:10 μmol/L IAA treatment. Different lower letters indicate significant differences at P<0.05 level. The same below
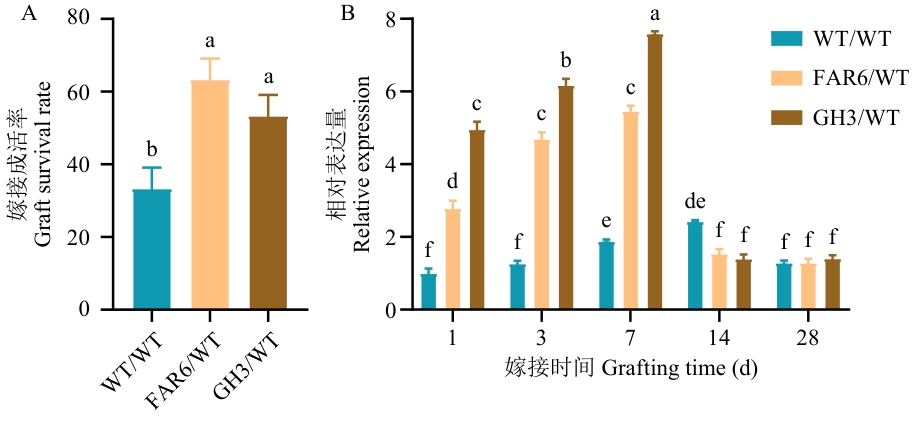
图3 嫁接成活率统计(A)及BGL基因表达量分析(B)WT:野生型杜仲;FAR6:转FAR6::BGL杜仲;GH3:转GH3::BGL杜仲。下同
Fig. 3 Statistics of graft survival rate (A) and analysis of BGL gene expression (B)WT: Wild type E. ulmoides; FAR6: FAR6::BGLE. ulmoides; GH3: GH3::BGLE. ulmoides. The same below

图4 转基因杜仲砧木的获得与鉴定A:农杆菌注射法遗传转化杜仲流程图(a:杜仲幼苗;b:拔除胚芽的杜仲幼苗;c:对杜仲幼苗注射菌液;d:转化15 d后的杜仲幼苗;e:转化2个月的杜仲幼苗);B:转基因杜仲GUS检测(WT:野生型杜仲;TP:转FAR6::BGL杜仲);C:转基因杜仲PCR扩增鉴定(GUS基因:304 bp),M:DNA DL 2 000 marker;1-10:转FAR6::BGL植株
Fig. 4 Acquisition and identification of transgenic E. ulmoides rootstocksA: Flow chart of genetic transformation of E. ulmoides by Agrobacterium injection (a: E. ulmoides seedlings. b: Remove the germ of E. ulmoides seedlings. c: Inject bacteria solution into E. ulmoides seedlings. d: E. ulmoides seedlings after 15 d of transformation. e: Converted E. ulmoides seedlings for two months). B: GUS detection of transgenic E. ulmoides (WT: Wild type E. ulmoides. TP: FAR6::BGLE. ulmoides). C: PCR amplification of transgenic E. ulmoides (GUS gene:304 bp) M: DNA DL 2 000 marker. 1-10: Transferring FAR6::BGL plant
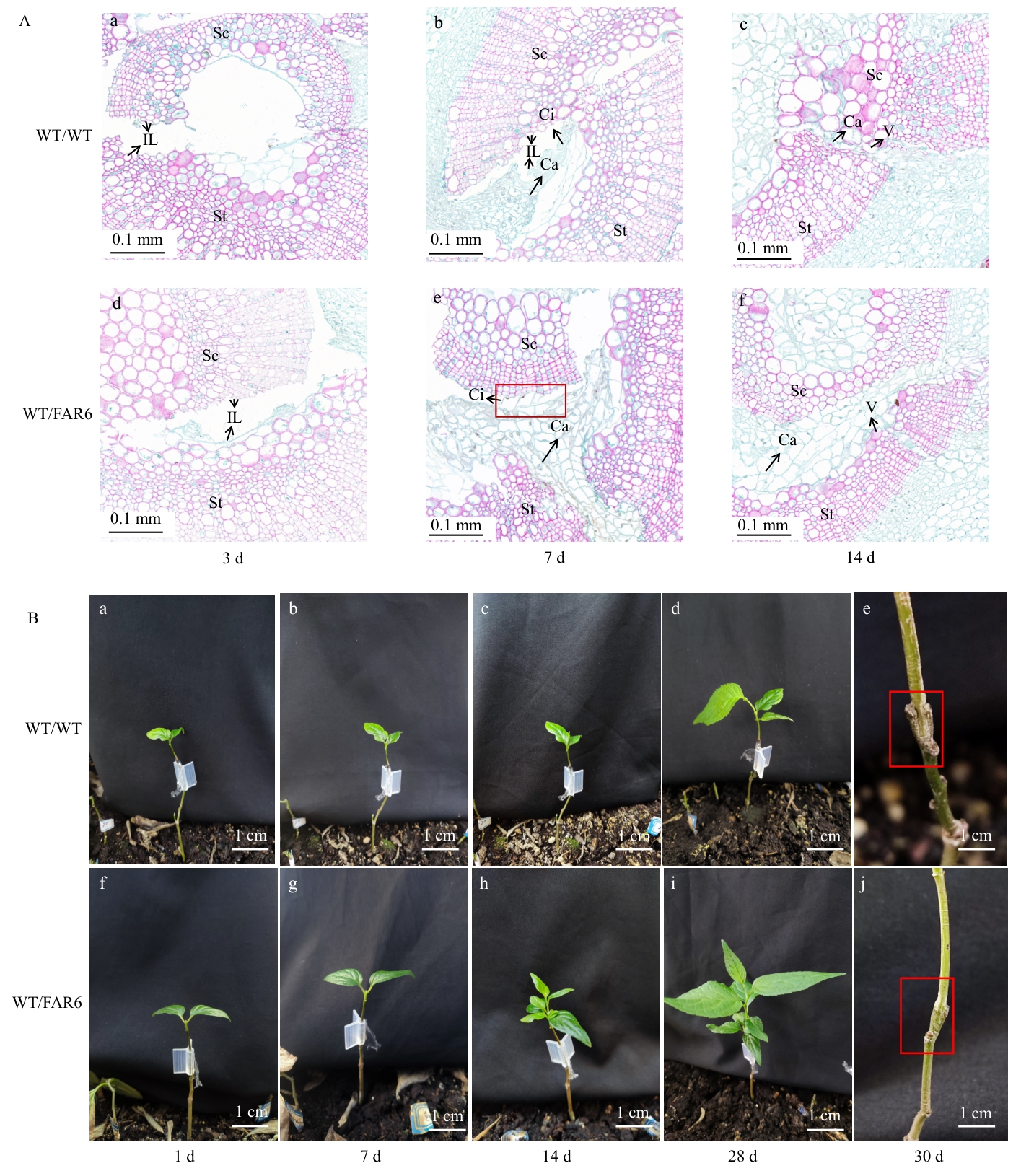
图6 转基因杜仲作砧木嫁接愈合过程解剖学(A)和外观形态学(B)观察St:砧木;Sc:接穗;IL:隔离层;Ci:愈伤组织;Ca:形成层;V:维管射线
Fig. 6 Observation of the anatomy (A) and appearance and morphology (B) of the grafting and healing process of transgenic E. ulmoides rootstockSt: Rootstock. Sc: Scion. IL: Isolation layer. Ci: Callus. Ca: Cambium. V: Vascular
砧木类别 Rootstock type | 嫁接株数 Number of grafis (plants) | 存活株数 Number of survivors (plants) | 成活率 Survival rate (%) |
|---|---|---|---|
野生型杜仲 WT E. ulmoides | 10 | 3 | 30 |
转FAR6::BGL杜仲 FAR6::BGL E. ulmoides | 10 | 8 | 80 |
表2 不同杜仲砧木的微嫁接成活率
Table 2 Survival rate of E. ulmoides micrografting of different rootstocks
砧木类别 Rootstock type | 嫁接株数 Number of grafis (plants) | 存活株数 Number of survivors (plants) | 成活率 Survival rate (%) |
|---|---|---|---|
野生型杜仲 WT E. ulmoides | 10 | 3 | 30 |
转FAR6::BGL杜仲 FAR6::BGL E. ulmoides | 10 | 8 | 80 |
| [1] | Qian CJ, Zhang RR, Li J, et al. The characteristics of habitat, functional traits and medicinal components of Eucommia ulmoides from Guizhou [J]. Environ Sci Pollut Res Int, 2022, 29(9): 12629-12647. |
| [2] | Qing J, Meng YD, He F, et al. Whole genome re-sequencing reveals the genetic diversity and evolutionary patterns of Eucommia ulmoides [J]. Mol Genet Genomics, 2022, 297(2): 485-494. |
| [3] | Du QX, Wu ZX, Liu PF, et al. The chromosome-level genome of Eucommia ulmoides provides insights into sex differentiation and α-linolenic acid biosynthesis [J]. Front Plant Sci, 2023, 14: 1118363. |
| [4] | 赵丹, 赵德刚, 李岩. EuFPS基因表达载体构建及对杜仲遗传转化的研究 [J]. 基因组学与应用生物学, 2009, 28(1): 27-33. |
| Zhao D, Zhao DG, Li Y. Studies on construction of EuFPS gene plant expression vector and Agrobacterium-mediated genetic transformation of Eucommia ulmoides oliv [J]. Genom Appl Biol, 2009, 28(1): 27-33. | |
| [5] | 王磊. 杜仲组培苗微嫁接技术研究 [D]. 贵阳: 贵州大学, 2023. |
| Wang L. Study on micrografting technology of Eucommia ulmoides oliver tissue culture plantlets [D]. Guiyang: Guizhou University, 2023. | |
| [6] | 陈晶晶, 李栋梁, 杨倩, 等. 植物嫁接再生机理研究进展 [J]. 植物生理学报, 2020, 56(8): 1690-1702. |
| Chen JJ, Li DL, Yang Q, et al. Research advances in the mechanism of plant graft regeneration [J]. Plant Physiol J, 2020, 56(8): 1690-1702. | |
| [7] | 范玉龙, 李兴群, 宋金修, 等. 蔬菜嫁接愈合过程及其光环境调控机制研究进展 [J]. 蔬菜, 2022(8): 42-48. |
| Fan YL, Li XQ, Song JX, et al. Research progress on grafting healing process and light environment regulation mechanism of vegetables [J]. Vegetables, 2022(8): 42-48. | |
| [8] | Nanda AK, Melnyk CW. The role of plant hormones during grafting [J]. J Plant Res, 2018, 131(1): 49-58. |
| [9] | Melnyk CW, Meyerowitz EM. Plant grafting [J]. Curr Biol, 2015, 25(5): R183-R188. |
| [10] | 田敏娇, 李乐, 李建设, 等. 土壤改良剂和嫁接栽培对日光温室土壤性状与番茄品质产量的影响 [J]. 西南农业学报, 2020, 33(11): 2577-2583. |
| Tian MJ, Li L, Li JS, et al. Effects of soil amendment and graft cultivation on soil and tomato yield and quality in solar greenhouse [J]. Southwest China J Agric Sci, 2020, 33(11): 2577-2583. | |
| [11] | 周洲. 不同中间砧长度对桃树营养生长和开花的影响 [J]. 中国果业信息, 2021, 38(6): 51. |
| Zhou Z. Effects of different interstock lengths on vegetative growth and flowering of peach trees [J]. China Fruit News, 2021, 38(6): 51. | |
| [12] | 付深造, 任君, 王鑫, 等. 南瓜砧木嫁接对黄瓜品种DUS测试基本性状和果实品质指标的影响 [J]. 园艺学报, 2023, 50(12): 2665-2679. |
| Fu SZ, Ren J, Wang X, et al. Effects of pumpkin rootstock grafting on the expressions of basic DUS characteristics and fruit quality indicators in cucumber [J]. Acta Hortic Sin, 2023, 50(12): 2665-2679. | |
| [13] | 朱瑜, 周信雁, 魏宇飞, 等. 嫁接对番茄植株根际微生物群落组成的影响 [J]. 西南农业学报, 2024, 37(1): 199-209. |
| Zhu Y, Zhou XY, Wei YF, et al. Effects of grafting on soil microbial community compositions in rhizospheres of tomatoes [J]. Southwest China J Agric Sci, 2024, 37(1): 199-209. | |
| [14] | 刘益勇, 周亚东, 申磊, 等. 嫁接对茄子耐冷性的影响 [J]. 安徽农业科学, 2022, 50(13): 52-55, 58. |
| Liu YY, Zhou YD, Shen L, et al. Effect of grafting on cold tolerance of eggplant [J]. J Anhui Agric Sci, 2022, 50(13): 52-55, 58. | |
| [15] | Chai SM, Zhang XL, Gao YJ, et al. Identification and characterization of a novel endo-β-1, 4-glucanase from a soil metagenomic library [J]. Carbohydr Res, 2021, 510: 108460. |
| [16] | Goodridge HS, Wolf AJ, Underhill DM. β-glucan recognition by the innate immune system [J]. Immunol Rev, 2009, 230(1): 38-50. |
| [17] | Kurotani KI, Wakatake T, Ichihashi Y, et al. Host-parasite tissue adhesion by a secreted type of β-1, 4-glucanase in the parasitic plant Phtheirospermum japonicum [J]. Commun Biol, 2020, 3(1): 407. |
| [18] | Kurotani KI, Huang CK, Okayasu K, et al. Interfamily grafting capacity of Petunia [J]. Hortic Res, 2022, 9: uhab056. |
| [19] | 王润英. 杜仲β-1,4葡聚糖酶基因克隆及酵母工程菌的制备 [D]. 贵阳: 贵州大学, 2022. |
| Wang RY. Cloning of Eucommia ulmoides oliver β-1,4 glucanase gene and preparation of yeast engineering bacteria [D]. Guiyang: Guizhou University, 2022. | |
| [20] | 周舒婷, 董旋, 赵懿琛, 等. 农杆菌介导EuABP2基因遗传转化杜仲下胚轴研究 [J]. 山地农业生物学报, 2018, 37(3): 33-39. |
| Zhou ST, Dong X, Zhao YC, et al. Genetic transformation of EuABP2 gene into the hypocotyl of Eucommia ulmoides oliv. [garryales: Eucommiaceae] via Agrobacterium-mediated method [J]. J Mt Agric Biol, 2018, 37(3): 33-39. | |
| [21] | 王玲, 董旋, 谭艾娟, 等. 转基因杜仲再生体系的优化 [J]. 种子, 2019, 38(5): 18-22, 27. |
| Wang L, Dong X, Tan AJ, et al. Optimization of regeneration system of transgenic Eucommia ulmoides oliver [J]. Seed, 2019, 38(5): 18-22, 27. | |
| [22] | 王超. 杜仲直接转化法研究 [D]. 贵阳: 贵州大学, 2021. |
| Wang C. Study on the direct transformation of Eucommia ulmoides oliver [D]. Guiyang, Guizhou University, 2021. | |
| [23] | Pedersen GB, Blaschek L, Frandsen KEH, et al. Cellulose synthesis in land plants [J]. Mol Plant, 2023, 16(1): 206-231. |
| [24] | Yang L, Chen Y, Liu XJ, et al. Genome-wide identification and expression analysis of xyloglucan endotransglucosylase/hydrolase genes family in Salicaceae during grafting [J]. BMC Genomics, 2023, 24(1): 676. |
| [25] | 胡瑞学. 玉米干旱诱导型基因启动子的克隆及功能分析 [D]. 长春: 吉林大学, 2011. |
| Hu RX. Cloning and function analysis of the promoters of drought-induced genes from maize [D]. Changchun: Jilin University, 2011. | |
| [26] | Jeong HJ, Jung KH. Rice tissue-specific promoters and condition-dependent promoters for effective translational application [J]. J Integr Plant Biol, 2015, 57(11): 913-924. |
| [27] | Nakashima K, Tran LP, Van Nguyen D, et al. Functional analysis of a NAC-type transcription factor OsNAC6 involved in abiotic and biotic stress-responsive gene expression in rice [J]. Plant J, 2007, 51(4): 617-630. |
| [28] | Wang L, Wang RY, Li Y, et al. Overexpression of β-1,4-glucanase gene EuEG1 improves micrografting of Eucommia ulmoides [J]. Phyton Int J Exp Bot, 2023, 92(11): 3063-3075. |
| [29] | Xu DB, Yang Y, Tao SC, et al. Identification and expression analysis of auxin-responsive GH3 family genes in Chinese hickory (Carya cathayensis) during grafting [J]. Mol Biol Rep, 2020, 47(6): 4495-4506. |
| [30] | Feng M, Zhang A, Nguyen V, et al. A conserved graft formation process in Norway spruce and Arabidopsis identifies the PAT gene family as central regulators of wound healing [J]. Nat Plants, 2024, 10(1): 53-65. |
| [31] | Zhang A, Matsuoka K, Kareem A, et al. Cell-wall damage activates DOF transcription factors to promote wound healing and tissue regeneration in Arabidopsis thaliana [J]. Curr Biol, 2022, 32(9): 1883-1894.e7. |
| [32] | 陈铭秋, 刘果, 林彦, 等. 木本植物组织培养及器官从头再生的研究进展 [J]. 桉树科技, 2023, 40(4): 85-96. |
| Chen MQ, Liu G, Lin Y, et al. Research progress in tissue culture and de novo organ regeneration of woody plants [J]. Eucalypt Sci Technol, 2023, 40(4): 85-96. | |
| [33] | 蔡方阳. 杜仲EuDIR4基因克隆及其对植物抗逆性影响研究 [D]. 贵阳: 贵州大学, 2021. |
| Cai FY. Cloning of EuDIR4 gene from Eucommia ulmoides and its effect on plant stress resistance [D]. Guiyang: Guizhou University, 2021. | |
| [34] | 李兰君. 杜仲几丁质酶基因EuCHIT30.7克隆及功能分析 [D]. 贵阳: 贵州大学, 2023. |
| Li LJ. Cloning and functional analysis of chitinase gene EuCHIT30.7 in Eucommia ulmoides [D]. Guiyang: Guizhou University, 2023. |
| [1] | 李正, 邱炜玥, 孙瑞雪, 赵筱. 不同启动子过表达xylR基因提高大肠杆菌的葡萄糖-木糖共利用能力[J]. 生物技术通报, 2026, 42(1): 329-337. |
| [2] | 张驰昊, 刘晋囡, 晁跃辉. 蒺藜苜蓿bZIP转录因子MtbZIP29的克隆及功能分析[J]. 生物技术通报, 2026, 42(1): 241-250. |
| [3] | 程婷婷, 刘俊, 王利丽, 练从龙, 魏文君, 郭辉, 吴尧琳, 杨晶凡, 兰金旭, 陈随清. 杜仲查尔酮异构酶基因家族全基因组鉴定及其表达模式分析[J]. 生物技术通报, 2025, 41(9): 242-255. |
| [4] | 付博晗, 毛华, 赵薪程, 陆虹, 欧庸彬, 姚银安. 不同杨树SOS1基因启动子的克隆及盐胁迫响应分析[J]. 生物技术通报, 2025, 41(7): 205-213. |
| [5] | 张泽, 杨秀丽, 宁东贤. 花生4CL基因家族鉴定及对干旱与盐胁迫响应分析[J]. 生物技术通报, 2025, 41(7): 117-127. |
| [6] | 梁丽存, 王克芬, 宋祖洹, 刘梦婷, 李佳玉, 罗会颖, 姚斌, 杨浩萌. 优化sgRNA提高塔宾曲霉基因编辑效率[J]. 生物技术通报, 2025, 41(3): 62-70. |
| [7] | 林紫依, 吴一舟, 叶芳贤, 朱淑颖, 刘燕敏, 刘骕骦. 大豆GmPM31基因启动子响应高温高湿胁迫的功能分析[J]. 生物技术通报, 2025, 41(3): 90-97. |
| [8] | 薛晓斌, 宁琳, 周鱼, 刘虹君, 高照祖, 王振平, 李栋梅. 酿酒葡萄VvOMTs基因家族鉴定及启动子功能分析[J]. 生物技术通报, 2025, 41(12): 168-176. |
| [9] | 王若若, 曲鹏坤, 张欣, 王珞, 朱英, 田双一, 赵德刚. 杜仲EuGIF1基因鉴定及其表达和蛋白互作分析[J]. 生物技术通报, 2025, 41(12): 267-279. |
| [10] | 张静安, 胡孝龙, 曹蓓蓓, 廖敏, 束长龙, 张杰, 王奎, 操海群. 苏云金芽胞杆菌可视化快速表达载体的构建与特性分析[J]. 生物技术通报, 2025, 41(1): 95-102. |
| [11] | 毋舒宁, 苏永平, 李冬雪, 柏映国, 刘波, 张志伟. 一种谷氨酸棒杆菌4-异丙基苯甲酸诱导型启动子的设计与应用[J]. 生物技术通报, 2024, 40(7): 108-116. |
| [12] | 李梦然, 叶伟, 李赛妮, 张维阳, 李建军, 章卫民. Lithocarols类化合物生物合成基因litI的表达及其启动子功能分析[J]. 生物技术通报, 2024, 40(6): 310-318. |
| [13] | 王周, 余杰, 王金华, 王永泽, 赵筱. 厌氧表达乳酸脱氢酶以提高大肠杆菌产D-乳酸光学纯度[J]. 生物技术通报, 2024, 40(5): 290-299. |
| [14] | 张清兰, 张亚冉, 鞠志花, 王秀革, 肖遥, 王金鹏, 魏晓超, 高亚平, 白福恒, 王洪程. 牛TARDBP基因核心启动子鉴定与转录调控分析[J]. 生物技术通报, 2024, 40(4): 306-318. |
| [15] | 刘玉萍, 张维阳, 章卫民, 叶伟, 李冬利. Phomopsis tersa FS441聚酮杂萜类化合物生物合成基因启动子的鉴定[J]. 生物技术通报, 2024, 40(12): 248-255. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||